Antibody data
- Antibody Data
- Antigen structure
- References [13]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [1]
- Immunohistochemistry [3]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-16730 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Laminin Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Other
- Description
- This antibody is pan-specific and reacts well with all Laminin isoforms tested: Laminin-1 (alpha-1, beta-1, and gamma-1) and Laminin-2 (alpha-2, beta-1, and gamma-1).
- Concentration
- 1 mg/mL
Submitted references Transcriptional and Epigenetic Consequences of DMSO Treatment on HepaRG Cells.
Extracellular Matrix Proteins Confer Cell Adhesion-Mediated Drug Resistance Through Integrin α (v) in Glioblastoma Cells.
Effects of Aerobic Exercise Training on MyomiRs Expression in Cachectic and Non-Cachectic Cancer Mice.
Cancer Cell Invasion of Mammary Organoids with Basal-In Phenotype.
The effect of resistance training, detraining and retraining on muscle strength and power, myofibre size, satellite cells and myonuclei in older men.
Endothelial Lactate Controls Muscle Regeneration from Ischemia by Inducing M2-like Macrophage Polarization.
Exercise promotes satellite cell contribution to myofibers in a load-dependent manner.
Effect of natalizumab treatment on metalloproteinases and their inhibitors in a mouse model of multiple sclerosis.
Succinate induces skeletal muscle fiber remodeling via SUNCR1 signaling.
Morphological and Molecular Changes in Juvenile Normal Human Fibroblasts Exposed to Simulated Microgravity.
Inflammatory-Driven Angiogenesis in Bone Augmentation with Bovine Hydroxyapatite, B-Tricalcium Phosphate, and Bioglasses: A Comparative Study.
Both brown adipose tissue and skeletal muscle thermogenesis processes are activated during mild to severe cold adaptation in mice.
A semi-interpenetrating network of polyacrylamide and recombinant basement membrane allows pluripotent cell culture in a soft, ligand-rich microenvironment.
Dubois-Pot-Schneider H, Aninat C, Kattler K, Fekir K, Jarnouen K, Cerec V, Glaise D, Salhab A, Gasparoni G, Takashi K, Ishida S, Walter J, Corlu A
Cells 2022 Jul 26;11(15)
Cells 2022 Jul 26;11(15)
Extracellular Matrix Proteins Confer Cell Adhesion-Mediated Drug Resistance Through Integrin α (v) in Glioblastoma Cells.
Yu Q, Xiao W, Sun S, Sohrabi A, Liang J, Seidlits SK
Frontiers in cell and developmental biology 2021;9:616580
Frontiers in cell and developmental biology 2021;9:616580
Effects of Aerobic Exercise Training on MyomiRs Expression in Cachectic and Non-Cachectic Cancer Mice.
Gomes JLP, Tobias GC, Fernandes T, Silveira AC, Negrão CE, Chammas R, Brum PC, Oliveira EM
Cancers 2021 Nov 16;13(22)
Cancers 2021 Nov 16;13(22)
Cancer Cell Invasion of Mammary Organoids with Basal-In Phenotype.
Parigoris E, Lee S, Mertz D, Turner M, Liu AY, Sentosa J, Djomehri S, Chang HC, Luker K, Luker G, Kleer CG, Takayama S
Advanced healthcare materials 2021 Feb;10(4):e2000810
Advanced healthcare materials 2021 Feb;10(4):e2000810
The effect of resistance training, detraining and retraining on muscle strength and power, myofibre size, satellite cells and myonuclei in older men.
Blocquiaux S, Gorski T, Van Roie E, Ramaekers M, Van Thienen R, Nielens H, Delecluse C, De Bock K, Thomis M
Experimental gerontology 2020 May;133:110860
Experimental gerontology 2020 May;133:110860
Endothelial Lactate Controls Muscle Regeneration from Ischemia by Inducing M2-like Macrophage Polarization.
Zhang J, Muri J, Fitzgerald G, Gorski T, Gianni-Barrera R, Masschelein E, D'Hulst G, Gilardoni P, Turiel G, Fan Z, Wang T, Planque M, Carmeliet P, Pellerin L, Wolfrum C, Fendt SM, Banfi A, Stockmann C, Soro-Arnáiz I, Kopf M, De Bock K
Cell metabolism 2020 Jun 2;31(6):1136-1153.e7
Cell metabolism 2020 Jun 2;31(6):1136-1153.e7
Exercise promotes satellite cell contribution to myofibers in a load-dependent manner.
Masschelein E, D'Hulst G, Zvick J, Hinte L, Soro-Arnaiz I, Gorski T, von Meyenn F, Bar-Nur O, De Bock K
Skeletal muscle 2020 Jul 9;10(1):21
Skeletal muscle 2020 Jul 9;10(1):21
Effect of natalizumab treatment on metalloproteinases and their inhibitors in a mouse model of multiple sclerosis.
Pyka-Fosciak G, Lis GJ, Litwin JA
Journal of physiology and pharmacology : an official journal of the Polish Physiological Society 2020 Apr;71(2)
Journal of physiology and pharmacology : an official journal of the Polish Physiological Society 2020 Apr;71(2)
Succinate induces skeletal muscle fiber remodeling via SUNCR1 signaling.
Wang T, Xu YQ, Yuan YX, Xu PW, Zhang C, Li F, Wang LN, Yin C, Zhang L, Cai XC, Zhu CJ, Xu JR, Liang BQ, Schaul S, Xie PP, Yue D, Liao ZR, Yu LL, Luo L, Zhou G, Yang JP, He ZH, Du M, Zhou YP, Deng BC, Wang SB, Gao P, Zhu XT, Xi QY, Zhang YL, Shu G, Jiang QY
EMBO reports 2019 Sep;20(9):e47892
EMBO reports 2019 Sep;20(9):e47892
Morphological and Molecular Changes in Juvenile Normal Human Fibroblasts Exposed to Simulated Microgravity.
Buken C, Sahana J, Corydon TJ, Melnik D, Bauer J, Wehland M, Krüger M, Balk S, Abuagela N, Infanger M, Grimm D
Scientific reports 2019 Aug 15;9(1):11882
Scientific reports 2019 Aug 15;9(1):11882
Inflammatory-Driven Angiogenesis in Bone Augmentation with Bovine Hydroxyapatite, B-Tricalcium Phosphate, and Bioglasses: A Comparative Study.
Anghelescu VM, Neculae I, Dincă O, Vlădan C, Socoliuc C, Cioplea M, Nichita L, Popp C, Zurac S, Bucur A
Journal of immunology research 2018;2018:9349207
Journal of immunology research 2018;2018:9349207
Both brown adipose tissue and skeletal muscle thermogenesis processes are activated during mild to severe cold adaptation in mice.
Bal NC, Singh S, Reis FCG, Maurya SK, Pani S, Rowland LA, Periasamy M
The Journal of biological chemistry 2017 Oct 6;292(40):16616-16625
The Journal of biological chemistry 2017 Oct 6;292(40):16616-16625
A semi-interpenetrating network of polyacrylamide and recombinant basement membrane allows pluripotent cell culture in a soft, ligand-rich microenvironment.
Price AJ, Huang EY, Sebastiano V, Dunn AR
Biomaterials 2017 Mar;121:179-192
Biomaterials 2017 Mar;121:179-192
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
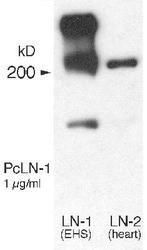
- Experimental details
- Western Blot detection of laminin by Western blot using Product # PA1-16730.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Laminin in 0.2 µg rat heart cells lysates (lane 1) and 0.2 µg of purified laminin protein from mouse EHS sarcoma (lane 2). Samples were incubated in Laminin polyclonal antibody (Product # PA1-16730). Laminin Antibody recognizes 3 laminin isotypes: alpha 1 (440 kDa), beta 1 (220 kDa) and gamma 1 (220 kDa). Also recognized is a laminin binding protein at 120 kDa in both rat heart lysates and purified laminin protein. Since this protein always coexpresses with laminin this crossreactivity is irrelevant. Theoretical molecular weight of LAMA1 is 337 kDa.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
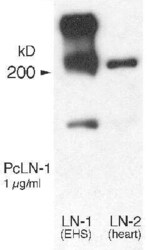
- Experimental details
- Western blot analysis of Laminin in mouse EHS tumor crude extracts (left) and Laminin-2 expression in rat heart crude extracts (right). Samples were incubated in Laminin polyclonal antibody (Product # PA1-16730 using a dilution of 1 µg/mL. Theoretical Molecular Weight is 337 kDa.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
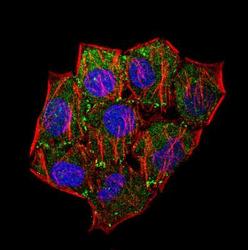
- Experimental details
- Immunocytochemistry analysis of Laminin in HeLa cells. Samples were incubated in Laminin polyclonal antibody (Product # PA1-16730) followed by Alexa Fluor 488-conjugated Goat to rabbit IgG secondary antibody (green, A). Actin filaments were labeled with Alexa Fluor 568 phalloidin (red, B). DAPI was used to stain the cell nuclei (blue, C).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
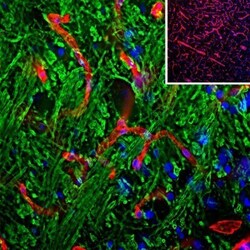
- Experimental details
- Immunohistochemical analysis of Laminin in brain stem sections. Samples were incubated in Laminin polyclonal antibody (Product # PA1-16730) using a dilution of 1:1,000 (red). Costained with chicken pAb to Myelin Basic Protein (MBP), dilution 1:5,000 in green. The blue is DAPI staining of nuclear DNA. Following transcardial perfusion of rat with 4% paraformaldehyde, brain was post fixed for 24 hours, cut to 45 µM, and free-floating sections were stained with the above antibodies. The laminin antibody is an excellent marker of basement membranes surrounding blood vessels, while the MBP antibody stains the myelin sheathes around axons.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
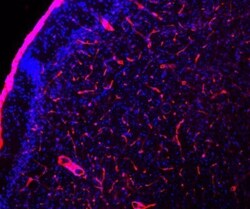
- Immunohistochemical analysis of Laminin in mouse sections of cortex. Samples were incubated in Laminin polyclonal antibody (Product # PA1-16730). Antibody is red. Blue is DAPI staining of DNA. This antibody reveals strong staining in the basement membranes of blood vessels.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
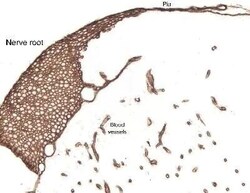
- Immunohistochemical analysis of Laminin in paraformaldehyde/paraffin-embedded rat spinal cord and dorsal root tissue. Samples were incubated in Laminin polyclonal antibody (Product # PA1-16730). Pepsin antigen retrieval was performed on this tissue sample.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
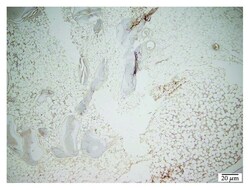
- Figure 13 TCP 40x laminin stain showing moderate scaring.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
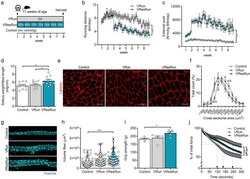
- Fig. 1 Running pattern and muscle hypertrophy are load-dependent. a Experimental set-up. b Average running distances and c average external work per night throughout the exercise protocol ( n = 16 mice per group). d Soleus weight normalized to tibia length. e Representative cross-sections stained for laminin (red) and f quantification ( n = 7-8 mice per group) of fiber cross-sectional area distribution in soleus muscle. g Representative pictures of single myofibers isolated from soleus and ( h quantification of fiber volume of 11-16 single myofibers per mouse ( n = 4 mice per group). i Muscle force production by grip strength and j time-to-fatigue in ex vivo stimulated muscle. Statistics, one-way ANOVA test with Tukey correction for multiple comparisons ( d , h , i , j ) or two-way ANOVA test with a Bonferroni post hoc test ( b , c , f , k ). (* p < 0.05; ** p < 0.01; *** p < 0.001). Each dot represents a single mouse ( d , i , j ) or muscle fiber ( h ). Bar graphs ( d , i , j ) and line graphs ( b , c , f , k ) represent mean +- SEM (error bars). Violin plot ( h ). Scale bars, 50 mum ( e ) and 25 mum ( g ). Pax7 mTmG/+ female mice on a C57BL/6J background were used for all analyses
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
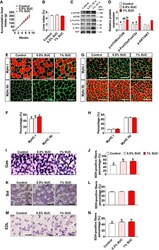
- Additional file 2: Figure S2. Muscle hypertrophy is muscle dependent, fiber type dependent and load-dependent. (A) Representative cross-sections for m. soleus (SOL) stained for MyHCI (green), MyHCIIa (blue) and Laminin (red). (B,C) Quantification of fiber cross-sectional area distribution and (D,E) average fiber cross-sectional area. (F) Representative cross-sections for plantaris (PLT) stained for MyHCIIa (blue), MyHCIIb (yellow) and laminin. (G,H) quantification of fiber cross-sectional area distribution and (I,J) average fiber cross-sectional area. (K) Fiber cross-sectional area distribution for gastrocnemius (GAS), (L) tibialis anterior (TA) and (M) extensor digitorum longus (EDL). Statistics: one-way ANOVA test with Tukey correction for multiple comparisons (*p < 0.05; **p < 0.01; ***p < 0.001) (a, p < 0.05 VResRun compared to Control; b, p < 0.05 VResRun compared to VRun; c, p < 0.05 VRun compared to Control). Bar graphs and line graphs represent mean +- SEM (error bars). Scale bar, 100 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
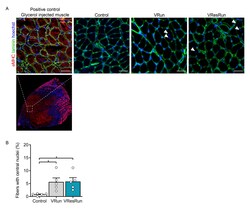
- Additional file 3: Figure S3. Fiber remodeling and central nuclei. (A) Myofibers stained for embryonic myosin heavy chain (eMHC), laminin and hoechst. No eMHC + fibers were detected in Control, VRun, VResRun. Glycerol injected muscle was used as a positive control. (B) Quantification of fibers containing one or more centrally located nucleus. Arrows indicate central nuclei. Bar graph represents mean +- SEM (error bars). Statistics: one-way ANOVA test with Tukey correction for multiple comparisons (*p < 0.05). Each dot represents a single mouse. Scale bar, 50 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
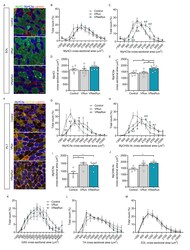
- Figure EV1 Effects of succinate on growth performance and muscle fiber composition in mice (related to Figs 1 and 3 ) Male C57BL/6J mice were fed with chow diet supplemented with 0, 0.5%, or 1% SUC for 8 weeks. A, B (A) Cumulative food intake and (B) liver index of mice treated with SUC for 8 weeks ( n = 8). C, D Immunoblots and quantification of p-mTOR, mTOR, p-FoxO3a, FoxO3a, p-AKT, and AKT protein in gastrocnemius ( n = 3). E-H Representative images and quantification of laminin (green), MyHC I (red), and MyHC IIb (red) immunofluorescent staining in the (E, F) soleus and (G, H) extensor digitorum longus muscle. The graphs show the MyHC I and MyHC IIb fiber ratios ( n = 6). Scale bar in (E, G) represents 100 mum. I-N The percentage of SDH positive in the (I, J) gastrocnemius, (K, L) soleus, and (M, N) extensor digitorum longus muscle is shown by SDH enzyme staining. Only darkly stained SDH fibers are treated as SDH-positive fibers. The graphs show the SDH-positive fiber ratios ( n = 4-6). Scale bar in I, K, and M represents 100 mum. Data information: Results are presented as mean +- SEM. Different letters between bars mean P
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot