Antibody data
- Antibody Data
- Antigen structure
- References [13]
- Comments [0]
- Validations
- Western blot [4]
- Other assay [8]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-758 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Nicastrin Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- PA1-758 detects nicastrin from human, mouse and rat samples. PA1-758 has been successfully used in Western blot procedures. By Western blot, this antibody detects an ~110 kDa protein representing nicastrin. PA1-758 immunizing peptide corresponds to amino acid residues 688-708 from human nicastrin. There is a single amino acid substitution between the human and mouse sequence. PA1-758 immunizing peptide (Cat. # PEP-131) is available for use in neutralization and control experiments.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 1 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references Upregulation of APP endocytosis by neuronal aging drives amyloid-dependent synapse loss.
Bin1 and CD2AP polarise the endocytic generation of beta-amyloid.
Presenilin transmembrane domain 8 conserved AXXXAXXXG motifs are required for the activity of the γ-secretase complex.
Chronic administration of anti-stroke herbal medicine TongLuoJiuNao reduces amyloidogenic processing of amyloid precursor protein in a mouse model of Alzheimer's disease.
Ubiquilin-1 modulates γ-secretase-mediated ε-site cleavage in neuronal cells.
Presenilins regulate neurotrypsin gene expression and neurotrypsin-dependent agrin cleavage via cyclic AMP response element-binding protein (CREB) modulation.
Potent amyloidogenicity and pathogenicity of Aβ43.
Calsenilin regulates presenilin 1/γ-secretase-mediated N-cadherin ε-cleavage and β-catenin signaling.
Ubiquilin 1 modulates amyloid precursor protein trafficking and Abeta secretion.
The presenilin proteins are components of multiple membrane-bound complexes that have different biological activities.
Presenilin-1, nicastrin, amyloid precursor protein, and gamma-secretase activity are co-localized in the lysosomal membrane.
APH-1 interacts with mature and immature forms of presenilins and nicastrin and may play a role in maturation of presenilin.nicastrin complexes.
Presenilin and nicastrin regulate each other and determine amyloid beta-peptide production via complex formation.
Burrinha T, Martinsson I, Gomes R, Terrasso AP, Gouras GK, Almeida CG
Journal of cell science 2021 May 1;134(9)
Journal of cell science 2021 May 1;134(9)
Bin1 and CD2AP polarise the endocytic generation of beta-amyloid.
Ubelmann F, Burrinha T, Salavessa L, Gomes R, Ferreira C, Moreno N, Guimas Almeida C
EMBO reports 2017 Jan;18(1):102-122
EMBO reports 2017 Jan;18(1):102-122
Presenilin transmembrane domain 8 conserved AXXXAXXXG motifs are required for the activity of the γ-secretase complex.
Marinangeli C, Tasiaux B, Opsomer R, Hage S, Sodero AO, Dewachter I, Octave JN, Smith SO, Constantinescu SN, Kienlen-Campard P
The Journal of biological chemistry 2015 Mar 13;290(11):7169-84
The Journal of biological chemistry 2015 Mar 13;290(11):7169-84
Chronic administration of anti-stroke herbal medicine TongLuoJiuNao reduces amyloidogenic processing of amyloid precursor protein in a mouse model of Alzheimer's disease.
He P, Li P, Hua Q, Liu Y, Staufenbiel M, Li R, Shen Y
PloS one 2013;8(3):e58181
PloS one 2013;8(3):e58181
Ubiquilin-1 modulates γ-secretase-mediated ε-site cleavage in neuronal cells.
Viswanathan J, Haapasalo A, Kurkinen KM, Natunen T, Mäkinen P, Bertram L, Soininen H, Tanzi RE, Hiltunen M
Biochemistry 2013 Jun 4;52(22):3899-912
Biochemistry 2013 Jun 4;52(22):3899-912
Presenilins regulate neurotrypsin gene expression and neurotrypsin-dependent agrin cleavage via cyclic AMP response element-binding protein (CREB) modulation.
Almenar-Queralt A, Kim SN, Benner C, Herrera CM, Kang DE, Garcia-Bassets I, Goldstein LS
The Journal of biological chemistry 2013 Dec 6;288(49):35222-36
The Journal of biological chemistry 2013 Dec 6;288(49):35222-36
Potent amyloidogenicity and pathogenicity of Aβ43.
Saito T, Suemoto T, Brouwers N, Sleegers K, Funamoto S, Mihira N, Matsuba Y, Yamada K, Nilsson P, Takano J, Nishimura M, Iwata N, Van Broeckhoven C, Ihara Y, Saido TC
Nature neuroscience 2011 Jul 3;14(8):1023-32
Nature neuroscience 2011 Jul 3;14(8):1023-32
Calsenilin regulates presenilin 1/γ-secretase-mediated N-cadherin ε-cleavage and β-catenin signaling.
Jang C, Choi JK, Na YJ, Jang B, Wasco W, Buxbaum JD, Kim YS, Choi EK
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2011 Dec;25(12):4174-83
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2011 Dec;25(12):4174-83
Ubiquilin 1 modulates amyloid precursor protein trafficking and Abeta secretion.
Hiltunen M, Lu A, Thomas AV, Romano DM, Kim M, Jones PB, Xie Z, Kounnas MZ, Wagner SL, Berezovska O, Hyman BT, Tesco G, Bertram L, Tanzi RE
The Journal of biological chemistry 2006 Oct 27;281(43):32240-53
The Journal of biological chemistry 2006 Oct 27;281(43):32240-53
The presenilin proteins are components of multiple membrane-bound complexes that have different biological activities.
Gu Y, Sanjo N, Chen F, Hasegawa H, Petit A, Ruan X, Li W, Shier C, Kawarai T, Schmitt-Ulms G, Westaway D, St George-Hyslop P, Fraser PE
The Journal of biological chemistry 2004 Jul 23;279(30):31329-36
The Journal of biological chemistry 2004 Jul 23;279(30):31329-36
Presenilin-1, nicastrin, amyloid precursor protein, and gamma-secretase activity are co-localized in the lysosomal membrane.
Pasternak SH, Bagshaw RD, Guiral M, Zhang S, Ackerley CA, Pak BJ, Callahan JW, Mahuran DJ
The Journal of biological chemistry 2003 Jul 18;278(29):26687-94
The Journal of biological chemistry 2003 Jul 18;278(29):26687-94
APH-1 interacts with mature and immature forms of presenilins and nicastrin and may play a role in maturation of presenilin.nicastrin complexes.
Gu Y, Chen F, Sanjo N, Kawarai T, Hasegawa H, Duthie M, Li W, Ruan X, Luthra A, Mount HT, Tandon A, Fraser PE, St George-Hyslop P
The Journal of biological chemistry 2003 Feb 28;278(9):7374-80
The Journal of biological chemistry 2003 Feb 28;278(9):7374-80
Presenilin and nicastrin regulate each other and determine amyloid beta-peptide production via complex formation.
Edbauer D, Winkler E, Haass C, Steiner H
Proceedings of the National Academy of Sciences of the United States of America 2002 Jun 25;99(13):8666-71
Proceedings of the National Academy of Sciences of the United States of America 2002 Jun 25;99(13):8666-71
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
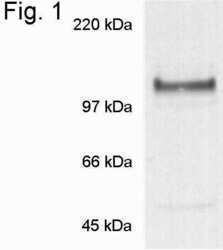
- Experimental details
- Western blot of nicastrin on mouse primary cultured neuronal cells using Product # PA1-758.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on membrane extracts (30 µglysate) of T98G (Lane 1), and RSC96 (Lane 2). The blots were probed with Rabbit Anti-Nicastrin Polyclonal Antibody (Product # PA1-758, 2 µg/mL) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A27036, 0.25 µg/mL, 1:4000 dilution). A 120 kDa band corresponding to immature Nicastrin was observed across the cell lines tested. In addition, a 140 kDa band corresponding to mature, glycosylated Nicastrin was observed in RSC96. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
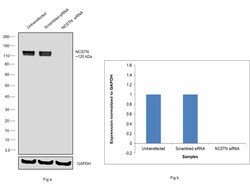
- Experimental details
- Knockdown of Nicastrin was achieved by transfecting HeLa with Nicastrin specific siRNAs (Silencer® select Product # s23708, s23707). Western blot analysis (Fig. a) was performed using Whole cell extracts from the Nicastrin knockdown cells (lane 3), non-targeting scrambled siRNA transfected cells (lane 2) and untransfected cells (lane 1). The blot was probed with Nicastrin Polyclonal Antibody (Product # PA1-758, 1:1000 dilution) and Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution). Densitometric analysis of this western blot is shown in histogram (Fig. b). Decrease in signal upon siRNA mediated knock down confirms that antibody is specific to Nicastrin.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Nicastrin Polyclonal Antibody (Product # PA1-758) and a 120 kDa band corresponding to Nicastrin was observed across cell lines and tissues tested along with an uncharacterized band (*) at 55 kDa. Whole cell extracts (60 µg lysate) of HeLa (Lane 1), IMR-32 (Lane 2), NIH/3T3 (Lane 3), L6 (Lane 4), Mouse Brain (Lane 5) and Rat Brain (Lane 6) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # LC2001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1 µg/mL) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
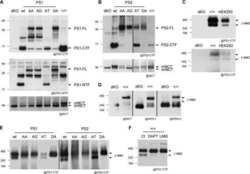
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
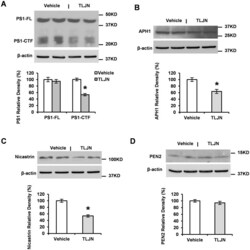
- Experimental details
- Figure 4 Herbal medicine TLJN reduces levels of gamma-secretase components. APP23 mice were treated with TLJN for 6 months. Levels of gamma-secretase components were analyzed by Western blotting. Representative images showed the bands of both PS1-CTF and PS1-FL ( A ), APH1 ( B ), nicastrin ( C ) and PEN2 ( D ). Then Spot density analysis was performed and a significant decrease was observed in PS1-CTF ( A ), APH1 ( B ) and nicastrin ( C ) but not in PS1-FL ( A ) and PEN2 ( D ). Significant differences were presented as * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- The gamma-secretase complex, but not its proteolytic activity, controls neurotrypsin expression and activity. A, Western blot analysis showing APP, N-cad, and APLP2 full length (FL) and corresponding CTFs in wild-type cells treated with DMSO (vehicle) or 0.5 µm Compound E (CE) for 24 h. Tubulin is shown as a loading control. B, RT-qPCR analysis of Tbp-normalized neurotrypsin mRNA expression in wild-type cells treated as in A (top panel) and Western blot analysis of agrin-90 in conditioned medium analyzed (bottom panel) using the same cells as in A. C, Western blot analysis of Psen1/2 dKO (+hPSEN1/2) or Psen1/2 dKO (+hPSEN1 D257A/hPSEN2) cells to detect PSEN1 CTF, PSEN1 full length (FL), APP full length, APP CTF, N-cad full length (FL), N-cad/CTF1, and mature (m.) and immature (im.) nicastrin. Tubulin is shown as a loading control. D, RT-qPCR analysis of Tbp-normalized neurotrypsin mRNA expression (top panel) and Western blot analysis of agrin-90 (bottom panel) in conditioned medium from the same cells as in C. E, Western blot analysis of wild-type (WT) and nicastrin KO cells of mature (m.) and immature (im.) nicastrin, APP full length (FL), APP CTF, N-cad full length, and N-cad/CTF1. Tubulin is shown as a loading control. F, RT-qPCR analysis of Tbp-normalized neurotrypsin mRNA expression (top panel) and Western blot analysis of agrin-90 in conditioned medium (bottom panel) from the same cells as in E. G, Western blot analysis of Psen1/2 dKO and Psen1/2 dKO cells rescued with deltaE9
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot