Antibody data
- Antibody Data
- Antigen structure
- References [36]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [2]
- Flow cytometry [4]
- Other assay [20]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA3100 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD31 Monoclonal Antibody (HEC7)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- MA3100 targets CD31 in ICC/IF and ACS applications and shows reactivity with Human and Bovine samples.
- Antibody clone number
- HEC7
- Concentration
- 1 mg/mL
Submitted references Dephosphorylation of annexin A2 by protein phosphatase 1 regulates endothelial cell barrier.
The Extracellular Matrix Environment of Clear Cell Renal Cell Carcinoma Determines Cancer Associated Fibroblast Growth.
L-selectin regulates human neutrophil transendothelial migration.
Design and Validation of a Human Brain Endothelial Microvessel-on-a-Chip Open Microfluidic Model Enabling Advanced Optical Imaging.
Vγ9Vδ2 T Cells Activation Through Phosphoantigens Can Be Impaired by a RHOB Rerouting in Lung Cancer.
Direct reprogramming of human smooth muscle and vascular endothelial cells reveals defects associated with aging and Hutchinson-Gilford progeria syndrome.
Modelling early events in Mycobacterium bovis infection using a co-culture model of the bovine alveolus.
Cardiovascular tissue regeneration system based on multiscale scaffolds comprising double-layered hydrogels and fibers.
Bovine Organospecific Microvascular Endothelial Cell Lines as New and Relevant In Vitro Models to Study Viral Infections.
Lamin B2 Levels Regulate Polyploidization of Cardiomyocyte Nuclei and Myocardial Regeneration.
Targeting of prostate-specific membrane antigen for radio-ligand therapy of triple-negative breast cancer.
Fat-Specific Protein 27 Regulation of Vascular Function in Human Obesity.
Vitexin reduces epilepsy after hypoxic ischemia in the neonatal brain via inhibition of NKCC1.
Mutations in sphingosine-1-phosphate lyase cause nephrosis with ichthyosis and adrenal insufficiency.
Azithromycin effectively inhibits tumor angiogenesis by suppressing vascular endothelial growth factor receptor 2-mediated signaling pathways in lung cancer.
CXCR7 expression disrupts endothelial cell homeostasis and causes ligand-dependent invasion.
Aberrant proliferation in CXCR7+ endothelial cells via degradation of the retinoblastoma protein.
Association of T-zone reticular networks and conduits with ectopic lymphoid tissues in mice and humans.
Adipocytes as immune cells: differential expression of TWEAK, BAFF, and APRIL and their receptors (Fn14, BAFF-R, TACI, and BCMA) at different stages of normal and pathological adipose tissue development.
Transmigration across activated endothelium induces transcriptional changes, inhibits apoptosis, and decreases antimicrobial protein expression in human monocytes.
Dendritic cell adhesion is enhanced on endothelial cells preexposed to calcineurin inhibitors.
Dendritic cell adhesion is enhanced on endothelial cells preexposed to calcineurin inhibitors.
Integrin alpha4beta1-VCAM-1-mediated adhesion between endothelial and mural cells is required for blood vessel maturation.
A chemokine-dependent stromal induction mechanism for aberrant lymphocyte accumulation and compromised lymphatic return in rheumatoid arthritis.
Endothelial cell activation by myeloblasts: molecular mechanisms of leukostasis and leukemic cell dissemination.
Cell contact-dependent activation of alpha3beta1 integrin modulates endothelial cell responses to thrombospondin-1.
Regulation of angiogenesis in vivo by ligation of integrin alpha5beta1 with the central cell-binding domain of fibronectin.
Functional roles for PECAM-1 (CD31) and VE-cadherin (CD144) in tube assembly and lumen formation in three-dimensional collagen gels.
A standardized, a computer-assisted in vitro assay for the assessment of neutrophil transmigration across endothelial monolayers.
Endotoxin-inducible granulocyte-mediated hepatocytotoxicity requires adhesion and serine protease release.
L- and P-selectins, but not CD49d (VLA-4) integrins, mediate monocyte initial attachment to TNF-alpha-activated vascular endothelium under flow in vitro.
PECAM-1 is required for transendothelial migration of leukocytes.
Molecular and cellular properties of PECAM-1 (endoCAM/CD31): a novel vascular cell-cell adhesion molecule.
PECAM-1 (CD31) cloning and relation to adhesion molecules of the immunoglobulin gene superfamily.
PECAM-1 (CD31) cloning and relation to adhesion molecules of the immunoglobulin gene superfamily.
A human endothelial cell-restricted, externally disposed plasmalemmal protein enriched in intercellular junctions.
Király N, Thalwieser Z, Fonódi M, Csortos C, Boratkó A
IUBMB life 2021 Oct;73(10):1257-1268
IUBMB life 2021 Oct;73(10):1257-1268
The Extracellular Matrix Environment of Clear Cell Renal Cell Carcinoma Determines Cancer Associated Fibroblast Growth.
Bond KH, Chiba T, Wynne KPH, Vary CPH, Sims-Lucas S, Coburn JM, Oxburgh L
Cancers 2021 Nov 23;13(23)
Cancers 2021 Nov 23;13(23)
L-selectin regulates human neutrophil transendothelial migration.
Rahman I, Collado Sánchez A, Davies J, Rzeniewicz K, Abukscem S, Joachim J, Hoskins Green HL, Killock D, Sanz MJ, Charras G, Parsons M, Ivetic A
Journal of cell science 2021 Feb 8;134(3)
Journal of cell science 2021 Feb 8;134(3)
Design and Validation of a Human Brain Endothelial Microvessel-on-a-Chip Open Microfluidic Model Enabling Advanced Optical Imaging.
Salman MM, Marsh G, Kusters I, Delincé M, Di Caprio G, Upadhyayula S, de Nola G, Hunt R, Ohashi KG, Gray T, Shimizu F, Sano Y, Kanda T, Obermeier B, Kirchhausen T
Frontiers in bioengineering and biotechnology 2020;8:573775
Frontiers in bioengineering and biotechnology 2020;8:573775
Vγ9Vδ2 T Cells Activation Through Phosphoantigens Can Be Impaired by a RHOB Rerouting in Lung Cancer.
Laplagne C, Meddour S, Figarol S, Michelas M, Calvayrac O, Favre G, Laurent C, Fournié JJ, Cabantous S, Poupot M
Frontiers in immunology 2020;11:1396
Frontiers in immunology 2020;11:1396
Direct reprogramming of human smooth muscle and vascular endothelial cells reveals defects associated with aging and Hutchinson-Gilford progeria syndrome.
Bersini S, Schulte R, Huang L, Tsai H, Hetzer MW
eLife 2020 Sep 8;9
eLife 2020 Sep 8;9
Modelling early events in Mycobacterium bovis infection using a co-culture model of the bovine alveolus.
Lee DF, Stewart GR, Chambers MA
Scientific reports 2020 Oct 28;10(1):18495
Scientific reports 2020 Oct 28;10(1):18495
Cardiovascular tissue regeneration system based on multiscale scaffolds comprising double-layered hydrogels and fibers.
Kook YM, Hwang S, Kim H, Rhee KJ, Lee K, Koh WG
Scientific reports 2020 Nov 23;10(1):20321
Scientific reports 2020 Nov 23;10(1):20321
Bovine Organospecific Microvascular Endothelial Cell Lines as New and Relevant In Vitro Models to Study Viral Infections.
Lagrée AC, Fasani F, Rouxel C, Pivet M, Pourcelot M, Fablet A, Romey A, Caignard G, Vitour D, Blaise-Boisseau S, Kieda C, Boulouis HJ, Haddad N, Grillon C
International journal of molecular sciences 2020 Jul 24;21(15)
International journal of molecular sciences 2020 Jul 24;21(15)
Lamin B2 Levels Regulate Polyploidization of Cardiomyocyte Nuclei and Myocardial Regeneration.
Han L, Choudhury S, Mich-Basso JD, Ammanamanchi N, Ganapathy B, Suresh S, Khaladkar M, Singh J, Maehr R, Zuppo DA, Kim J, Eberwine JH, Wyman SK, Wu YL, Kühn B
Developmental cell 2020 Apr 6;53(1):42-59.e11
Developmental cell 2020 Apr 6;53(1):42-59.e11
Targeting of prostate-specific membrane antigen for radio-ligand therapy of triple-negative breast cancer.
Morgenroth A, Tinkir E, Vogg ATJ, Sankaranarayanan RA, Baazaoui F, Mottaghy FM
Breast cancer research : BCR 2019 Oct 22;21(1):116
Breast cancer research : BCR 2019 Oct 22;21(1):116
Fat-Specific Protein 27 Regulation of Vascular Function in Human Obesity.
Karki S, Farb MG, Sharma VM, Jash S, Zizza EJ, Hess DT, Carmine B, Carter CO, Pernar LI, Apovian CM, Puri V, Gokce N
Journal of the American Heart Association 2019 Jun 4;8(11):e011431
Journal of the American Heart Association 2019 Jun 4;8(11):e011431
Vitexin reduces epilepsy after hypoxic ischemia in the neonatal brain via inhibition of NKCC1.
Luo WD, Min JW, Huang WX, Wang X, Peng YY, Han S, Yin J, Liu WH, He XH, Peng BW
Journal of neuroinflammation 2018 Jun 20;15(1):186
Journal of neuroinflammation 2018 Jun 20;15(1):186
Mutations in sphingosine-1-phosphate lyase cause nephrosis with ichthyosis and adrenal insufficiency.
Lovric S, Goncalves S, Gee HY, Oskouian B, Srinivas H, Choi WI, Shril S, Ashraf S, Tan W, Rao J, Airik M, Schapiro D, Braun DA, Sadowski CE, Widmeier E, Jobst-Schwan T, Schmidt JM, Girik V, Capitani G, Suh JH, Lachaussée N, Arrondel C, Patat J, Gribouval O, Furlano M, Boyer O, Schmitt A, Vuiblet V, Hashmi S, Wilcken R, Bernier FP, Innes AM, Parboosingh JS, Lamont RE, Midgley JP, Wright N, Majewski J, Zenker M, Schaefer F, Kuss N, Greil J, Giese T, Schwarz K, Catheline V, Schanze D, Franke I, Sznajer Y, Truant AS, Adams B, Désir J, Biemann R, Pei Y, Ars E, Lloberas N, Madrid A, Dharnidharka VR, Connolly AM, Willing MC, Cooper MA, Lifton RP, Simons M, Riezman H, Antignac C, Saba JD, Hildebrandt F
The Journal of clinical investigation 2017 Mar 1;127(3):912-928
The Journal of clinical investigation 2017 Mar 1;127(3):912-928
Azithromycin effectively inhibits tumor angiogenesis by suppressing vascular endothelial growth factor receptor 2-mediated signaling pathways in lung cancer.
Li F, Huang J, Ji D, Meng Q, Wang C, Chen S, Wang X, Zhu Z, Jiang C, Shi Y, Liu S, Li C
Oncology letters 2017 Jul;14(1):89-96
Oncology letters 2017 Jul;14(1):89-96
CXCR7 expression disrupts endothelial cell homeostasis and causes ligand-dependent invasion.
Totonchy JE, Clepper L, Phillips KG, McCarty OJ, Moses AV
Cell adhesion & migration 2014;8(2):165-76
Cell adhesion & migration 2014;8(2):165-76
Aberrant proliferation in CXCR7+ endothelial cells via degradation of the retinoblastoma protein.
Totonchy JE, Osborn JM, Botto S, Clepper L, Moses AV
PloS one 2013;8(7):e69828
PloS one 2013;8(7):e69828
Association of T-zone reticular networks and conduits with ectopic lymphoid tissues in mice and humans.
Link A, Hardie DL, Favre S, Britschgi MR, Adams DH, Sixt M, Cyster JG, Buckley CD, Luther SA
The American journal of pathology 2011 Apr;178(4):1662-75
The American journal of pathology 2011 Apr;178(4):1662-75
Adipocytes as immune cells: differential expression of TWEAK, BAFF, and APRIL and their receptors (Fn14, BAFF-R, TACI, and BCMA) at different stages of normal and pathological adipose tissue development.
Alexaki VI, Notas G, Pelekanou V, Kampa M, Valkanou M, Theodoropoulos P, Stathopoulos EN, Tsapis A, Castanas E
Journal of immunology (Baltimore, Md. : 1950) 2009 Nov 1;183(9):5948-56
Journal of immunology (Baltimore, Md. : 1950) 2009 Nov 1;183(9):5948-56
Transmigration across activated endothelium induces transcriptional changes, inhibits apoptosis, and decreases antimicrobial protein expression in human monocytes.
Williams MR, Sakurai Y, Zughaier SM, Eskin SG, McIntire LV
Journal of leukocyte biology 2009 Dec;86(6):1331-43
Journal of leukocyte biology 2009 Dec;86(6):1331-43
Dendritic cell adhesion is enhanced on endothelial cells preexposed to calcineurin inhibitors.
Schlichting CL, Schareck WD, Weis M
Journal of cardiovascular pharmacology 2005 Sep;46(3):250-4
Journal of cardiovascular pharmacology 2005 Sep;46(3):250-4
Dendritic cell adhesion is enhanced on endothelial cells preexposed to calcineurin inhibitors.
Schlichting CL, Schareck WD, Weis M
Journal of cardiovascular pharmacology 2005 Sep;46(3):250-4
Journal of cardiovascular pharmacology 2005 Sep;46(3):250-4
Integrin alpha4beta1-VCAM-1-mediated adhesion between endothelial and mural cells is required for blood vessel maturation.
Garmy-Susini B, Jin H, Zhu Y, Sung RJ, Hwang R, Varner J
The Journal of clinical investigation 2005 Jun;115(6):1542-51
The Journal of clinical investigation 2005 Jun;115(6):1542-51
A chemokine-dependent stromal induction mechanism for aberrant lymphocyte accumulation and compromised lymphatic return in rheumatoid arthritis.
Burman A, Haworth O, Hardie DL, Amft EN, Siewert C, Jackson DG, Salmon M, Buckley CD
Journal of immunology (Baltimore, Md. : 1950) 2005 Feb 1;174(3):1693-700
Journal of immunology (Baltimore, Md. : 1950) 2005 Feb 1;174(3):1693-700
Endothelial cell activation by myeloblasts: molecular mechanisms of leukostasis and leukemic cell dissemination.
Stucki A, Rivier AS, Gikic M, Monai N, Schapira M, Spertini O
Blood 2001 Apr 1;97(7):2121-9
Blood 2001 Apr 1;97(7):2121-9
Cell contact-dependent activation of alpha3beta1 integrin modulates endothelial cell responses to thrombospondin-1.
Chandrasekaran L, He CZ, Al-Barazi H, Krutzsch HC, Iruela-Arispe ML, Roberts DD
Molecular biology of the cell 2000 Sep;11(9):2885-900
Molecular biology of the cell 2000 Sep;11(9):2885-900
Regulation of angiogenesis in vivo by ligation of integrin alpha5beta1 with the central cell-binding domain of fibronectin.
Kim S, Bell K, Mousa SA, Varner JA
The American journal of pathology 2000 Apr;156(4):1345-62
The American journal of pathology 2000 Apr;156(4):1345-62
Functional roles for PECAM-1 (CD31) and VE-cadherin (CD144) in tube assembly and lumen formation in three-dimensional collagen gels.
Yang S, Graham J, Kahn JW, Schwartz EA, Gerritsen ME
The American journal of pathology 1999 Sep;155(3):887-95
The American journal of pathology 1999 Sep;155(3):887-95
A standardized, a computer-assisted in vitro assay for the assessment of neutrophil transmigration across endothelial monolayers.
Gröger M, Matsumura T, Kohrgruber N, Maurer D, Wolff K, Petzelbauer P
Journal of immunological methods 1999 Jan 1;222(1-2):101-9
Journal of immunological methods 1999 Jan 1;222(1-2):101-9
Endotoxin-inducible granulocyte-mediated hepatocytotoxicity requires adhesion and serine protease release.
Sauer A, Hartung T, Aigner J, Wendel A
Journal of leukocyte biology 1996 Nov;60(5):633-43
Journal of leukocyte biology 1996 Nov;60(5):633-43
L- and P-selectins, but not CD49d (VLA-4) integrins, mediate monocyte initial attachment to TNF-alpha-activated vascular endothelium under flow in vitro.
Luscinskas FW, Ding H, Tan P, Cumming D, Tedder TF, Gerritsen ME
Journal of immunology (Baltimore, Md. : 1950) 1996 Jul 1;157(1):326-35
Journal of immunology (Baltimore, Md. : 1950) 1996 Jul 1;157(1):326-35
PECAM-1 is required for transendothelial migration of leukocytes.
Muller WA, Weigl SA, Deng X, Phillips DM
The Journal of experimental medicine 1993 Aug 1;178(2):449-60
The Journal of experimental medicine 1993 Aug 1;178(2):449-60
Molecular and cellular properties of PECAM-1 (endoCAM/CD31): a novel vascular cell-cell adhesion molecule.
Albelda SM, Muller WA, Buck CA, Newman PJ
The Journal of cell biology 1991 Sep;114(5):1059-68
The Journal of cell biology 1991 Sep;114(5):1059-68
PECAM-1 (CD31) cloning and relation to adhesion molecules of the immunoglobulin gene superfamily.
Newman PJ, Berndt MC, Gorski J, White GC 2nd, Lyman S, Paddock C, Muller WA
Science (New York, N.Y.) 1990 Mar 9;247(4947):1219-22
Science (New York, N.Y.) 1990 Mar 9;247(4947):1219-22
PECAM-1 (CD31) cloning and relation to adhesion molecules of the immunoglobulin gene superfamily.
Newman PJ, Berndt MC, Gorski J, White GC 2nd, Lyman S, Paddock C, Muller WA
Science (New York, N.Y.) 1990 Mar 9;247(4947):1219-22
Science (New York, N.Y.) 1990 Mar 9;247(4947):1219-22
A human endothelial cell-restricted, externally disposed plasmalemmal protein enriched in intercellular junctions.
Muller WA, Ratti CM, McDonnell SL, Cohn ZA
The Journal of experimental medicine 1989 Aug 1;170(2):399-414
The Journal of experimental medicine 1989 Aug 1;170(2):399-414
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-CD31 Monoclonal Antibody (HEC7) (Product # MA3100) and a 130kDa band corresponding to Platelet endothelial cell adhesion molecule was observed across cell lines tested. Membrane enriched extracts (40 µg lysate) of HUVEC (Lane 1), HEL 92.1.7 (Lane 2) and MDA-MB-231 (Lane 3) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0322BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23002) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using SuperSignal™ West Atto Ultimate Sensitivity Substrate (Product # A38556).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of CD31 (green) showing staining in the membrane and cytoplasm of THP-1 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a CD31 monoclonal antibody (Product # MA3100) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
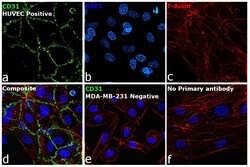
- Immunofluorescence analysis of Platelet endothelial cell adhesion molecule was performed using 70% confluent log phase HUVEC cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with CD31 Monoclonal Antibody (HEC7) (Product # MA3100) at 1:100 dilution in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing Cell Junctional localization. Panel e represents no expression in MDA-MB-231. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
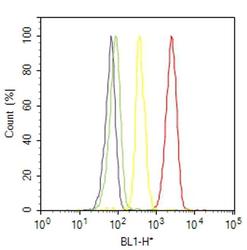
- Flow cytometry analysis of CD31 / PECAM-1 was done on THP-1 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with CD31 / PECAM-1 Mouse Monoclonal Antibody (MA3100, red histogram) or with mouse isotype control (yellow histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
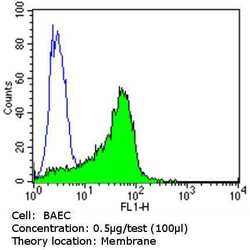
- Flow cytometry analysis of CD31 in BAEC cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD31 monoclonal antibody (Product # MA3100) at a dilution of 0.5 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Flow cytometry analysis of CD31 in PBMC cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD31 monoclonal antibody (Product # MA3100) at a dilution of 0.5 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
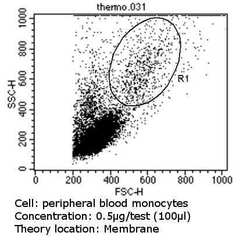
- Experimental details
- Flow cytometry analysis of CD31 in PBMC cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD31 monoclonal antibody (Product # MA3100) at a dilution of 0.5 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
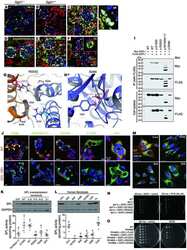
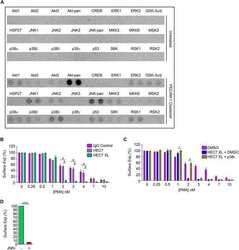
- Fig. 4. Clustering neutrophil PECAM-1 activates Akt family kinases and MAPKs, and potentiates ectodomain shedding of L-selectin. (A) MAPK array (R&D Systems) of primary human neutrophils clustered with HEC7 and subsequently cross-linked with secondary antibody. Upper panel represents untreated cells and lower panel is PECAM-1-clustered cells. See Materials and Methods section for more detail. (B) Control neutrophils isolated from healthy volunteers were labelled with either control IgG (purple bars), HEC7 (grey bars) or HEC7 plus clustering secondary antibody (turquoise bars). All labelling procedures were performed in the presence of Fc receptor block to avoid any non-specific binding and clustering of neutrophil Fc receptors (see Materials and Methods for more details). Antibody-labelled neutrophils were subsequently challenged with an increasing range of PMA concentrations (0.25-10 nM) and incubated for 30 min at 37degC. Alexa Fluor (r) 647-conjugated DREG56 monoclonal antibody was used to monitor L-selectin expression by flow cytometry. Fluorescence values were compared against untreated cells, which in turn were normalised to a value of 100%. Bar graph shows mean+-s.e.m. of three independent experiments using three different donors. # P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
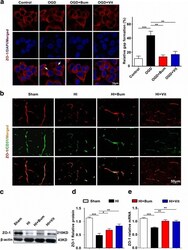
- Fig. 4 Vitexin inhibited HI-induced BBB destruction assayed by tight junction-related ZO-1. a TJs are characteristically located at cell-cell contact sites and are intact under physiological conditions. Confocal image of ZO-1 demonstrated disruption of the tight junctions and gap formation following OGD in RBMECs. Arrows indicate tight junction disruption. Scale bar = 50 mum. b Immunofluorescence staining for ZO-1 (red) and CD31 (green), a capillary endothelia marker, in the ischemic cortex of the sham, HI, HI+Bum, and HI+Vit groups 24 h after HI. Merged images of ZO-1 and CD31 staining are also shown. Scale bar = 50 mum. c , d Representative western blot for ZO-1 protein levels in the cerebral cortex from rats of each group. Densitometric value of the protein bands normalized to the respective beta-actin is also shown. * P < 0.05, ** P < 0.01, *** P < 0.001. e The mRNA expression of ZO-1 in the ipsi-ischemic brain tissue of each group was analyzed by real-time quantitative PCR. Data are shown as the mean +- SEM; ** P < 0.01, *** P < 0.001 in comparison with the HI group, n = 4~6 per group, based on a one-way ANOVA
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
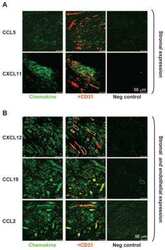
- Figure 5 Maintenance of the ectopic TRC network and the conduit system in RIP-CXCL13 tg + pancreas is partially LTbetaR dependent. Immunofluorescence analysis of 8-mum-thick pancreas sections from adult RIP-CXCL13 tg + mice (aged 4 months) treated twice per week for 22 days with 100 mug of soluble LTbetaR-Fc or control LFA-3-Fc fusion proteins. Data are representative of three mice per group. A: Serial frozen sections were stained for CD3 + T cells (red) and B220 + B cells (green) and for podoplanin (red) and CD31 (green). Scale bar = 100 mum. B: Staining of an infiltrated islet for podoplanin (red), lyve-1 (green), and DAPI + nuclei (blue), with the clustered DAPI + cells representing mainly infiltrating T and B cells. Lymphatic vessels appear in yellow (podoplanin + lyve1 + ); and reticular fibroblasts in red (podoplanin + lyve1 - ). Right: A higher magnification of the infiltrate is shown with the vascular marker CD31 instead of DAPI (blue). Most podoplanin + cells ( arrowheads ) show reticular morphological features and absence of vascular markers (Lyve1 - CD31 - ). C: Confocal images showing podoplanin + cells (red) and fibronectin + conduits (green) in the T-cell-rich region. Scale bar = 10 mum. D: Immunofluorescence analysis of CCL21 (green) expression in the podoplanin + region. Scale bar = 100 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
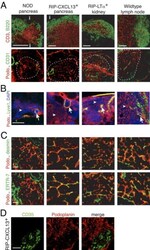
- Figure 3 The TLTs developing in murine models of chronic inflammation form a network of podoplanin + stromal cells in the T zone, similar to LNs. Immunofluorescence analysis of frozen sections of pancreas from NOD and RIP-CXCL13 tg + mice and kidney from RIP-LTalpha tg + mice and peripheral LNs. A: B220 + B cells (green) and CD3 + T cells (red), the TRC marker podoplanin (podo, red) combined with the endothelial marker CD31 (green) in serial sections. Tg - pancreata did not show reticular podoplanin staining typical for TRCs (data not shown). Scale bar = 100 mum. i indicates islet. B: Infiltrate labeled with podoplanin (red), the lymphatic vessel marker Lyve-1 (green), and DAPI + nuclei (blue). Arrows indicate lymphatic vessels; and arrowheads , the more abundant fibroblastic reticular cells. Scale bar = 100 mum. C: Higher-magnification images of the T-cell-rich zone of the infiltrate labeled with podoplanin (red) and desmin and the conduit marker ER-TR7 (green). Scale bar = 20 mum. D: The podoplanin (red) and CD35 (green) staining showing two distinct stromal cell networks in infiltrates of RIP-CXCL13 + pancreas. Scale bar = 100 mum. Data are representative of at least three independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
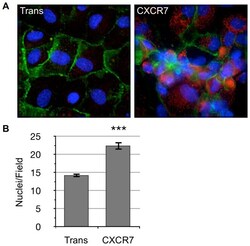
- Experimental details
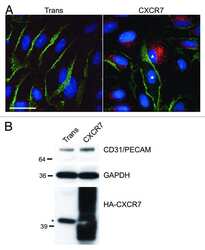
- Figure 2 CXCR7+ EC are not contact inhibited. (A) pLEC were infected with Trans only or Trans+CXCR7. At 20 hours post-infection cells were trypsinized, counted and replated onto chamber slides at confluence and allowed to attach for a further 20 hours. Resulting monolayers were fixed and stained for DAPI (blue), CD31 (green), and CXCR7 (red). (B) Nuclei were counted in 10 random fields from 2 independent wells from 2 independent experiments (n = 40, P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
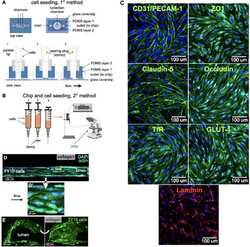
- Experimental details
- FIGURE 2 Cell seeding procedures and formation of a human brainmicrovessel using TY10 cells. (A) Cell seeding, 1st method. Representation of the steps used to increase the cell concentration before injection into the chip. Cells are allowed to settle by gravity (left and central panels) in the cell seeder and then injected as a bolus into the hollow lumen. Prior to injection, the chamber is capped with a plugged pipette tip (central panel). (B) Cell seeding, 2nd method. Representation of the steps used to inject cells into the chip in a more controlled and uniform manner compared to the 1st method. Cells are allowed to settle at the bottom of a syringe, and are then delivered into the hollow lumen at constant flow controlled by a syringe pump. (C) Immunostaining of TY10 monolayers: endothelial cell marker PECAM-1/CD31, transporters Glut-1 and transferrin receptor (TfR), and tight junction proteins ZO-1, Occludin and Claudin-5. Staining for laminin provides evidence that TY10 cells deposit extracellular matrix while in culture. Scale bar, 100 mum. (D) Representative image of a chemically fixed sample of TY10 cells after they were grown in the brain microvessel-on-a-chip with medium flowing from left to right at 1 mµL/min for 7 days. Volumetric image was obtained using a spinning disk confocal microscope. Maximum z-projection is shown for a sample stained with DAPI (nuclei, blue) and phalloidin (actin, green). Scale bar, 100 mum. (D') Representative image of a chemically
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
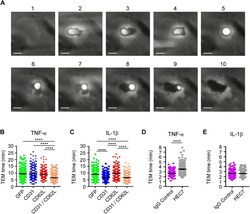
- Experimental details
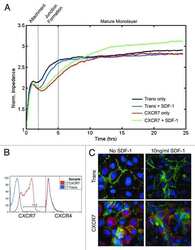
- Fig. 5. Co-expression of L-selectin and PECAM-1 in dHL-60 cells significantly increases TTT across TNF-activated HUVECs, and functionally blocking PECAM-1 in primary human neutrophils significantly reduces TTT across TNF-activated HUVEC. (A) Time series of frames (from Movie 3 , which includes the time code) showing how TEM of HL-60 cells is quantified. Cells transitioning from phase bright (image 2) to completely phase dark (image 10). Scale bars: 5 um. (B) Quantification of average TTT for dHL-60 cells transmigrating across TNF-activated HUVECs. The dHL-60 cells analysed are stably expressing GFP alone (green column scatter), PECAM-1-GFP alone (blue column scatter), L-selectin-RFP alone (red column scatter) or fluorescently tagged constructs of both PECAM-1 and L-selectin (orange column scatter). The TTT of at least 180 cells were quantified (mean+-s.e.m.) per group across three independent experiments. **** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
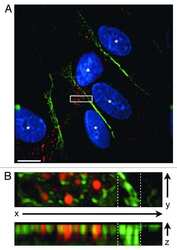
- Figure 3 Z-stack confocal images of co-cultures, at 48 h.p.i by BCG, moving from the apical (top left) through to basolateral (bottom right) aspect for each panel. ( a ) Co-cultures of endothelial BPAECs overlaid with the BATII epithelial cell line stained with the ATII marker SFTPC (green), phalloidin (red) and the endothelial marker CD31 (cyan). Moving left to right, a tight epithelial cell layer was observed (red and green fluorescence; green box), giving way to the underlying endothelial BPAEC (red and cyan fluorescence, blue box). ( b ) At 48 h.p.i, the epithelial BATII layer displayed regions of decimation, as evidenced by bare patches in the epithelial layer (red box); however phalloidin staining was still present underneath, in co-localisation with CD31 (blue box), suggesting that endothelial BPAECs were relatively unscathed. ( c ) xz visualisation of the untreated co-culture reflects the integral layers shown in the xy z-stack images of ( a ). ( d ) xz visualisation of the BCG infected co-culture, including regions of catastrophic damage to the epithelial BATII layer (white brackets), whilst CD31 staining is unaffected, suggesting that detrimental effects of BCG infection are limited to the epithelial component of the model. Images acquired using a 20 x objective on a Nikon Ti confocal microscope.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
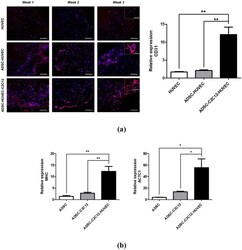
- Experimental details
- Figure 5 In vitro cardiovascular tissue formation in the multiscale scaffold. ( a ) (left) Immunofluorescence images for CD31 in the fibrin hydrogel region of the multiscale scaffold at each time point. Yellow dashed rectangle insets show x 40 magnified images of each group. Scale bars = 200 mum and 100 mum in low magnification and high magnification images, respectively. (right) Quantitative analysis of CD31 expression by qRT-PCR at 3-week culture. ( b ) Quantitative expression of cardiac markers (MHC, ACTC1) from ADSCs in the multiscale scaffold by qRT-PCR at 3-week culture. The expression levels to relative to the housekeeping gene (GAPDH) were calculated for analysis of qRT-PCR results (n = 3). Data are mean +- s.d. One-way ANOVA with Tukey's post-hoc test was used. ***P < 0.001, **P < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Cardiovascular tissue regeneration capacity of cell-laden multiscale scaffolds after subcutaneous implantation. ( a ) Photos showing the experimental procedure for in vivo studies. ( b ) Description of experimental design for in vivo studies. Immunofluorescence images and qRT-PCR results for ( c ) CD31, ( d ) cTNT, and ( e ) s.alpha. actinin. Scale bar = 200 mum and 50 mum for low magnification images and high magnification images, respectively. Data are mean +- s.d. One-way ANOVA with Tukey's post-hoc test was used, compare to ADSC and HUVEC monocultured group, respectively. ***P < 0.001, **P < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
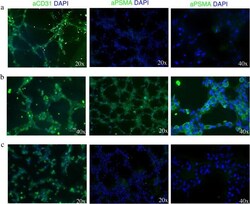
- Experimental details
- Fig. 2 TNBC cells induce PSMA expression on endothelial tubes. Fluorescence microscopy analysis of tube-associated expression of CD31 and PSMA on HUVEC cells incubated in a EBM-2 growth media supplemented with endothelial growth factors, b MDA-MB231-conditioned medium, and c MCF-7-conditioned medium
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
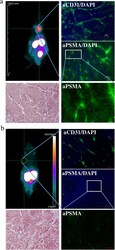
- Experimental details
- Fig. 6 PSMA-addressing tracer accumulates efficiently and specifically in PSMA-expressing tumor tissue. a muPET analysis of [ 68 Ga]-PSMA-11 biodistribution 30 min p.i. in MDA-MB231 subcutaneously xenografted NUDE mice with fluorescence microscopy imaging of tumor tissue section stained with CD31- and PSMA-specific antibodies and DAPI, and with Masson's Trichrome stain. b muPET analysis of [ 68 Ga]-PSMA-11 biodistribution 30 min p.i. in MCF-7 subcutaneously xenografted NUDE mice with fluorescence microscopy imaging of tumor tissue section stained with CD31- and PSMA-specific antibodies and DAPI, and with Masson's Trichrome stain ( n = 5)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Comparison of FSP 27 (fat-specific protein 27) protein in primary endothelial cells from subcutaneous vs visceral fat of obese subjects. A , Representative immunofluorescence image of isolated endothelial cells demonstrating significantly reduced expression of FSP 27 protein (red= FSP 27, green= CD 31, endothelial cell marker, blue=DAPI, a nuclear stain) in endothelial cells isolated from visceral compared with subcutaneous depots from obese subjects. B , Quantification of FSP 27 protein expression in endothelial cells isolated from subcutaneous and visceral fat (n=14, P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. Azithromycin inhibits lung tumor growth and tumor angiogenesis in xenograft mice. (A) Azithromycin significantly inhibits A549 lung tumor growth. SCID mice bearing A549 lung tumor xenografts at the flanks were treated with equal volume of vehicles, azithromycin at 20 mg/kg daily by intraperitoneal injection (n=10). (B) Azithromycin significantly inhibits tumor angiogenesis. Tumor blood vessels were visualized by CD31 staining. Nucleus was stained blue by 4,6-diamidino-2-phenylindole. Scale bar represents 20 um. (C) The average number of vessels were analyzed from three microscopic fields per tumor section, three tumor sections per tumor and ten tumors for each experiment group. (D and E) Decreased protein levels of VEGF and VEGFR2, but not HIF-1alpha, were observed in azithromycin-treated tumors. Representative western blot analysis images of tumor tissues were captured and presented. The protein levels of HIF-1alpha, VEGF and VEGFR were quantified using Image-J software with beta-actin levels for normalization. *P
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot