Antibody data
- Antibody Data
- Antigen structure
- References [35]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [4]
- Immunohistochemistry [2]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-12826 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- MAP2 Monoclonal Antibody (AP18)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- MA5-12826 has been successfully used in immunofluorescence analysis of Map2 in human glutamate neurons derived from iPSCs.
- Antibody clone number
- AP18
- Concentration
- 0.2 mg/mL
Submitted references Human iPSC Modeling of Genetic Febrile Seizure Reveals Aberrant Molecular and Physiological Features Underlying an Impaired Neuronal Activity.
Purine nucleoside phosphorylase deficiency induces p53-mediated intrinsic apoptosis in human induced pluripotent stem cell-derived neurons.
A microengineered Brain-Chip to model neuroinflammation in humans.
microRNAs (miR 9, 124, 155 and 224) transdifferentiate mouse macrophages to neurons.
Protocol for High-Throughput Screening of Neural Cell or Brain Tissue Protein Using a Dot-Blot Technique with Near-Infrared Imaging.
Anti-aging effects of Ribes meyeri anthocyanins on neural stem cells and aging mice.
Brain-derived neurotrophic factor mediates macrophage migration inhibitory factor to protect neurons against oxygen-glucose deprivation.
Nrg1 Intracellular Signaling Is Neuroprotective upon Stroke.
Detecting Neurodevelopmental Toxicity of Domoic Acid and Ochratoxin A Using Rat Fetal Neural Stem Cells.
Brain-specific Drp1 regulates postsynaptic endocytosis and dendrite formation independently of mitochondrial division.
Intra-cerebral implantation of a variety of collagenous scaffolds with nervous embryonic cells.
Silver nanoparticles induce neurotoxicity in a human embryonic stem cell-derived neuron and astrocyte network.
Neuronal sphingosine kinase 2 subcellular localization is altered in Alzheimer's disease brain.
MAP2 Defines a Pre-axonal Filtering Zone to Regulate KIF1- versus KIF5-Dependent Cargo Transport in Sensory Neurons.
Silver nanoparticles exhibit coating and dose-dependent neurotoxicity in glutamatergic neurons derived from human embryonic stem cells.
Rapid generation of sub-type, region-specific neurons and neural networks from human pluripotent stem cell-derived neurospheres.
NRP1-mediated Sema3A signals coordinate laminar formation in the developing chick optic tectum.
AAD-2004 Attenuates Progressive Neuronal Loss in the Brain of Tg-betaCTF99/B6 Mouse Model of Alzheimer Disease.
Intracranial extraskeletal myxoid chondrosarcoma : case report and literature review.
Semaphorin 3A induces CaV2.3 channel-dependent conversion of axons to dendrites.
Intramedullary papillary ependymoma with choroid plexus differentiation and cerebrospinal fluid dissemination to the brain.
Successful isolation and long-term establishment of a cell line with stem cell-like features from an anaplastic medulloblastoma.
S100B induces tau protein hyperphosphorylation via Dickopff-1 up-regulation and disrupts the Wnt pathway in human neural stem cells.
Electrophysiological recording of re-aggregating brain cell cultures on multi-electrode arrays to detect acute neurotoxic effects.
Enzymatic properties and localization of motopsin (PRSS12), a protease whose absence causes mental retardation.
Translational control of glial glutamate transporter EAAT2 expression.
Differential localization of ErbB receptor ensembles influences their signaling in hippocampal neurons.
Immunolocalization of MAP-2 in routinely formalin-fixed, paraffin-embedded guinea pig brain sections using microwave irradiation: a comparison of different combinations of antibody clones and antigen retrieval buffer solutions.
Involvement of glial cells in the neurotoxicity of parathion and chlorpyrifos.
Fetal human neural progenitors can be the target for tumor transformation.
Altered levels and distribution of microtubule-associated proteins before disease onset in a mouse model of amyotrophic lateral sclerosis.
Involvement of microglia-neuron interactions in the tumor necrosis factor-alpha release, microglial activation, and neurodegeneration induced by trimethyltin.
Simultaneous suppression of cdc2 and cdk2 activities induces neuronal differentiation of PC12 cells.
Suppression of sprouting: An early function of NMDA receptors in the absence of AMPA/kainate receptor activity.
Suppression of sprouting: An early function of NMDA receptors in the absence of AMPA/kainate receptor activity.
Scalise S, Zannino C, Lucchino V, Lo Conte M, Scaramuzzino L, Cifelli P, D'Andrea T, Martinello K, Fucile S, Palma E, Gambardella A, Ruffolo G, Cuda G, Parrotta EI
Biomedicines 2022 May 5;10(5)
Biomedicines 2022 May 5;10(5)
Purine nucleoside phosphorylase deficiency induces p53-mediated intrinsic apoptosis in human induced pluripotent stem cell-derived neurons.
Tsui M, Biro J, Chan J, Min W, Dobbs K, Notarangelo LD, Grunebaum E
Scientific reports 2022 May 31;12(1):9084
Scientific reports 2022 May 31;12(1):9084
A microengineered Brain-Chip to model neuroinflammation in humans.
Pediaditakis I, Kodella KR, Manatakis DV, Le CY, Barthakur S, Sorets A, Gravanis A, Ewart L, Rubin LL, Manolakos ES, Hinojosa CD, Karalis K
iScience 2022 Aug 19;25(8):104813
iScience 2022 Aug 19;25(8):104813
microRNAs (miR 9, 124, 155 and 224) transdifferentiate mouse macrophages to neurons.
Challagundla N, Agrawal-Rajput R
Experimental cell research 2021 May 1;402(1):112563
Experimental cell research 2021 May 1;402(1):112563
Protocol for High-Throughput Screening of Neural Cell or Brain Tissue Protein Using a Dot-Blot Technique with Near-Infrared Imaging.
Chlebowski AC, Kisby GE
STAR protocols 2020 Sep 18;1(2)
STAR protocols 2020 Sep 18;1(2)
Anti-aging effects of Ribes meyeri anthocyanins on neural stem cells and aging mice.
Gao J, Wu Y, He D, Zhu X, Li H, Liu H, Liu H
Aging 2020 Sep 12;12(17):17738-17753
Aging 2020 Sep 12;12(17):17738-17753
Brain-derived neurotrophic factor mediates macrophage migration inhibitory factor to protect neurons against oxygen-glucose deprivation.
Bae SH, Yoo MR, Kim YY, Hong IK, Kim MH, Lee SH, Kim DY
Neural regeneration research 2020 Aug;15(8):1483-1489
Neural regeneration research 2020 Aug;15(8):1483-1489
Nrg1 Intracellular Signaling Is Neuroprotective upon Stroke.
Navarro-González C, Huerga-Gómez A, Fazzari P
Oxidative medicine and cellular longevity 2019;2019:3930186
Oxidative medicine and cellular longevity 2019;2019:3930186
Detecting Neurodevelopmental Toxicity of Domoic Acid and Ochratoxin A Using Rat Fetal Neural Stem Cells.
Gill S, Kumara VMR
Marine drugs 2019 Oct 4;17(10)
Marine drugs 2019 Oct 4;17(10)
Brain-specific Drp1 regulates postsynaptic endocytosis and dendrite formation independently of mitochondrial division.
Itoh K, Murata D, Kato T, Yamada T, Araki Y, Saito A, Adachi Y, Igarashi A, Li S, Pletnikov M, Huganir RL, Watanabe S, Kamiya A, Iijima M, Sesaki H
eLife 2019 Oct 11;8
eLife 2019 Oct 11;8
Intra-cerebral implantation of a variety of collagenous scaffolds with nervous embryonic cells.
Drobnik J, Pietrucha K, Janczar K, Polis L, Polis B, Safandowska M, Szymański J
Experimental and therapeutic medicine 2019 Dec;18(6):4758-4764
Experimental and therapeutic medicine 2019 Dec;18(6):4758-4764
Silver nanoparticles induce neurotoxicity in a human embryonic stem cell-derived neuron and astrocyte network.
Repar N, Li H, Aguilar JS, Li QQ, Drobne D, Hong Y
Nanotoxicology 2018 Mar;12(2):104-116
Nanotoxicology 2018 Mar;12(2):104-116
Neuronal sphingosine kinase 2 subcellular localization is altered in Alzheimer's disease brain.
Dominguez G, Maddelein ML, Pucelle M, Nicaise Y, Maurage CA, Duyckaerts C, Cuvillier O, Delisle MB
Acta neuropathologica communications 2018 Apr 3;6(1):25
Acta neuropathologica communications 2018 Apr 3;6(1):25
MAP2 Defines a Pre-axonal Filtering Zone to Regulate KIF1- versus KIF5-Dependent Cargo Transport in Sensory Neurons.
Gumy LF, Katrukha EA, Grigoriev I, Jaarsma D, Kapitein LC, Akhmanova A, Hoogenraad CC
Neuron 2017 Apr 19;94(2):347-362.e7
Neuron 2017 Apr 19;94(2):347-362.e7
Silver nanoparticles exhibit coating and dose-dependent neurotoxicity in glutamatergic neurons derived from human embryonic stem cells.
Begum AN, Aguilar JS, Elias L, Hong Y
Neurotoxicology 2016 Dec;57:45-53
Neurotoxicology 2016 Dec;57:45-53
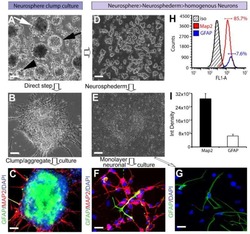
Rapid generation of sub-type, region-specific neurons and neural networks from human pluripotent stem cell-derived neurospheres.
Begum AN, Guoynes C, Cho J, Hao J, Lutfy K, Hong Y
Stem cell research 2015 Nov;15(3):731-741
Stem cell research 2015 Nov;15(3):731-741
NRP1-mediated Sema3A signals coordinate laminar formation in the developing chick optic tectum.
Watanabe Y, Sakuma C, Yaginuma H
Development (Cambridge, England) 2014 Sep;141(18):3572-82
Development (Cambridge, England) 2014 Sep;141(18):3572-82
AAD-2004 Attenuates Progressive Neuronal Loss in the Brain of Tg-betaCTF99/B6 Mouse Model of Alzheimer Disease.
Baek IS, Kim TK, Seo JS, Lee KW, Lee YA, Cho J, Gwag BJ, Han PL
Experimental neurobiology 2013 Mar;22(1):31-7
Experimental neurobiology 2013 Mar;22(1):31-7
Intracranial extraskeletal myxoid chondrosarcoma : case report and literature review.
Park JH, Kim MJ, Kim CJ, Kim JH
Journal of Korean Neurosurgical Society 2012 Sep;52(3):246-9
Journal of Korean Neurosurgical Society 2012 Sep;52(3):246-9
Semaphorin 3A induces CaV2.3 channel-dependent conversion of axons to dendrites.
Nishiyama M, Togashi K, von Schimmelmann MJ, Lim CS, Maeda S, Yamashita N, Goshima Y, Ishii S, Hong K
Nature cell biology 2011 Jun;13(6):676-85
Nature cell biology 2011 Jun;13(6):676-85
Intramedullary papillary ependymoma with choroid plexus differentiation and cerebrospinal fluid dissemination to the brain.
Dulai MS, Caccamo DV, Briley AL, Edwards MS, Fisher PG, Lehman NL
Journal of neurosurgery. Pediatrics 2010 May;5(5):511-7
Journal of neurosurgery. Pediatrics 2010 May;5(5):511-7
Successful isolation and long-term establishment of a cell line with stem cell-like features from an anaplastic medulloblastoma.
Raso A, Negri F, Gregorio A, Nozza P, Mascelli S, De Marco P, Merello E, Milanaccio C, Ravegnani M, Cama A, Garrè ML, Capra V
Neuropathology and applied neurobiology 2008 Jun;34(3):306-15
Neuropathology and applied neurobiology 2008 Jun;34(3):306-15
S100B induces tau protein hyperphosphorylation via Dickopff-1 up-regulation and disrupts the Wnt pathway in human neural stem cells.
Esposito G, Scuderi C, Lu J, Savani C, De Filippis D, Iuvone T, Steardo L Jr, Sheen V, Steardo L
Journal of cellular and molecular medicine 2008 Jun;12(3):914-27
Journal of cellular and molecular medicine 2008 Jun;12(3):914-27
Electrophysiological recording of re-aggregating brain cell cultures on multi-electrode arrays to detect acute neurotoxic effects.
van Vliet E, Stoppini L, Balestrino M, Eskes C, Griesinger C, Sobanski T, Whelan M, Hartung T, Coecke S
Neurotoxicology 2007 Nov;28(6):1136-46
Neurotoxicology 2007 Nov;28(6):1136-46
Enzymatic properties and localization of motopsin (PRSS12), a protease whose absence causes mental retardation.
Mitsui S, Yamaguchi N, Osako Y, Yuri K
Brain research 2007 Mar 9;1136(1):1-12
Brain research 2007 Mar 9;1136(1):1-12
Translational control of glial glutamate transporter EAAT2 expression.
Tian G, Lai L, Guo H, Lin Y, Butchbach ME, Chang Y, Lin CL
The Journal of biological chemistry 2007 Jan 19;282(3):1727-37
The Journal of biological chemistry 2007 Jan 19;282(3):1727-37
Differential localization of ErbB receptor ensembles influences their signaling in hippocampal neurons.
Chen J, Tseng HC, Dichter MA, Zhang H, Greene MI
DNA and cell biology 2005 Sep;24(9):553-62
DNA and cell biology 2005 Sep;24(9):553-62
Immunolocalization of MAP-2 in routinely formalin-fixed, paraffin-embedded guinea pig brain sections using microwave irradiation: a comparison of different combinations of antibody clones and antigen retrieval buffer solutions.
Kan RK, Pleva CM, Hamilton TA, Petrali JP
Microscopy and microanalysis : the official journal of Microscopy Society of America, Microbeam Analysis Society, Microscopical Society of Canada 2005 Apr;11(2):175-80
Microscopy and microanalysis : the official journal of Microscopy Society of America, Microbeam Analysis Society, Microscopical Society of Canada 2005 Apr;11(2):175-80
Involvement of glial cells in the neurotoxicity of parathion and chlorpyrifos.
Zurich MG, Honegger P, Schilter B, Costa LG, Monnet-Tschudi F
Toxicology and applied pharmacology 2004 Dec 1;201(2):97-104
Toxicology and applied pharmacology 2004 Dec 1;201(2):97-104
Fetal human neural progenitors can be the target for tumor transformation.
Wang Y, Bai Y, Li X, Hu Q, Lin C, Xiao Z, Liu Y, Xu J, Shen L, Li L
Neuroreport 2004 Aug 26;15(12):1907-12
Neuroreport 2004 Aug 26;15(12):1907-12
Altered levels and distribution of microtubule-associated proteins before disease onset in a mouse model of amyotrophic lateral sclerosis.
Farah CA, Nguyen MD, Julien JP, Leclerc N
Journal of neurochemistry 2003 Jan;84(1):77-86
Journal of neurochemistry 2003 Jan;84(1):77-86
Involvement of microglia-neuron interactions in the tumor necrosis factor-alpha release, microglial activation, and neurodegeneration induced by trimethyltin.
Eskes C, Juillerat-Jeanneret L, Leuba G, Honegger P, Monnet-Tschudi F
Journal of neuroscience research 2003 Feb 15;71(4):583-90
Journal of neuroscience research 2003 Feb 15;71(4):583-90
Simultaneous suppression of cdc2 and cdk2 activities induces neuronal differentiation of PC12 cells.
Dobashi Y, Shoji M, Kitagawa M, Noguchi T, Kameya T
The Journal of biological chemistry 2000 Apr 28;275(17):12572-80
The Journal of biological chemistry 2000 Apr 28;275(17):12572-80
Suppression of sprouting: An early function of NMDA receptors in the absence of AMPA/kainate receptor activity.
Lin SY, Constantine-Paton M
The Journal of neuroscience : the official journal of the Society for Neuroscience 1998 May 15;18(10):3725-37
The Journal of neuroscience : the official journal of the Society for Neuroscience 1998 May 15;18(10):3725-37
Suppression of sprouting: An early function of NMDA receptors in the absence of AMPA/kainate receptor activity.
Lin SY, Constantine-Paton M
The Journal of neuroscience : the official journal of the Society for Neuroscience 1998 May 15;18(10):3725-37
The Journal of neuroscience : the official journal of the Society for Neuroscience 1998 May 15;18(10):3725-37
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-MAP2 Monoclonal Antibody (AP18) (Product # MA5-12826) and a 160 kDa band corresponding to MAP2 was observed across SH-SY5Y and HEK-293. Whole Cell Extract-WCL (30 µg lysate) of SH-SY5Y (Lane 1), HEK-293 (Lane 2), Hep G2 (Lane 3), Jurkat (Lane 4), BeWo (Lane 5) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (0.4 µg/mL) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using SuperSignal™ West Dura Extended Duration Substrate (Product # 34076).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
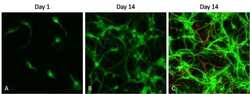
- Experimental details
- Immunofluorescent analysis of microtubule-associated protein 2 (MAP2, green) in cultured primary cortical neurons at different developmental stages (day 1 and day 14). Primary cortical neurons were isolated and cultured using the Primary Neuron Isolation Kit (Product # 88280). Neurons were fixed with 4% paraformaldehyde, permeablilized with 0.1% Triton X-100 in HBSS for 10 minutes at room temperature, and blocked with 3% BSA in PBS (Product # 37525) for 30 minutes at room temperature. Cells were probed with a MAP2 monoclonal antibody, clone AP18 (Product # MA5-12826) at a dilution of 1:500 for 2 hours at room temperature (or overnight at 4°C), washed with HBSS, and incubated with a DyLight 488 goat anti-mouse IgG (Product # 35502) secondary antibody at a dilution of 1:500 for 1 hour at room temperature (panels A and B). In panel C, cells were also probed with a glial fibrillary acidic protein (GFAP) polyclonal antibody at a dilution of 1:500 for 2 hours at room temperature (or overnight at 4°C), washed with HBSS, and incubated with a DyLight 680 goat anti-rabbit IgG (Product # 35568) secondary antibody at a dilution of 1:500 for 1 hour at room temperature. Images were taken at 20X magnification on a Carl Zeiss microscope (AxioVision Rel. 4.7).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
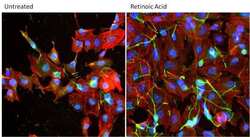
- Experimental details
- Immunofluorescent analysis of MAP2 a, b, c (green) in SH-SY5Y cells either left untreated (left panel) or treated with 10uM all-trans retinoic acid for 72 hours (right panel). Formalin fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 10 minutes at room temperature and blocked with 1% Blocker BSA (Product # 37525) for 15 minutes at room temperature. Cells were probed with a MAP2 a, b, c monoclonal antibody (Product # MA5-12826) at a dilution of 1:200 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat anti-mouse IgG secondary antibody (Product # 35502) at a dilution of 1:400 for 30 minutes at room temperature. F-Actin (red) was stained with DyLight 554 Phalloidin (Product # 21834) and nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ArrayScan or a ToxInsight Instrument at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
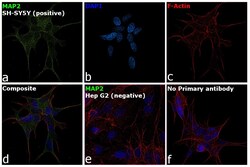
- Experimental details
- Immunofluorescence analysis of MAP2 was performed using 70% confluent log phase SH-SY5Y cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with MAP2 Monoclonal Antibody (AP18) (Product # MA5-12826) at 1:500 dilution in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766, 1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing cytoplasmic localization. Panel e represents Hep G2 cells having no expression of MAP2. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
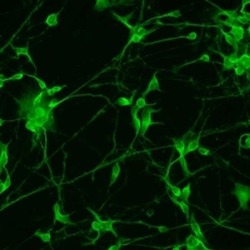
- Experimental details
- Immunofluorescence analysis of Map2 (green) in human glutamate neurons derived from iPSCs. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.2% Triton X-100 in PBS for 10 minutes, and blocked with 5% donkey serum in PBS for 15 minutes at room temperature. Cells were stained with a Map2 monoclonal antibody (Product # MA5-12826) diluted at 1:1000 in 5% donkey serum overnight at 4°C, and then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:1000 for 1 hour at room temperature (green). Note: Data courtesy of Innovators Program.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Formalin-fixed, paraffin-embedded human brain stained with Microtubulessociated Protein 2a, b, c antibody using peroxidase-conjugate and AEC chromogen. Note cytoplasmic staining of neurons and dendrites.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
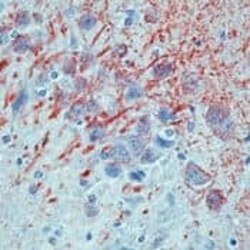
- Formalin-fixed, paraffin-embedded rat brain stained with MAP antibody using peroxidase-conjugate and AEC chromogen. Note cytoplasmic staining of neurons and fibers.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
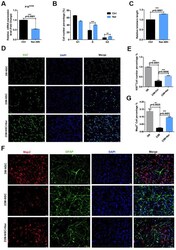
- Figure 3 Effect of naringenin (Nar) on senescence of mouse neural stem cells (mNSCs). ( A ) p16 ink4a mRNA expression was measured by qRT-PCR in 23M-NSCs with and without treatment of 6.8 mug/mL Nar for 48 h. ( B ) Cell cycle phase distributions of 23M-NSCs treated with Nar for 48 h and control cells. ( C ) The relative telomere length of 23M-NSCs increased significantly with Nar treatment. ( D ) Immunofluorescence Ki67 staining of mNSCs treated with Nar for 48 h, with DAPI nuclear labeling. ( E ) Quantification of ( D ). ( F ) Representative fields of MAP2 and GFAP immunofluorescence staining of cultured mNSCs after control and Nar treatment. ( G ) Quantification of ( F ). Data are presented as the mean +- SD of three independent experiments. * P < 0.05, ** P < 0.01, and *** P < 0.0001 compared with untreated cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
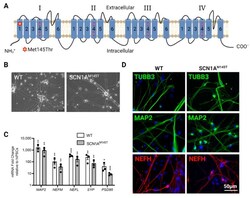
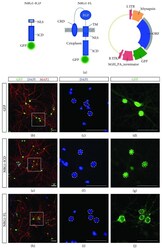
- Characterization of idNs. ( A ) Representation of Na V 1.1 channel. The star in segment 1 of domain I shows the localization of the mutated aminoacid (Met145Thr). The relative missense mutation c.434T > C is found in the exon 3 of the translated sequence. ( B ) Bright-field images of idNs from WT and SCN1A M145T -iPSCs (20x magnification). ( C ) Differentiated idNs show high expression levels of neuronal specific genes such as MAP2 , NEFM , NEFL, SYP and PSD95 compared to their undifferentiated counterparts (iPSCs). GAPDH was used as a housekeeping control. Data are presented as mean +- SEM of three biological replicates (black dots), * p < 0.05, ** p < 0.01, *** p < 0.001, t -test has been calculated vs. expression in iPSCs. ( D ) Immunostaining of neuronal markers TUBB3 (neurites marker), MAP2 (cell body and dendrites marker), and NEFH (axonal marker) in WT and SCN1A M145T idNs. DAPI nuclear counterstain is shown in all images in blue (63x magnification).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
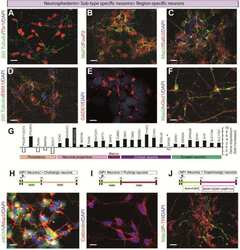
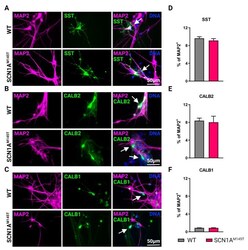
- Types of interneurons generated from iPSCs. Immunofluorescence analysis of idNs stained with antibodies against interneuronal subtypes markers ( A ) somatostatin (SST), ( B ) calretinin (CALB2), and ( C ) calbindin (CALB1). In each group of images, WT cells are shown in the upper panel, while SCN1A M145T idNs are shown in the lower panel. White arrows in the merged images indicate neurons expressing the interneuronal makers indicated (63x magnification). ( D - F ) Quantification of percentage of MAP2 + neurons co-expressing interneuronal markers immunostained in panels ( A - C ). About 9-1% of idNs express SST and CALB2, while CALB1 is present in less than 1% percent of neurons. At least 200 cells were counted for each bar, and data are presented as mean +- SEM of two independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
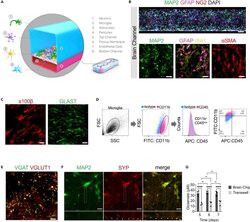
- Reconstruction of the neurovascular unit in the Brain-Chip (A) Schematic illustration of the Brain-Chip, a two-channel microengineered chip including iPSC-derived brain endothelial-like cells cultured on all surfaces of the bottom channel, and iPSC-derived Glutamatergic and GABAergic neurons, primary human brain astrocytes, pericytes, and microglia on the surface of the top channel. (B) Confocal images of the cell coverage in the brain channel on day 7 of culture. Top image: Immunofluorescence staining of the brain channel including MAP2 (green), GFAP (magenta), NG2 (red), and DAPI (blue). Bottom images: Representative merged confocal image of the brain channel on culture day 7, stained for neurons (MAP2, green), astrocytes (GFAP, magenta, IBA1, yellow), and pericytes (alphaSMA, red) (bar, 50 mum). (C) Representative immunofluorescent staining for s100beta (red) and GLAST (green) (bar,100 mum). (D) FACS analysis of cell-specific markers of microglia: Total population of microglia within the brain channel (gray), CD11b-positive population (magenta), CD45-positive population (magenta), quantification of CD11b:CD45-positive cells. (E) Representative merged confocal image of the brain channel co-stained with VGAT (green) for GABAergic neurons and VGlut1 (red) for Glutamatergic neurons (bar, 100 mum). (F) Immunofluorescence staining of the brain channel including MAP2 (green) and SYP (red) (bar, 100 mum). (G) Levels of secreted glutamate in the brain channel on culture days 5, 6,
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
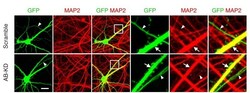
- Figure 4--figure supplement 1. The number of axons is not affected by Drp1 ABCD knockdown. Cultured hippocampal neurons were transfected at 3 weeks with the indicated shRNA plasmids carrying GFP as a cytosolic marker. Cells were subjected to immunofluorescence microscopy with anti-MAP2 antibodies. Arrowheads indicate axons, and arrows indicate dendrites. Bar, 20 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 2 Generation and validation of viral vectors for Nrg1 expression. (a) Schematic representation of the constructs for the expression of Nrg1-ICD (including the nuclear localization signal (NLS)) and Nrg1-FL both fused to GFP. The schema of the vector for the production of AAV particles to express Nrg1 under human Synapsin promoter is depicted on the right. CRD: cysteine-rich domain; EGF: epithelial growth factor domain; TM: transmembrane domain; ITR: inverted terminal repeat; ORF: open reading frame; hGH PA: human growth hormone polyadenylation signal. (b-j) Immunofluorescent labeling of primary cortical neurons infected to express GFP, Nrg1-ICD, or Nrg1-FL and fixed at D14. The neurons were labeled with DAPI and microtubule-associated protein 2 (MAP2) to visualize neuronal dendrites. The boxed area in (b, e, h) depicts the area magnified in (d, g, j). The dotted lines delimit the nuclei to highlight the specific localization of Nrg1 constructs. Scale bar, 50 mu m.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
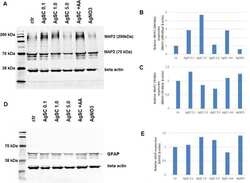
- Figure 3 Western blot results of neuroblastoma cell protein expression under control, OGD/R conditions, and OGD/R conditions treated with exogenous MIF or MIF antagonist ISO-1. (A) The expression levels of immature and mature BDNF and MAP2 in the MIF group were significantly higher than those in the OGD/R group (immature BDNF: P < 0.05; mature BDNF: P < 0.01). (C) The expression levels of Bcl2, Caspase-3, and Bax in the MIF group were significantly lower than those in the OGD/R group (Caspase-3: P < 0.01; Bax: P = 0.02). The expression levels of Bcl2 and MAP2 were significantly higher in the MIF group and lower in the ISO-1 group than those in the OGD/R group (Bcl2: P < 0.01; MAP2: P < 0.05). The expression levels of Caspase-3 and Bax were significantly lower in the MIF group and higher in the ISO-1 group than those in the OGD/R group (Caspase-3: P < 0.01; Bax: P = 0.02). (B and D) The representative bands of each group in western blot are presented. * P < 0.05, ** P < 0.01, vs . OGD/R group; one-way analysis of variance followed by the Bonferroni post-hoc analysis (number of independent cell culture experiments ( n ): immature BDNF, n = 5; mature BDNF, n = 6; Bcl2, n = 6, MAP2, n = 8; Caspase-3, n = 7; Bax, n = 8). BDNF: Brain-derived neurotrophic factor; ISO-1: MIF antagonist; MAP2: microtubule-associated protein 2; MIF: macrophage migration inhibitory factor recombinant; OGD/R: oxygen-glucose deprivation and reperfusion.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot