Antibody data
- Antibody Data
- Antigen structure
- References [10]
- Comments [0]
- Validations
- Immunohistochemistry [3]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA3-945 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Calpastatin Monoclonal Antibody (2G11D6)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- MA3-945 detects calpastatin from bovine, human, canine, pig, rabbit, and rat tissues and cells. This antibody does not cross-react with calpains or calmodulin.
- Antibody clone number
- 2G11D6
- Concentration
- Conc. Not Determined
Submitted references Postmortem proteolysis in three muscles from growing and mature beef cattle.
Isolation and characterization of mu-calpain, m-calpain, and calpastatin from postmortem muscle. I. Initial steps.
Purification and characterization of calpain and calpastatin from rainbow trout, Oncorhynchus mykiss.
Calpain is activated in degenerating photoreceptors in the rd1 mouse.
Effect of nutrient restriction on calpain and calpastatin content of skeletal muscle from cows and fetuses.
Effect of dietary protein on calpastatin in canine skeletal muscle.
Subcellular localization and in vivo subunit interactions of ubiquitous mu-calpain.
Fiber type-specific expression of major proteolytic systems in fast- to slow-transforming rabbit muscle.
Fiber type-specific expression of major proteolytic systems in fast- to slow-transforming rabbit muscle.
A comparison of the intracellular distribution of mu-calpain, m-calpain, and calpastatin in proliferating human A431 cells.
Cruzen SM, Paulino PV, Lonergan SM, Huff-Lonergan E
Meat science 2014 Feb;96(2 Pt A):854-61
Meat science 2014 Feb;96(2 Pt A):854-61
Isolation and characterization of mu-calpain, m-calpain, and calpastatin from postmortem muscle. I. Initial steps.
Camou JP, Mares SW, Marchello JA, Vazquez R, Taylor M, Thompson VF, Goll DE
Journal of animal science 2007 Dec;85(12):3400-14
Journal of animal science 2007 Dec;85(12):3400-14
Purification and characterization of calpain and calpastatin from rainbow trout, Oncorhynchus mykiss.
Saito M, Li H, Thompson VF, Kunisaki N, Goll DE
Comparative biochemistry and physiology. Part B, Biochemistry & molecular biology 2007 Apr;146(4):445-55
Comparative biochemistry and physiology. Part B, Biochemistry & molecular biology 2007 Apr;146(4):445-55
Calpain is activated in degenerating photoreceptors in the rd1 mouse.
Paquet-Durand F, Azadi S, Hauck SM, Ueffing M, van Veen T, Ekström P
Journal of neurochemistry 2006 Feb;96(3):802-14
Journal of neurochemistry 2006 Feb;96(3):802-14
Effect of nutrient restriction on calpain and calpastatin content of skeletal muscle from cows and fetuses.
Du M, Zhu MJ, Means WJ, Hess BW, Ford SP
Journal of animal science 2004 Sep;82(9):2541-7
Journal of animal science 2004 Sep;82(9):2541-7
Effect of dietary protein on calpastatin in canine skeletal muscle.
Helman EE, Huff-Lonergan E, Davenport GM, Lonergan SM
Journal of animal science 2003 Sep;81(9):2199-205
Journal of animal science 2003 Sep;81(9):2199-205
Subcellular localization and in vivo subunit interactions of ubiquitous mu-calpain.
Gil-Parrado S, Popp O, Knoch TA, Zahler S, Bestvater F, Felgenträger M, Holloschi A, Fernández-Montalván A, Auerswald EA, Fritz H, Fuentes-Prior P, Machleidt W, Spiess E
The Journal of biological chemistry 2003 May 2;278(18):16336-46
The Journal of biological chemistry 2003 May 2;278(18):16336-46
Fiber type-specific expression of major proteolytic systems in fast- to slow-transforming rabbit muscle.
Sultan KR, Dittrich BT, Leisner E, Paul N, Pette D
American journal of physiology. Cell physiology 2001 Feb;280(2):C239-47
American journal of physiology. Cell physiology 2001 Feb;280(2):C239-47
Fiber type-specific expression of major proteolytic systems in fast- to slow-transforming rabbit muscle.
Sultan KR, Dittrich BT, Leisner E, Paul N, Pette D
American journal of physiology. Cell physiology 2001 Feb;280(2):C239-47
American journal of physiology. Cell physiology 2001 Feb;280(2):C239-47
A comparison of the intracellular distribution of mu-calpain, m-calpain, and calpastatin in proliferating human A431 cells.
Lane RD, Allan DM, Mellgren RL
Experimental cell research 1992 Nov;203(1):5-16
Experimental cell research 1992 Nov;203(1):5-16
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry was performed on cancer biopsies of deparaffinized Human breast carcinoma tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:1000 with a mouse monoclonal antibody recognizing Calpastatin (Product # MA3-945) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
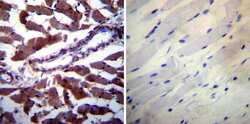
- Experimental details
- Immunohistochemistry was performed on biopsies of deparaffinized Human skeletal muscle tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing Calpastatin (Product # MA3-945) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
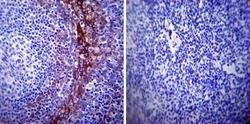
- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Human tonsil tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:500 with a mouse monoclonal antibody recognizing Calpastatin (Product # MA3-945) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
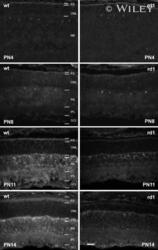
- Experimental details
- 3 Calpastatin immunofluorescence in the wt and rd1 retina during postnatal development. At PN4 and PN8 no significant differences were apparent. From PN11 onwards, the intensity of calpastatin immunofluorescence in the rd1 decreased compared with that in a wt animal of the same age. This was particularly evident in the rd1 PS where the staining also appeared to be more diffuse and less condensed than in wt. Images are representative of immunofluorescent staining from at least six different wt/rd1 animals. NB, neuroblast layer. Scale bar 50 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
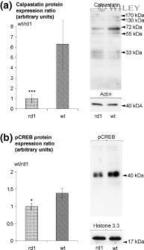
- Experimental details
- 4 Comparison of calpastatin and pCREB levels in the rd1 and wt retina at PN11 as assessed by immunoblotting. (a) Expression levels of calpastatin were approximately 6.3 times higher in wt than in rd1. Values are mean +- SD from four independent experiments each containing retinas from 4-5 wt and 4-5 rd1 animals. (b) Levels of pCREB in the wt retina were approximately 1.4 times higher than those in the rd1. Values are mean +- SD from three independent experiments each containing retinas from five wt and five rd1 animals. Arrowheads indicate positions of molecular weight standards; actin and histone immunolabelling were used as loading controls in (a) and (b) respectively. * p < 0.05, *** p < 0.001 versus wt (two-tailed Student's t -test).
 Explore
Explore Validate
Validate Learn
Learn Immunohistochemistry
Immunohistochemistry