Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [2]
- Flow cytometry [3]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-12484 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD235a Monoclonal Antibody (JC159)
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- JC159
- Vial size
- 500 µL
- Concentration
- 0.2 mg/mL
- Storage
- 4° C
Submitted references Erythrocytic α-synuclein contained in microvesicles regulates astrocytic glutamate homeostasis: a new perspective on Parkinson's disease pathogenesis.
Sustained maternal smoking-associated changes in the physico-chemical properties of fetal RBC membranes might serve as early markers for vascular comorbidities.
Transmission of α-synuclein-containing erythrocyte-derived extracellular vesicles across the blood-brain barrier via adsorptive mediated transcytosis: another mechanism for initiation and progression of Parkinson's disease?
Elevated levels of macromolecular damage are correlated with increased nitric oxide synthase expression in erythrocytes isolated from twin neonates.
Histopathological, immunophenotypic and clinical particularities and evolution of a case of hepatosplenic T-cell lymphoma in transformation to leukemia.
Sheng L, Stewart T, Yang D, Thorland E, Soltys D, Aro P, Khrisat T, Xie Z, Li N, Liu Z, Tian C, Bercow M, Matsumoto J, Zabetian CP, Peskind E, Quinn JF, Shi M, Zhang J
Acta neuropathologica communications 2020 Jul 8;8(1):102
Acta neuropathologica communications 2020 Jul 8;8(1):102
Sustained maternal smoking-associated changes in the physico-chemical properties of fetal RBC membranes might serve as early markers for vascular comorbidities.
Balogh G, Chakraborty P, Dugmonits KN, Péter M, Végh AG, Vígh L, Hermesz E
Biochimica et biophysica acta. Molecular and cell biology of lipids 2020 Apr;1865(4):158615
Biochimica et biophysica acta. Molecular and cell biology of lipids 2020 Apr;1865(4):158615
Transmission of α-synuclein-containing erythrocyte-derived extracellular vesicles across the blood-brain barrier via adsorptive mediated transcytosis: another mechanism for initiation and progression of Parkinson's disease?
Matsumoto J, Stewart T, Sheng L, Li N, Bullock K, Song N, Shi M, Banks WA, Zhang J
Acta neuropathologica communications 2017 Sep 13;5(1):71
Acta neuropathologica communications 2017 Sep 13;5(1):71
Elevated levels of macromolecular damage are correlated with increased nitric oxide synthase expression in erythrocytes isolated from twin neonates.
Dugmonits KN, Ferencz Á, Zahorán S, Lázár R, Talapka P, Orvos H, Hermesz E
British journal of haematology 2016 Sep;174(6):932-41
British journal of haematology 2016 Sep;174(6):932-41
Histopathological, immunophenotypic and clinical particularities and evolution of a case of hepatosplenic T-cell lymphoma in transformation to leukemia.
Benedek Lázár E, Köpeczi JB, Tunyogi AB, Kakucs E, Horváth E, Turcu M, Benedek I
Romanian journal of morphology and embryology = Revue roumaine de morphologie et embryologie 2013;54(4):1135-9
Romanian journal of morphology and embryology = Revue roumaine de morphologie et embryologie 2013;54(4):1135-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
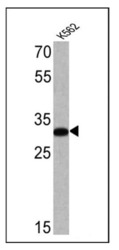
- Experimental details
- Western blot analysis of Glycophorin A was performed by loading 25 µg of K562 cell lysate onto an SDS polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked at 4ºC overnight. The membrane was probed with a Glycophorin A monoclonal antibody (Product # MA5-12484) at a dilution of 1:100 overnight at 4°C, washed in TBST, and probed with an HRP-conjugated secondary antibody for 1 hr at room temperature in the dark. Chemiluminescent detection was performed using Pierce ECL Plus Western Blotting Substrate (Product # 32132). Results show a band at ~32 kDa.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
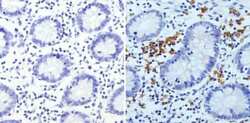
- Experimental details
- Immunohistochemistry analysis of Glycophorin A showing staining in the membrane of paraffin-embedded human colon tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Glycophorin A monoclonal antibody (Product # MA5-12484) diluted in 3% BSA-PBS at a dilution of 1:200 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
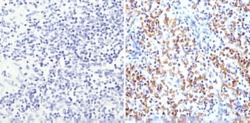
- Experimental details
- Immunohistochemistry analysis of Glycophorin A showing staining in the membrane of paraffin-embedded human spleen tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Glycophorin A monoclonal antibody (Product # MA5-12484) diluted in 3% BSA-PBS at a dilution of 1:200 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
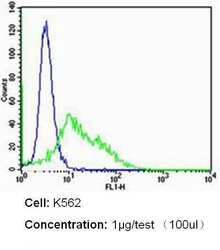
- Experimental details
- Flow cytometry analysis of CD235a/Glycophorin A in K562 cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD235a/Glycophorin A monoclonal antibody (Product # MA5-12484) at a dilution of 1 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
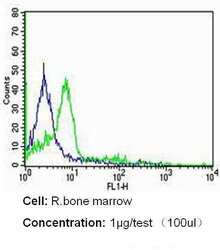
- Experimental details
- Flow cytometry analysis of CD235a/Glycophorin A in rat bone marrow cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD235a/Glycophorin A monoclonal antibody (Product # MA5-12484) at a dilution of 1 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Flow cytometry analysis of CD235a/Glycophorin A in rat bone marrow cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD235a/Glycophorin A monoclonal antibody (Product # MA5-12484) at a dilution of 1 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 The characterization of extracellular vesicles from packed red blood cells (RBC-EVs). SEC was used to separate EVs (peak 1) from low molecular weight protein contaminants [peak 2; co-eluted with BSA (66 kDa)]. The presence of (EV surface) proteins was monitored by UV absorbance at 280 nm ( a ), and the number of particles in each fraction was further measured by NTA ( b ). The total particle number of RBC-EVs ( c ) and the distribution of RBC-EVs ( d ) were measured in each peak by NTA (Nanosight NC300). Representative images of Western blots showing Alix and CD235a in RBC-EVs and RBC cell lysate e Three independently cultured and prepared samples of RBC lysate and EVs (50 mug of total proteins in each sample) were used for western blot analysis and were tested by immunoblot with antibodies against Alix (exosome marker) and CD235a (RBC marker). The alpha-syn level in RBC-EVs was measured using Luminex immunoassay ( f ), compared to the alpha-syn level in RBC cell lysates. Values are means +- S.E.M, n = 3
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot