Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 25-0036-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD3 Monoclonal Antibody (SK7), PE-Cyanine7, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The SK7 monoclonal antibody reacts with human and chimpanzee CD3e, a 20 kDa subunit of the TCR complex. Along with the other CD3 subunits gamma and delta, the epsilon chain is required for proper assembly, trafficking and surface expression of the TCR complex. CD3 is expressed by thymocytes in a developmentally regulated manner and by all mature T cells. The SK7 and UCHT1 monoclonal antibodies cross-block binding, suggesting recognition of overlapping epitope. In contrast, clones OKT3 and SK7 see different epitopes. The antibody SK7 recognizes chimpanzee CD3. Applications Reported: This SK7 antibody has been reported for use in flow cytometric analysis. Applications Tested: This SK7 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Light sensitivity: This tandem dye is sensitive to photo-induced oxidation. Please protect this vial and stained samples from light. Fixation: Samples can be stored in IC Fixation Buffer (Product # 00-8222) (100 µL of cell sample + 100 µL of IC Fixation Buffer) or 1-step Fix/Lyse Solution (Product # 00-5333) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically. Excitation: 488-561 nm; Emission: 775 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- SK7
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD19 eFluor® 450 (Product # 48-0199-42) and Mouse IgG1 K Isotype Control PE-Cyanine7 (Product # 25-4714-80) (left) or Anti-Human CD3 PE-Cyanine7 (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
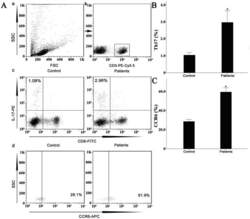
- Experimental details
- Figure 7 Circulating percentages of Th17 cells and CCR6-positive cells in peripheral blood are increased in IVD degenerated patients when compared with controls. Heparinized peripheral whole blood cells from 20 patients and 15 healthy controls were stimulated with phorbol myristate acetate (PMA), ionomycin, and monensin for 4 h and subsequently stained with fluorochrome-labeled antibodies as described in Materials and Methods. A(a) Lymphocytes were gated by flow cytometry. A(b) CD3 + T subsets were gated by flow cytometry; the plots in the inset box represent the CD3 + T cells. A(c) Representative IL-17 expression levels in the CD3 + CD8 - T subsets (CD4 + T subsets) from each group are shown. The percentages of positive cells are shown in the upper left panels. A(d) Representative surface CCR6 expression levels on the CD3 + CD8 - IL-17 + subsets from each group are shown. The percentages of positive cells are shown in the right panel. (B) The percentage of circulating Th17 cells was significantly higher in IVD degenerated patients (2.973+-0.689%) than in the control group (1.039+-0.156%; *, p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 In vitro T cell proliferation. Mononuclear cells from psoriatic patients (red curve) and healthy controls (blue curve) were labelled with 1 uM CFSE prior to culturing and incubated for 5 days alone, with 1 ug/ml PHA ( A ) or in the presence of recombinant proteins K17 ( B ), S1 ( C ), S4 ( D ), PS1 ( E ) and PK ( F ) at concentration of 10 ug/ml. After 5 days, cells were labelled with a PE-conjugated anti-CD3 antibody and 7-AAD prior to flow cytometry analysis. Gated CD3+ lymphocytes are shown on the CFSE fluorescence histograms to demonstrate the decrease in fluorescence intensity during divisions. The higher peak for both patients and healthy controls represents a larger population of non-dividing parental cells (P). The signal of interest is the smaller peak that corresponds to dividing cells (shown using arrow under F ), and such cells are only observed in patents when treated with K17 ( B ), S1 ( C ), or S4 ( D ) proteins. Figures represent the percentage of CFSE low CD3+ cells (proliferating population).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
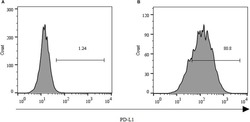
- Experimental details
- FIGURE 2 Expression of PD-L1 on human HCC PLC/PRF/5 cells. (A) Human HCC PLC/PRF/5 cells were cultured alone in the absence of GPC3-CAR T cells in RPMI 1640 medium containing 10% FBS. (B) Human HCC PLC/PRF/5 cells were cocultured with GPC3-CAR T cells at an effector:Target ratio of 1:1 for 18 h in RPMI 1640 medium containing 10% FBS. PD-L1 was determined by flow cytometry in the CD3-negative gate, and the fixable, viable stain 780 was used for discriminating live from dead cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 4 Efficient disruption of PD-1 expression on the surface of GPC3-CAR T cells. PD-1 and CAR expression on the surface of T cells were detected by flow cytometry on day 3 after the re-stimulation with anti-CD3/anti-CD28 beads. UTD, untransduced T cells; WT, wild type.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Identification of key immune cell types of MYJ1633 following ex vivo expansion. a The distribution of NK cells (CD3 - CD16 + CD56 + ), NKT cells (CD3 + CD16 + CD56 + ), and T cells (CD3 + CD16 - CD56 - ) of freshly isolated PBMCs and MYJ1633 was examined by flow cytometry. b Proportion of helper T cells (Th cells; CD4 + ) and cytotoxic T cells (Tc cells; CD8 + ) among CD3 + cells of MYJ1633. These data were analyzed from 6 individuals (Additional file 1 : Figure S1). Significant differences between groups were determined by Student's t test. The data represented as mean +- SEM
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 HIV-1 Cap-RNA 58 -treated DCs mediate T cell proliferation and differentiation. DCs were unstimulated or treated with 1 nM HIV-1 Cap-RNA 58 or LPS for 48 h, cocultured with CellTrace Violet-labeled peripheral blood lymphocytes and harvested after 5 days. Proliferation was determined in (A) live CD3 + CD4 + cells and (B) CD3 + CD8 + T cells by flow cytometry. (C,D) DCs were treated with vehicle control, 1 nM HIV-1 control RNA, 1 nM HIV-1 Cap-RNA 58 , LPS, LPS + PGE 2 , or LPS + IFNgamma for 48 h and cocultured with naive T cells. After 11-13 days, cells were restimulated and intracellular expression levels of IFNgamma (T H 1) and IL-4 (T H 2) were analyzed using flow cytometry. Number in plots indicate percentage of cells in the quadrant. Data are representative of collated data of three (A,B) or four (D) donors, or four donors (C) of different experiments (mean +- s.d.). * P < 0.05, ** P < 0.01, Student's t -test. NS, not significant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 CG-745 increases helper T cells, cytotoxic T cells and natural killer T cells, and decreases Treg: (A) hPBMCs were incubated with CG (CG-745) for 36 hours and a subset of hPBMCs was analyzed using the antibodies indicated in the text; (B) hPBMCs were co-cultured with Huh7, Hep3B, HepG2 or PLC/PRF/5 cells for 36 hours with or without CG, and a subset of hPBMCs was analyzed by Attune Nxt (Invitrogen, USA).
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry