12-7229-41
antibody from Invitrogen Antibodies
Targeting: IL22
IL-21, IL-22, IL-D110, IL-TIF, ILTIF, MGC79382, MGC79384, TIFa, TIFIL-23, zcyto18
Antibody data
- Antibody Data
- Antigen structure
- References [22]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-7229-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-22 Monoclonal Antibody (22URTI), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 22URTI monoclonal antibody reacts with human interleukin(IL)-22. IL-22 is a 20 kDa member of the IL-10 cytokine family that is secreted primarily by Th17 and NK cells. Nevertheless, other T cells have also been shown to produce IL-22. In in vitro Th17 cultures, induction of IL-22 expression is greater in response to IL-23 than IL-6 or TGF beta, suggesting that this cytokine may be secreted by more fully differentiated Th17 cells in vivo. A heterodimer consisting of IL-10R2 and IL-22R1 serves as the receptor for IL-22. Applications Reported: This 22URTI antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This 22URTI antibody has been pre-titrated and tested by intracellular staining followed by flow cytometric analysis of restimulated, Th17-polarized CD4+ normal human peripheral blood cells or stimulated total normal human peripheral blood cells. This can be used at 5 µL (0.01 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- 22URTI
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Decreased Frequencies of Gamma/Delta T Cells Expressing Th1/Th17 Cytokine, Cytotoxic, and Immune Markers in Latent Tuberculosis-Diabetes/Pre-Diabetes Comorbidity.
MicroRNA‑21‑5p protects melanocytes via targeting STAT3 and modulating Treg/Teff balance to alleviate vitiligo.
Expanded IL-22(+) Group 3 Innate Lymphoid Cells and Role of Oxidized LDL-C in the Pathogenesis of Axial Spondyloarthritis with Dyslipidaemia.
TIRC7 inhibits Th1 cells by upregulating the expression of CTLA‑4 and STAT3 in mice with acute graft‑versus‑host disease.
Human Fc Receptor-like 3 Inhibits Regulatory T Cell Function and Binds Secretory IgA.
CD62L Is a Functional and Phenotypic Marker for Circulating Innate Lymphoid Cell Precursors.
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Hypothermia Promotes Interleukin-22 Expression and Fine-Tunes Its Biological Activity.
Celiac Disease-Related Inflammation Is Marked by Reduction of Nkp44/Nkp46-Double Positive Natural Killer Cells.
Pathological Roles of Interleukin-22 in the Development of Recurrent Hepatitis C after Liver Transplantation.
Characterization and Quantification of Innate Lymphoid Cell Subsets in Human Lung.
Fetal CD103+ IL-17-Producing Group 3 Innate Lymphoid Cells Represent the Dominant Lymphocyte Subset in Human Amniotic Fluid.
Downregulation of RUNX3 moderates the frequency of Th17 and Th22 cells in patients with psoriasis.
A novel, dual cytokine-secretion assay for the purification of human Th22 cells that do not co-produce IL-17A.
Schistosoma mansoni Infection in Ugandan Men Is Associated with Increased Abundance and Function of HIV Target Cells in Blood, but Not the Foreskin: A Cross-sectional Study.
Vitamin D regulates cytokine patterns secreted by dendritic cells to promote differentiation of IL-22-producing T cells.
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Increased circulating T‑helper 22 cells in patients with dilated cardiomyopathy.
BET bromodomain inhibition suppresses TH17-mediated pathology.
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
Complement component C5a promotes expression of IL-22 and IL-17 from human T cells and its implication in age-related macular degeneration.
Human Th17 cells comprise heterogeneous subsets including IFN-gamma-producing cells with distinct properties from the Th1 lineage.
Kathamuthu GR, Kumar NP, Moideen K, Menon PA, Babu S
Frontiers in cellular and infection microbiology 2021;11:756854
Frontiers in cellular and infection microbiology 2021;11:756854
MicroRNA‑21‑5p protects melanocytes via targeting STAT3 and modulating Treg/Teff balance to alleviate vitiligo.
Huo J, Liu T, Li F, Song X, Hou X
Molecular medicine reports 2021 Jan;23(1)
Molecular medicine reports 2021 Jan;23(1)
Expanded IL-22(+) Group 3 Innate Lymphoid Cells and Role of Oxidized LDL-C in the Pathogenesis of Axial Spondyloarthritis with Dyslipidaemia.
Min HK, Moon J, Lee SY, Lee AR, Lee CR, Lee J, Kwok SK, Cho ML, Park SH
Immune network 2021 Dec;21(6):e43
Immune network 2021 Dec;21(6):e43
TIRC7 inhibits Th1 cells by upregulating the expression of CTLA‑4 and STAT3 in mice with acute graft‑versus‑host disease.
Zhu F, Qiu T, Zhu S, Zhao K, Chen C, Qiao J, Pan B, Yan Z, Chen W, Liu Q, Wu Q, Cao J, Sang W, Zeng L, Sun H, Li Z, Xu K
Oncology reports 2020 Jul;44(1):43-54
Oncology reports 2020 Jul;44(1):43-54
Human Fc Receptor-like 3 Inhibits Regulatory T Cell Function and Binds Secretory IgA.
Agarwal S, Kraus Z, Dement-Brown J, Alabi O, Starost K, Tolnay M
Cell reports 2020 Feb 4;30(5):1292-1299.e3
Cell reports 2020 Feb 4;30(5):1292-1299.e3
CD62L Is a Functional and Phenotypic Marker for Circulating Innate Lymphoid Cell Precursors.
Bar-Ephraim YE, Koning JJ, Burniol Ruiz E, Konijn T, Mourits VP, Lakeman KA, Boon L, Bögels M, van Maanen JP, Den Haan JMM, van Egmond M, Bouma G, Reijmers RM, Mebius RE
Journal of immunology (Baltimore, Md. : 1950) 2019 Jan 1;202(1):171-182
Journal of immunology (Baltimore, Md. : 1950) 2019 Jan 1;202(1):171-182
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Chen X, Wang Y, Wang J, Wen J, Jia X, Wang X, Zhang H
Oncology letters 2018 Jul;16(1):253-261
Oncology letters 2018 Jul;16(1):253-261
Hypothermia Promotes Interleukin-22 Expression and Fine-Tunes Its Biological Activity.
Chichelnitskiy E, Himmelseher B, Bachmann M, Pfeilschifter J, Mühl H
Frontiers in immunology 2017;8:742
Frontiers in immunology 2017;8:742
Celiac Disease-Related Inflammation Is Marked by Reduction of Nkp44/Nkp46-Double Positive Natural Killer Cells.
Marafini I, Monteleone I, Di Fusco D, Sedda S, Cupi ML, Fina D, Paoluzi AO, Pallone F, Monteleone G
PloS one 2016;11(5):e0155103
PloS one 2016;11(5):e0155103
Pathological Roles of Interleukin-22 in the Development of Recurrent Hepatitis C after Liver Transplantation.
Gao Y, Ren H, Meng F, Li J, Cheung E, Li H, Zhao J, Liu H, Liu Z, Zhang M
PloS one 2016;11(4):e0154419
PloS one 2016;11(4):e0154419
Characterization and Quantification of Innate Lymphoid Cell Subsets in Human Lung.
De Grove KC, Provoost S, Verhamme FM, Bracke KR, Joos GF, Maes T, Brusselle GG
PloS one 2016;11(1):e0145961
PloS one 2016;11(1):e0145961
Fetal CD103+ IL-17-Producing Group 3 Innate Lymphoid Cells Represent the Dominant Lymphocyte Subset in Human Amniotic Fluid.
Marquardt N, Ivarsson MA, Sundström E, Åkesson E, Martini E, Eidsmo L, Mjösberg J, Friberg D, Kublickas M, Ek S, Tegerstedt G, Seiger Å, Westgren M, Michaëlsson J
Journal of immunology (Baltimore, Md. : 1950) 2016 Oct 15;197(8):3069-3075
Journal of immunology (Baltimore, Md. : 1950) 2016 Oct 15;197(8):3069-3075
Downregulation of RUNX3 moderates the frequency of Th17 and Th22 cells in patients with psoriasis.
Fu D, Song X, Hu H, Sun M, Li Z, Tian Z
Molecular medicine reports 2016 Jun;13(6):4606-12
Molecular medicine reports 2016 Jun;13(6):4606-12
A novel, dual cytokine-secretion assay for the purification of human Th22 cells that do not co-produce IL-17A.
Wawrzyniak M, Ochsner U, Wirz O, Wawrzyniak P, van de Veen W, Akdis CA, Akdis M
Allergy 2016 Jan;71(1):47-57
Allergy 2016 Jan;71(1):47-57
Schistosoma mansoni Infection in Ugandan Men Is Associated with Increased Abundance and Function of HIV Target Cells in Blood, but Not the Foreskin: A Cross-sectional Study.
Prodger JL, Ssemaganda A, Ssetaala A, Kitandwe PK, Muyanja E, Mpendo J, Nanvubya A, Wambuzi M, Nielsen L, Kiwanuka N, Kaul R
PLoS neglected tropical diseases 2015;9(9):e0004067
PLoS neglected tropical diseases 2015;9(9):e0004067
Vitamin D regulates cytokine patterns secreted by dendritic cells to promote differentiation of IL-22-producing T cells.
Sommer A, Fabri M
PloS one 2015;10(6):e0130395
PloS one 2015;10(6):e0130395
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Wang WB, Yen ML, Liu KJ, Hsu PJ, Lin MH, Chen PM, Sudhir PR, Chen CH, Chen CH, Sytwu HK, Yen BL
Stem cell reports 2015 Sep 8;5(3):392-404
Stem cell reports 2015 Sep 8;5(3):392-404
Increased circulating T‑helper 22 cells in patients with dilated cardiomyopathy.
Kong Q, Li X, Wu W, Yang F, Liu Y, Lai W, Pan X, Gao M, Xue Y
Molecular medicine reports 2014 Jul;10(1):359-64
Molecular medicine reports 2014 Jul;10(1):359-64
BET bromodomain inhibition suppresses TH17-mediated pathology.
Mele DA, Salmeron A, Ghosh S, Huang HR, Bryant BM, Lora JM
The Journal of experimental medicine 2013 Oct 21;210(11):2181-90
The Journal of experimental medicine 2013 Oct 21;210(11):2181-90
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
Zhang L, Li YG, Li YH, Qi L, Liu XG, Yuan CZ, Hu NW, Ma DX, Li ZF, Yang Q, Li W, Li JM
PloS one 2012;7(4):e31000
PloS one 2012;7(4):e31000
Complement component C5a promotes expression of IL-22 and IL-17 from human T cells and its implication in age-related macular degeneration.
Liu B, Wei L, Meyerle C, Tuo J, Sen HN, Li Z, Chakrabarty S, Agron E, Chan CC, Klein ML, Chew E, Ferris F, Nussenblatt RB
Journal of translational medicine 2011 Jul 15;9:1-12
Journal of translational medicine 2011 Jul 15;9:1-12
Human Th17 cells comprise heterogeneous subsets including IFN-gamma-producing cells with distinct properties from the Th1 lineage.
Boniface K, Blumenschein WM, Brovont-Porth K, McGeachy MJ, Basham B, Desai B, Pierce R, McClanahan TK, Sadekova S, de Waal Malefyt R
Journal of immunology (Baltimore, Md. : 1950) 2010 Jul 1;185(1):679-87
Journal of immunology (Baltimore, Md. : 1950) 2010 Jul 1;185(1):679-87
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Intracellular staining of CD4-enriched Th17-polarized (using Human IL-23 Recombinant Protein (Product # 14-8239-63), normal human peripheral blood cells with Anti-Human CD4 FITC (Product # 11-0049-42) and Anti-Human IL-22 PE. Cultures were treated with Protein Transport Inhibitor Cocktail (Product # 00-4980-03) (left) alone (Product # 00-4506-51) Cell Stimulation Cocktail (plus protein transport inhibitors) (Product # 00-4975-03) (right) for 5 hours prior to intracellular staining using the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00). Cells in the lymphocyte gate were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
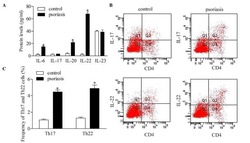
- Figure 5 Inhibition of RUNX3 regulates the frequencies of Th17 and Th22 in CD4 + T cells from patients with psoriasis. The frequencies of Th17 and Th22 cells in CD4 + T cells from patients with psoriasis transfected with RUNX3 siRNA or an siRNA control was detected using flow cytometry. (A and B) Flow cytometric analysis of Th17 in CD4 + T cells from patients with psoriasis. The percentages of cells in the Q2 region represent the percentage of Th17 cells. (C and D) Flow cytometric analysis of Th22 in CD4 + T cells from patients with psoriasis. The percentages of cells in the Q2 region represent the percentage of Th22 cells. (E) Percentages of Th17 and Th22 cells in CD4 + T cells from patients with psoriasis transfected with RUNX3 siRNA or an siRNA control. Data are presented as the mean +- standard deviation of three experiments. * P
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Decreased frequencies of gammadelta T cells expressing Th17 cytokines in LTB comorbidities. PBMCs were either untreated or treated with Mtb or positive control antigens for 18 h. The absolute (unstimulated, UNS) and antigen-stimulated (PPD, WCL, P/I) net frequencies of Th17 (IL-17A, IL-17F, IL-22) cytokines were shown in LTB DM (n = 20), LTB PDM (n = 20), and LTB NDM (n = 20) groups. Geometric mean values were represented using bars, and every circle denotes a single individual. Kruskal-Wallis test with multiple Dunn's comparison was used to determine the p values.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 8. Changes in cytokines in recipient mice on day 21 post-allo-BMT. (A) Changes in IFN-gamma-positive Th1 in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. (B) Changes in IL-4-positive Th2 cells in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. (C) Changes in IL-17-positive Tg17 cells in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. (D) Changes in IL-22-positive Th22 cells in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. Allo-BMT, allogeneic bone marrow transplant; Th, T helper; IFN, interferon; IL, interleukin.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Multipotent Human Mesenchymal Stromal Cells (hMSCs) Suppress Th17 Responses (A-D) Human peripheral blood CD3 + leukocytes (PBLs) (A, representative data; B, pooled data of 17 PBL donors co-cultured with all three hMSC donors) or CD3 + CD4 T cells (C, representative data; D, pooled data of 11 PBL donors co-cultured with all three hMSC donors) were co-cultured without (left) or with (right) hMSCs ex vivo, followed by PMA/ionomycin stimulation for 6 hr. (E-H) IL-17A production in ex-vivo-cultured CD3 + T cells was assessed by intracellular staining. IL-17A and IFN-gamma production in CD3 + PBLs (E, representative data; F, pooled data) or CD3 + CD4 T cells (G, representative data; H, pooled data) without and with co-culture of hMSCs was analyzed by flow cytometry. Representative intracellular staining is shown for IL-17A + IFN-gamma - - CD3 + T cells (R3 region) and IL-17A + IFN-gamma + (R5 region) CD3 + T cells, and pooled data from PBLs (n = 4) or CD4 T cells (n = 4) co-cultured with two hMSC donors (donors A and B) are provided in (F) and (H), respectively. Gray bars represent the percentages of IL-17A + IFN-gamma - - CD3 + T cells, whereas white bars represent the percentages of IL-17A + IFN-gamma + T cells. (I and J) IL-22 production in four donors of CD3 + CD4 T cells (I, representative data; J, pooled data) without and with co-culture of two donors of hMSCs (donors A and B) was assessed by intracellular staining. Cell percentages are denoted in the dotplot quadran
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Flow cytometric analysis was used to determine the distribution of Th22, Th17 and Th1 cells in EGC, HE and HY ( Fig. 1 ). Flow cytometric analysis of Th22, Th17 and Th1 cells in peripheral whole blood from EGC (n=39), HE (n=32) and HY (n=31). (A) Lymphocytes were gated in P1 using flow cytometry. CD4 + IFN-gamma - lymphocytes were gated in P2 using flow cytometry, and representative results of flow cytometric analyses for (B) Th1 (CD4 + IFN-gamma + ), (C) Th22 (CD4 + IFN-gamma - IL-17 - IL-22 + ) and Th17 (CD4 + IFN-gamma - IL-17 + IL-22 - ) cells in the three groups of subjects are presented. The number of cells stained in EGC, HE and HY in P2 were 2,654, 4,696 and 5,185, respectively. The proportion of (D) Th22, (E) Th17 and (F) Th1 cells in the three groups of subjects. The proportion of (G) Th22 and (H) Th17 cells in peripheral whole blood derived from patients with early (n=13) or advanced (n=26) gastric cancer. The association between the proportion of (I) Th22 and Th17 cells, (J) Th22 and Th1 cells, and (K) Th17 and Th1 cells, in peripheral whole blood of all subjects. *P
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry