MA3-004
antibody from Invitrogen Antibodies
Targeting: VCP
CDC48, IBMPFD, p97, TERA
 Western blot
Western blot Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [34]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [4]
- Immunohistochemistry [2]
- Other assay [44]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA3-004 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- VCP Monoclonal Antibody (5)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Description
- MA3-004 detects VCP protein in human, mouse, and rat samples. MA3-004 has successfully been used in Western blot immunoprecipitation, and immunohistochemical procedures. By Western blot, this antibody detects an ~97 kDa protein representing VCP from total lysate of cultured human B cells. The MA3-004 immunogen is a synthetic peptide corresponding to residues C G(792) G S V Y T E D N D D D L Y G(806) of mouse VCP. This peptide (Cat. # PEP-239) is available for use in neutralization and control experiments.
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 5
- Vial size
- 100 µg
- Concentration
- 1.0 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references VCP/p97 cofactor UBXN1/SAKS1 regulates mitophagy by modulating MFN2 removal from mitochondria.
An autopsy case of amyotrophic lateral sclerosis with striatonigral and pallidoluysian degeneration and cat's-eye-shaped neuronal nuclear inclusions.
Impacts of p97 on Proteome Changes in Human Cells during Coronaviral Replication.
Functional Inhibition of Valosin-Containing Protein Induces Cardiac Dilation and Dysfunction in a New Dominant-Negative Transgenic Mouse Model.
Ubiquitination of G3BP1 mediates stress granule disassembly in a context-specific manner.
Site-Specific Photo-Crosslinking Proteomics Reveal Regulation of IFITM3 Trafficking and Turnover by VCP/p97 ATPase.
Parkin Coordinates Platelet Stress Response in Diabetes Mellitus: A Big Role in a Small Cell.
ULK1 and ULK2 Regulate Stress Granule Disassembly Through Phosphorylation and Activation of VCP/p97.
Parkin is a disease modifier in the mutant SOD1 mouse model of ALS.
UBXD1 is a mitochondrial recruitment factor for p97/VCP and promotes mitophagy.
Dioxins and related environmental contaminants increase TDP-43 levels.
An aberrant phase transition of stress granules triggered by misfolded protein and prevented by chaperone function.
Novel eosinophilic neuronal cytoplasmic inclusions in the external cuneate nucleus of humans.
The Myoblast C2C12 Transfected with Mutant Valosin-Containing Protein Exhibits Delayed Stress Granule Resolution on Oxidative Stress.
Pathogenic Mutations in the Valosin-containing Protein/p97(VCP) N-domain Inhibit the SUMOylation of VCP and Lead to Impaired Stress Response.
The aspartyl protease DDI2 activates Nrf1 to compensate for proteasome dysfunction.
PFKM gene defect and glycogen storage disease GSDVII with misleading enzyme histochemistry.
Ubiquitin-negative, eosinophilic neuronal cytoplasmic inclusions associated with stress granules and autophagy: an immunohistochemical investigation of two cases.
Ubiquitin-negative, eosinophilic neuronal cytoplasmic inclusions associated with stress granules and autophagy: an immunohistochemical investigation of two cases.
Eukaryotic stress granules are cleared by autophagy and Cdc48/VCP function.
Amyotrophic lateral sclerosis (ALS)-associated VAPB-P56S inclusions represent an ER quality control compartment.
Valosin-containing protein immunoreactivity in tauopathies, synucleinopathies, polyglutamine diseases and intranuclear inclusion body disease.
Neuronal-specific overexpression of a mutant valosin-containing protein associated with IBMPFD promotes aberrant ubiquitin and TDP-43 accumulation and cognitive dysfunction in transgenic mice.
VCP is essential for mitochondrial quality control by PINK1/Parkin and this function is impaired by VCP mutations.
SEL1L protein critically determines the stability of the HRD1-SEL1L endoplasmic reticulum-associated degradation (ERAD) complex to optimize the degradation kinetics of ERAD substrates.
Herp regulates Hrd1-mediated ubiquitylation in a ubiquitin-like domain-dependent manner.
Developmental expression of p97/VCP (Valosin-containing protein) and Jab1/CSN5 in the rat testis and epididymis.
Different p97/VCP complexes function in retrotranslocation step of mammalian ER-associated degradation (ERAD).
Differential regulation of CFTRDeltaF508 degradation by ubiquitin ligases gp78 and Hrd1.
VCP/p97 is essential for maturation of ubiquitin-containing autophagosomes and this function is impaired by mutations that cause IBMPFD.
VCP/p97 is essential for maturation of ubiquitin-containing autophagosomes and this function is impaired by mutations that cause IBMPFD.
Nuclear localization of valosin-containing protein in normal muscle and muscle affected by inclusion-body myositis.
IgE modulates neutrophil survival in asthma: role of mitochondrial pathway.
Selective inhibition of endoplasmic reticulum-associated degradation rescues DeltaF508-cystic fibrosis transmembrane regulator and suppresses interleukin-8 levels: therapeutic implications.
Mengus C, Neutzner M, Bento ACPF, Bippes CC, Kohler C, Decembrini S, Häusel J, Hemion C, Sironi L, Frank S, Scholl HPN, Neutzner A
Autophagy 2022 Jan;18(1):171-190
Autophagy 2022 Jan;18(1):171-190
An autopsy case of amyotrophic lateral sclerosis with striatonigral and pallidoluysian degeneration and cat's-eye-shaped neuronal nuclear inclusions.
Kon T, Mori F, Kinoshita I, Nakamura T, Nishijima H, Suzuki C, Goto S, Kijima H, Tomiyama M, Wakabayashi K
Neuropathology : official journal of the Japanese Society of Neuropathology 2022 Aug;42(4):329-338
Neuropathology : official journal of the Japanese Society of Neuropathology 2022 Aug;42(4):329-338
Impacts of p97 on Proteome Changes in Human Cells during Coronaviral Replication.
Cheng KW, Li S, Wang F, Ruiz-Lopez NM, Houerbi N, Chou TF
Cells 2021 Oct 29;10(11)
Cells 2021 Oct 29;10(11)
Functional Inhibition of Valosin-Containing Protein Induces Cardiac Dilation and Dysfunction in a New Dominant-Negative Transgenic Mouse Model.
Sun X, Zhou N, Ma B, Wu W, Stoll S, Lai L, Qin G, Qiu H
Cells 2021 Oct 26;10(11)
Cells 2021 Oct 26;10(11)
Ubiquitination of G3BP1 mediates stress granule disassembly in a context-specific manner.
Gwon Y, Maxwell BA, Kolaitis RM, Zhang P, Kim HJ, Taylor JP
Science (New York, N.Y.) 2021 Jun 25;372(6549):eabf6548
Science (New York, N.Y.) 2021 Jun 25;372(6549):eabf6548
Site-Specific Photo-Crosslinking Proteomics Reveal Regulation of IFITM3 Trafficking and Turnover by VCP/p97 ATPase.
Wu X, Spence JS, Das T, Yuan X, Chen C, Zhang Y, Li Y, Sun Y, Chandran K, Hang HC, Peng T
Cell chemical biology 2020 May 21;27(5):571-585.e6
Cell chemical biology 2020 May 21;27(5):571-585.e6
Parkin Coordinates Platelet Stress Response in Diabetes Mellitus: A Big Role in a Small Cell.
Lee SH, Du J, Hwa J, Kim WH
International journal of molecular sciences 2020 Aug 15;21(16)
International journal of molecular sciences 2020 Aug 15;21(16)
ULK1 and ULK2 Regulate Stress Granule Disassembly Through Phosphorylation and Activation of VCP/p97.
Wang B, Maxwell BA, Joo JH, Gwon Y, Messing J, Mishra A, Shaw TI, Ward AL, Quan H, Sakurada SM, Pruett-Miller SM, Bertorini T, Vogel P, Kim HJ, Peng J, Taylor JP, Kundu M
Molecular cell 2019 May 16;74(4):742-757.e8
Molecular cell 2019 May 16;74(4):742-757.e8
Parkin is a disease modifier in the mutant SOD1 mouse model of ALS.
Palomo GM, Granatiero V, Kawamata H, Konrad C, Kim M, Arreguin AJ, Zhao D, Milner TA, Manfredi G
EMBO molecular medicine 2018 Oct;10(10)
EMBO molecular medicine 2018 Oct;10(10)
UBXD1 is a mitochondrial recruitment factor for p97/VCP and promotes mitophagy.
Bento AC, Bippes CC, Kohler C, Hemion C, Frank S, Neutzner A
Scientific reports 2018 Aug 17;8(1):12415
Scientific reports 2018 Aug 17;8(1):12415
Dioxins and related environmental contaminants increase TDP-43 levels.
Ash PEA, Stanford EA, Al Abdulatif A, Ramirez-Cardenas A, Ballance HI, Boudeau S, Jeh A, Murithi JM, Tripodis Y, Murphy GJ, Sherr DH, Wolozin B
Molecular neurodegeneration 2017 May 5;12(1):35
Molecular neurodegeneration 2017 May 5;12(1):35
An aberrant phase transition of stress granules triggered by misfolded protein and prevented by chaperone function.
Mateju D, Franzmann TM, Patel A, Kopach A, Boczek EE, Maharana S, Lee HO, Carra S, Hyman AA, Alberti S
The EMBO journal 2017 Jun 14;36(12):1669-1687
The EMBO journal 2017 Jun 14;36(12):1669-1687
Novel eosinophilic neuronal cytoplasmic inclusions in the external cuneate nucleus of humans.
Ito M, Nakamura K, Mori F, Miki Y, Tanji K, Wakabayashi K
Neuropathology : official journal of the Japanese Society of Neuropathology 2016 Oct;36(5):441-447
Neuropathology : official journal of the Japanese Society of Neuropathology 2016 Oct;36(5):441-447
The Myoblast C2C12 Transfected with Mutant Valosin-Containing Protein Exhibits Delayed Stress Granule Resolution on Oxidative Stress.
Rodriguez-Ortiz CJ, Flores JC, Valenzuela JA, Rodriguez GJ, Zumkehr J, Tran DN, Kimonis VE, Kitazawa M
The American journal of pathology 2016 Jun;186(6):1623-34
The American journal of pathology 2016 Jun;186(6):1623-34
Pathogenic Mutations in the Valosin-containing Protein/p97(VCP) N-domain Inhibit the SUMOylation of VCP and Lead to Impaired Stress Response.
Wang T, Xu W, Qin M, Yang Y, Bao P, Shen F, Zhang Z, Xu J
The Journal of biological chemistry 2016 Jul 1;291(27):14373-14384
The Journal of biological chemistry 2016 Jul 1;291(27):14373-14384
The aspartyl protease DDI2 activates Nrf1 to compensate for proteasome dysfunction.
Koizumi S, Irie T, Hirayama S, Sakurai Y, Yashiroda H, Naguro I, Ichijo H, Hamazaki J, Murata S
eLife 2016 Aug 16;5
eLife 2016 Aug 16;5
PFKM gene defect and glycogen storage disease GSDVII with misleading enzyme histochemistry.
Auranen M, Palmio J, Ylikallio E, Huovinen S, Paetau A, Sandell S, Haapasalo H, Viitaniemi K, Piirilä P, Tyynismaa H, Udd B
Neurology. Genetics 2015 Jun;1(1):e7
Neurology. Genetics 2015 Jun;1(1):e7
Ubiquitin-negative, eosinophilic neuronal cytoplasmic inclusions associated with stress granules and autophagy: an immunohistochemical investigation of two cases.
Mori F, Watanabe Y, Miki Y, Tanji K, Odagiri S, Eto K, Wakabayashi K
Neuropathology : official journal of the Japanese Society of Neuropathology 2014 Apr;34(2):140-7
Neuropathology : official journal of the Japanese Society of Neuropathology 2014 Apr;34(2):140-7
Ubiquitin-negative, eosinophilic neuronal cytoplasmic inclusions associated with stress granules and autophagy: an immunohistochemical investigation of two cases.
Mori F, Watanabe Y, Miki Y, Tanji K, Odagiri S, Eto K, Wakabayashi K
Neuropathology : official journal of the Japanese Society of Neuropathology 2014 Apr;34(2):140-7
Neuropathology : official journal of the Japanese Society of Neuropathology 2014 Apr;34(2):140-7
Eukaryotic stress granules are cleared by autophagy and Cdc48/VCP function.
Buchan JR, Kolaitis RM, Taylor JP, Parker R
Cell 2013 Jun 20;153(7):1461-74
Cell 2013 Jun 20;153(7):1461-74
Amyotrophic lateral sclerosis (ALS)-associated VAPB-P56S inclusions represent an ER quality control compartment.
Kuijpers M, van Dis V, Haasdijk ED, Harterink M, Vocking K, Post JA, Scheper W, Hoogenraad CC, Jaarsma D
Acta neuropathologica communications 2013 Jun 12;1:24
Acta neuropathologica communications 2013 Jun 12;1:24
Valosin-containing protein immunoreactivity in tauopathies, synucleinopathies, polyglutamine diseases and intranuclear inclusion body disease.
Mori F, Tanji K, Toyoshima Y, Sasaki H, Yoshida M, Kakita A, Takahashi H, Wakabayashi K
Neuropathology : official journal of the Japanese Society of Neuropathology 2013 Dec;33(6):637-44
Neuropathology : official journal of the Japanese Society of Neuropathology 2013 Dec;33(6):637-44
Neuronal-specific overexpression of a mutant valosin-containing protein associated with IBMPFD promotes aberrant ubiquitin and TDP-43 accumulation and cognitive dysfunction in transgenic mice.
Rodriguez-Ortiz CJ, Hoshino H, Cheng D, Liu-Yescevitz L, Blurton-Jones M, Wolozin B, LaFerla FM, Kitazawa M
The American journal of pathology 2013 Aug;183(2):504-15
The American journal of pathology 2013 Aug;183(2):504-15
VCP is essential for mitochondrial quality control by PINK1/Parkin and this function is impaired by VCP mutations.
Kim NC, Tresse E, Kolaitis RM, Molliex A, Thomas RE, Alami NH, Wang B, Joshi A, Smith RB, Ritson GP, Winborn BJ, Moore J, Lee JY, Yao TP, Pallanck L, Kundu M, Taylor JP
Neuron 2013 Apr 10;78(1):65-80
Neuron 2013 Apr 10;78(1):65-80
SEL1L protein critically determines the stability of the HRD1-SEL1L endoplasmic reticulum-associated degradation (ERAD) complex to optimize the degradation kinetics of ERAD substrates.
Iida Y, Fujimori T, Okawa K, Nagata K, Wada I, Hosokawa N
The Journal of biological chemistry 2011 May 13;286(19):16929-39
The Journal of biological chemistry 2011 May 13;286(19):16929-39
Herp regulates Hrd1-mediated ubiquitylation in a ubiquitin-like domain-dependent manner.
Kny M, Standera S, Hartmann-Petersen R, Kloetzel PM, Seeger M
The Journal of biological chemistry 2011 Feb 18;286(7):5151-6
The Journal of biological chemistry 2011 Feb 18;286(7):5151-6
Developmental expression of p97/VCP (Valosin-containing protein) and Jab1/CSN5 in the rat testis and epididymis.
Cayli S, Ocakli S, Erdemir F, Tas U, Aslan H, Yener T, Karaca Z
Reproductive biology and endocrinology : RB&E 2011 Aug 19;9:117
Reproductive biology and endocrinology : RB&E 2011 Aug 19;9:117
Different p97/VCP complexes function in retrotranslocation step of mammalian ER-associated degradation (ERAD).
Ballar P, Pabuccuoglu A, Kose FA
The international journal of biochemistry & cell biology 2011 Apr;43(4):613-21
The international journal of biochemistry & cell biology 2011 Apr;43(4):613-21
Differential regulation of CFTRDeltaF508 degradation by ubiquitin ligases gp78 and Hrd1.
Ballar P, Ors AU, Yang H, Fang S
The international journal of biochemistry & cell biology 2010 Jan;42(1):167-73
The international journal of biochemistry & cell biology 2010 Jan;42(1):167-73
VCP/p97 is essential for maturation of ubiquitin-containing autophagosomes and this function is impaired by mutations that cause IBMPFD.
Tresse E, Salomons FA, Vesa J, Bott LC, Kimonis V, Yao TP, Dantuma NP, Taylor JP
Autophagy 2010 Feb;6(2):217-27
Autophagy 2010 Feb;6(2):217-27
VCP/p97 is essential for maturation of ubiquitin-containing autophagosomes and this function is impaired by mutations that cause IBMPFD.
Tresse E, Salomons FA, Vesa J, Bott LC, Kimonis V, Yao TP, Dantuma NP, Taylor JP
Autophagy 2010 Feb;6(2):217-27
Autophagy 2010 Feb;6(2):217-27
Nuclear localization of valosin-containing protein in normal muscle and muscle affected by inclusion-body myositis.
Greenberg SA, Watts GD, Kimonis VE, Amato AA, Pinkus JL
Muscle & nerve 2007 Oct;36(4):447-54
Muscle & nerve 2007 Oct;36(4):447-54
IgE modulates neutrophil survival in asthma: role of mitochondrial pathway.
Saffar AS, Alphonse MP, Shan L, Hayglass KT, Simons FE, Gounni AS
Journal of immunology (Baltimore, Md. : 1950) 2007 Feb 15;178(4):2535-41
Journal of immunology (Baltimore, Md. : 1950) 2007 Feb 15;178(4):2535-41
Selective inhibition of endoplasmic reticulum-associated degradation rescues DeltaF508-cystic fibrosis transmembrane regulator and suppresses interleukin-8 levels: therapeutic implications.
Vij N, Fang S, Zeitlin PL
The Journal of biological chemistry 2006 Jun 23;281(25):17369-17378
The Journal of biological chemistry 2006 Jun 23;281(25):17369-17378
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
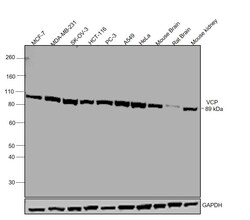
- Western blot was performed using Anti-VCP Polyclonal Antibody (Product # MA3-004) and a 89 kDa band corresponding to VCP was observed in across all tested cell lines and tissues. Whole cell extracts (30 µg lysate) of MCF-7 (Lane 1), MDA-MB-231 (Lane 2), SK-OV-3 (Lane 3), HCT-116 (Lane 4), PC-3 (Lane 5), A549 (Lane 6), HeLa (Lane 7), Mouse brain (Lane 8), Rat brain (Lane 9), Mouse Kidney (Lane 10) were electrophoresed using NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005)..
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Knockdown of VCP was achieved by transfecting MCF-7 with VCP specific siRNAs (Silencer® select Product # s14765). Western blot analysis (Fig. a) was performed using whole cell extracts from the VCP knockdown cells (Lane 3), non-specific scrambled siRNA transfected cells (Lane 2) and untransfected cells (Lane 1). The blot was probed with VCP Polyclonal Antibody (Product # MA3-004, 1:1000 dilution) and Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution). Densitometric analysis of this western blot is shown in histogram (Fig. b). Loss of signal upon siRNA mediated knock down confirms that antibody is specific to VCP.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Western blot was performed using Anti-VCP Polyclonal Antibody (Product # MA3-004) and a 89 kDa band corresponding to VCP was observed in across all tested cell lines and tissues. Whole cell extracts (30 µg lysate) of MCF-7 (Lane 1), MDA-MB-231 (Lane 2), SK-OV-3 (Lane 3), HCT-116 (Lane 4), PC-3 (Lane 5), A549 (Lane 6), HeLa (Lane 7), Mouse brain (Lane 8), Rat brain (Lane 9), Mouse Kidney (Lane 10) were electrophoresed using NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005)..
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of VCP using VCP Monoclonal antibody (5) (Product # MA3-004) shows staining in C6 glioma cells. VCP staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing VCP (Product # MA3-004) at a dilution of 1:20-1:200 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of VCP using VCP Monoclonal antibody (5) (Product # MA3-004) shows staining in HeLa cells. VCP staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing VCP (Product # MA3-004) at a dilution of 1:20-1:200 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of VCP using VCP Monoclonal antibody (5) (Product # MA3-004) shows staining in WiDr colon carcinoma cells. VCP staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing VCP (Product # MA3-004) at a dilution of 1:20-1:200 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of VCP was performed using HeLa cells. The cells were fixed and permeabilized with ice cold 100% Acetone for 5 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with VCP Monoclonal Antibody (5) (Product # MA3-004) at 5 microgram/mL concentration in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 (Product # A28175) for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry was performed on normal deparaffinized Human brain tissue tissues. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:1000 with a mouse monoclonal antibody recognizing Anti-VCP (Product # MA3-004) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
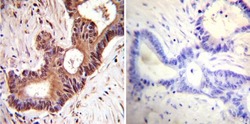
- Experimental details
- Immunohistochemistry was performed on cancer biopsies of deparaffinized Human colon carcinoma tissues. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:1000 with a mouse monoclonal antibody recognizing Anti-VCP (Product # MA3-004) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
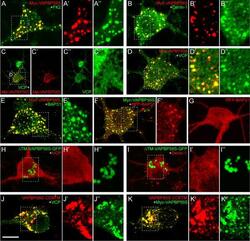
- Figure 5 UBXD1 recruits p97 to mitochondria under mitophagic conditions. ( A ) HeLa cells transfected with expression plasmids for FLAG-UBXD1 and mitoYFP-T2A-Parkin-myc3 were treated with CCCP for 6 hours or left untreated as control. Cells were fixed, stained using rabbit anti-FLAG and mouse anti-p97 antibodies and analyzed by confocal microscopy. ( B ) HeLa cells transfected with expression plasmids for FLAG-UBXD1 or vector control and mitoYFP-T2A-Parkin-myc3 were treated as in A. To quantify p97 redistribution to mitochondria, the ratio of mitochondrial p97 to total p97 was determined by image analysis of confocal images. Shown are box plots of three independent experiments with at least 15 cells per experiment and condition. Statistical significance was assessed by ANOVA followed by Student's t-test using Bonferroni correction to account for multiple comparisons. ***Denotes p-values < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 The UBX domain of UBXD1 is essential for mitochondrial translocation of p97. HeLa cells transfected with expression plasmids for FLAG-UBXD1 or variants of FLAG-UBXD1 and mitoYFP-T2A-Parkin-myc3 were treated with CCCP for 6 hours or left untreated as control. Fixed cells were stained using rabbit anti-FLAG and mouse anti-p97 antibodies and analyzed by confocal microscopy. The box plots represent three independent experiments with at least 15 cells/experiment/condition. Statistical significance was assessed by ANOVA followed by Student's t-test using Bonferroni correction to account for multiple comparisons. *denotes p-values < 0.05, ***p-values < 0.001, n.s. - no significant difference.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 The VIM domain of UBXD1 is not involved in mitochondrial translocation and p97 recruitment. HeLa cells transfected with expression plasmid for FLAG-VIMonly and mitoYFP-T2A-Parkin-myc3 were treated with CCCP for 6 hours or left untreated as control. Fixed cells were stained using rabbit anti-FLAG and mouse anti-p97 antibodies and analyzed by confocal microscopy. The box plots represent three independent experiments with at least 15 cells/experiment/condition. Statistical significance was assessed by ANOVA followed by Student's t-test using Bonferroni correction to account for multiple comparisons. n.s. - no significant difference.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 9 HeLa cells transfected with expression plasmids for mitochondria-targeted dsRED (mitodsRED) and YFP-UBXD1 or YFP fused to the mitochondrial membrane targeting signal ActA were fixed, stained using mouse anti-p97 antibodies, and analyzed by confocal microscopy. Shown are representative images out of three independent experiments. The box plots represent three independent experiments with at least 15 cells/experiment/condition. Statistical significance was assessed by by Student's t-test. ***denotes p < 0.001, n.s. - no significant difference.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
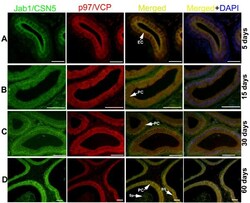
- Figure 1 Localisation of p97/VCP and Jab1/CSN5 in the developing rat testis . Immunohistochemistry was used to show the cellular localisation of p97/VCP and Jab1/CSN5 in the testis at days 5 (A, G), 15 (B, H), 30 (C, D, I), and 60 (E, F, J, K, L) after birth. The negative controls (A, C, G, H, L, inserts). A: Positive staining of p97/VCP expression was demonstrated in the nuclear and cytoplasmic regions of gonocytes (GC) at day 5. Interstitial cells (I) show weak immunoreactivity, and peritubuler cells (P) present no immunoreactivity. B: p97/VCP is highly expressed in spermatogonia (Sg), spermatocytes (Sc) and Sertoli cells (Se) at day 15. Some I show moderate immunostaining for p97/VCP. C, D: Elongating (eSt) and round (rSt) spermatids, Se and Sg show moderate to strong immunostaining for p97/VCP, while cytoplasm of Sc are moderately labelled with p97/VCP. E, F: In the adult testis, p97/VCP is weakly to moderately expressed in the cytoplasm of Sc, whereas Se, Sg, rSt and eSt are moderate to strong immunopositive for p97/VCP. G: Moderate Jab1/CSN5 expression is seen at day 5 in GC. H: Jab1/CSN5 is strongly expressed in Sg and Sc while Se exhibit moderate expression at day 15. I: Sg, Sc, eSt and Se show moderate to strong immunostaining for Jab1/CSN5. J-L: In the adult testis, Jab1/CSN5 is strongly expressed in Sg and Sc, while rSt and eSt show weak to moderate immunostaining for Jab1/CSN5. Roman numerals indicate stages of the seminiferous epithelial cycle. M: H-SCORE of the
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Immunohistochemical analysis of Sertoli cells at day 5 (A), 15 (B), 60 (C) and colocalisation of p97/VCP - p27 kip1 (D-F) and Jab1/CSN5 - p27 kip1 (G-I) in rat testis . Cytokeratin 18 (CK) are detected in immature Sertoli cells (Se) at day 15 (A) and p27 kip1 is observed in mature Sertoli cells (Se) of day 30 and 60 (B, C) in rat testis. Mature Sertoli cells (Se) are immunopositive for p27 kip1 (green, D, G), p97/VCP (red, E) and Jab1/CSN5 (red, H), resulting in a yellow color (overlay, F, I). Scale bars: 25 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Distribution of p97/VCP in different regions of the rat epididymis during postnatal development . p97/VCP immunopositivity is detected in the CT, CS and CA of the epididymis at days 5 (A, B, C) , 15 (D, E, F) , 30 (G, H, I) and 60 (J, K, L) . p97/VCP expression is seen in the epithelial cells (EC) of the CT, CS and CA epididymis at day 5 (A, B, C). No significant staining is observed in the negative controls (at the corner of panels A-L). p97/VCP is localised both in the basal (BS) and principal (PC) cells of the epididymis at days 15, 30 and 60 (D-L). Of note, p97/VCP is also expressed in the apical layer of the cytoplasm in all epididymal regions at days 30 and 60 (G-L). Immunopositivity for p97/VCP is detected both in the principal cells and basal epithelial cells, while clear cells (C) show no immunoreactivity at day 60 (J-L). Spermatozoa (Sp) are weakly immunopositive for p97/VCP. CT: caput, CS: corpus, CA: cauda. Scale bars: 25 mum (A-C); scale bars: 50 mum (D-L).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Double immunohistochemistry representing the colocalisation of p97/VCP and Jab1/CSN5 in the developing rat testis (A-D) . Detection of Jab1/CSN5 (green), p97/VCP (red), and double-stained (yellow) positive cells merged with DAPI nuclear staining on day 5 (A), day 15 (B), day 30 (C) and day 60 (D). A: Day 5 gonocytes (GC) are double labelled with p97/VCP and Jab1/CSN5. B: Double-labelled spermatocytes (Sc), spermatogonia (Sg) and Sertoli cells (Se) are shown on day 15, and the nuclear regions of some spermatocytes are only positive for Jab1/CSN5. C: Jab1/CSN5 and p97/VCP positivity (yellow) is observed in spermatocytes (Sc), spermatogonia (Sg), Sertoli cells (Se) and elongating spermatids (eSt) on day 30. D: Double staining (yellow) is detected in the cytoplasm of spermatocytes (Sc), the nuclear region of spermatogonia (Sg) and elongating spermatids (eSt) and Sertoli cells (Se) in the 60-day-old rat testis. Scale bars: 25 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 6 Colocalisations of Jab1/CSN5 and p97/VCP in the developing rat epididymis (A-D) . Detection of Jab1/CSN5 (green), p97/VCP (red), and double (yellow) positive cells merged with DAPI nuclear staining on day 5 (A), 15 (B), 30 (C) and 60 (D) CS epididymis. Both the basal (BS) and principal cells (PC) of CS epididymis are immunopositive for Jab1/CSN5 (green) and for p97/VCP (red), resulting a yellow color. Spermatozoa (Sp) are weakly immunopositive for Jab1/CSN5 and p97/VCP on day 60. Scale bars: 25 mum (A), 50 mum (B-D).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Mutant VAPB inclusions are ubiquitin-positive and enriched in ERAD components. A - C ) Representative confocal images of the distribution of ubiquitinated epitopes labeled with the FK2 antibody in motor neurons of mutant VAPB line VM1 with ( A ) or without ( B ) inclusions, and non-transgenic littermates ( C ). Note high level of FK2-immunoreactivity in inclusions ( A ) and diffuse increase as compared to non-transgenic motor neurons in VM1 motor neurons without inclusions ( B ). D ) Bar graph of FK2-immunofluoresencesignal in non-transgenic versus VM1 and VM3 motor neurons without inclusions. Values are means +- SE and are from lumbar L4 segments from 4 non-transgenic, 2 VM1 and 2 VM3 mice embedded in a single gelatin block (see methods). E - I ) Confocal image of mutant VAPB (line VM1, E - G ) or non-transgenic ( H, I ) spinal motor neurons double-labeled for VAPB or HA and ERAD components, i.e. VCP/p97 ( E , I ), derlin-1 ( F , H ), and BAP31 ( G ). Note, enrichment of VCP ( E ) and BAP31 ( G ) staining in the mutant VAPB inclusions; derlin-1 although present does not show preferential enrichment is not selectively enriched in the mutant VAPB inclusions. Scale bars: 10 mum ( A , H ), 25 mum ( E ).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 7 Mutant VAPB inclusions in primary cultured hippocampal neurons are enriched in ERAD components. A - E ) Maximal projections of confocal stacks of primary cultured hippocampal neurons transfected with Myc-VAPB-P56S showing intense staining of ubiquitinated epitopes (FK2, A ), derlin-1 ( B ), VCP ( C , D ) and BAP31 ( E ) in mutant VAPB inclusions. F , G ) Co-expression of Myc-VAPB-P56S and RFP-BAP31 shows that mutant VAPB inclusions recruits a large portion of RFP-BAP31 ( F ), while in single RFP-BAP31 transfected neurons, RFP-BAP31 has a diffuse distribution ( G ). H , I ) Hippocampal neurons transfected with GFP-tagged VAPB-P56S lacking the transmembrane domain (DeltaTM) develop inclusions that are immunonegative for VCP ( H ) and Derlin-1 ( I ). J , K ) Transfection of hippocampal neurons with VAPB-P56S-CD8TM with ( K ) or without ( J ) myc-VAPB-P56S (green) shows that VAPB-P56S with the transmembrane domain of CD8 (immunostained with anti-CD8 antibody) accumulates in the same inclusions as VAPB-P56S. Scale bar, 10 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. DDI2 is involved in Nrf1 processing and translocation to the nucleus. ( A ) Representative images of Nrf1 localization. HEK293A cells were transfected with a non-targeting control (negative control), DDI2, or p97 siRNA and then treated with 50 nM bortezomib for 14 hr before fixation. ( B ) Immunoblotting of whole-cell lysates of cells in ( A ) treated with or without bortezomib. Nrf1 is detected as three different forms; a glycosylated form (G), full-length form (FL), and processed form (P). ( C ) Immunoblotting of Nrf1 after deglycosylation treatment. HEK293A cells were transfected with DDI2 or p97 siRNA, followed by transfection with Nrf1-3xFlag, and then treated with or without 50 nM bortezomib. The cell lysates were treated with or without Endo H. DeG denotes deglycosylated Nrf1. DOI: http://dx.doi.org/ Figure 2--figure supplement 1. The expression and localization of DDI2 were not affected by bortezomib treatment. ( A ) Relative mRNA expression of DDI2 in HCT116 cells treated with or without bortezomib (50 nM, 14 hr). The data represent mean + standard error of the mean (SEM) (n = 3, biological replicates). ( B ) Subcellular localization of DDI2. HEK293A cells stably expressing Venus-Sec61beta (ER marker) were transfected with Flag-DDI2 and treated with or without bortezomib. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Confirmation of interactions between Parkin and selected target proteins in human and murine platelets. ( A ) Immunoprecipitation of each specific antibody in pooled human DM platelets (4 HC, 5DM1, and 6DM2). We incubated 500 mug protein lysates incubated with specific antibodies overnight at 4 degC with 10% input as control. G1 represents interacting protein groups from LC-MS/MS results. G2 represents the non-interacting proteins group in LC-MS/MS results. G3 represents protein that interacted with Parkin and that were not found in the LC-MS/MS results. ( B ) Immunoprecipitation of each specific antibody in pooled murine platelets (3 WT and 5DM). We incubated 500 mug protein lysates incubated with specific antibodies overnight at 4 degC with 10% input as control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
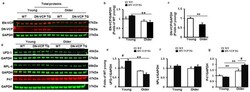
- Figure 1 Identification and characterization of cardiac-specific VCP dominant-negative transgenic mouse (DN-VCP TG) at the age of 10 weeks. ( a ). The construct of the cardiac-specific DN-VCP TG mouse model; ( b , c ). Identification of DN-VCP TG by genotyping with PCR ( b ) and protein expression in the hearts by Western blots ( c ); ( d ). the relative value of endogenous VCP (EN-VCP) and overexpressed DN-VCP in TG and WT mice of both genders; ( e - h ). The morphology and function of DN-VCP TG vs. WT by echocardiography at a young age, in terms of left ventricle (LV) wall thickness including LV anterior and posterior wall thickness at end-diastole and end-systole (LVAWd, LVAWs, LVPWd, and LVPWs) ( e ), LV internal end-diastolic and end-systolic dimensions (LVEDd and LVEDs) ( f ), heart rates ( g ), and contractility reflected by LV ejection fraction (EF) and fractional shortening (FS) ( h ). **, p < 0.01 vs. age-matched WT mice. N = 11-12/group; ( i ). The examples of the heart shape and size of DN-VCPTG vs. WT mice at the age of 10 weeks.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 3 DN-VCP aggravates the age-related alterations in EN-VCP and its co-factors in TG mouse hearts. ( a ). The representative images of Western blots reflect the total protein levels of endogenous VCP (EN-VCP) and dominant-negative VCP (DN-VCP) in DN-VCP TG mice compared to their age-matched WT mice at both young and older ages. GAPDH was used as a loading control; ( b , c ). The quantitated values of the total proteins of EN-VCP ( b ) and DN-VCP ( c ) normalized to the GAPDH in the hearts; ( d ). The representative Western blots reflect the total protein levels of VCP's co-factors in DN-VCP TG mice compared to their age-matched WT mice. GAPDH was used as a loading control of the total proteins; ( e - g ). The relative total protein value of the VCP-cofactors, ubiquitin fusion degradation protein 1 (UFD-1) ( e ), nuclear protein localization protein 4 (NPL-4) ( f ), and P47 ( g ), in TG and WT mice. N = 4/group. **, p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 DN-VCP impairs its nuclear localization and attenuates the age-related cytoplasmic/nuclear shuffling of the EN-VCP and its co-factors/adaptor in TG mouse hearts. ( a , b ). The subcellular distributions of overexpressed full-length VCP (FL-VCP) and dominant-negative VCP (DN-VCP) in the cytoplasmic and nuclear fractions of the cells, with a specific antibody against the tag-protein-Flag, by Western blots ( a ) and cytoimmunostaining ( b ). Plasmid with vector-only was used as control (Con); ( c ). The representing Western blots reflected the subcellular compartmentalization of the corresponding proteins in the DN-VCP TG mouse heart tissues and compared to their age-matched WT mice at both young and older ages, including endogenous VCP (EN VCP), dominant-negative VCP (DN-VCP) and co-factors, ubiquitin fusion degradation protein 1 (UFD-1), nuclear protein localization protein 4 (NPL-4), and P47. GAPDH was used as a loading control of the cytoplasmic proteins, and Histon 3 (His 3) was used as a loading control of nuclear proteins; ( d ). The relative values of EN-VCP in cytoplasmic and nuclear fractions, respectively; (e - g ). The relative values of VCP-cofactors in cytoplasmic and nuclear fractions, respectively, including UFD-1 ( e ), NPL-4 ( f ), and P47 ( g ). The cytoplasmic proteins were normalized to the GAPDH, and the nuclear proteins were normalized to the Histon 3 (His 3). The relative value was presented by the fold of the WT young mice. *, p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 DN-VCP disrupts the VCP-cofactor interaction and leads to a binding preference of EN-VCP with the repressive protein P47. ( a ). Immonuoparticipation (IP) with anti-Flag and the Western blotting with the antibodies against the corresponding proteins in the cells overexpressed full-length VCP (FL-VCP) and dominant-negative VCP (DN-VCP) compared to the vector control. C: control plasmid with vector; FL: overexpressed full-length VCP, DN overexpressed DN-VCP; M: Molecular weight marker; ( b , c ). The representative images of the Western blots with the antibodies against the corresponding proteins after IP in cytoplasmic fractions of mouse heart tissues with anti-VCP ( b ) or anti-P47 ( c ). Input proteins were used as positive control and the supernatants of the IP were used as the negative control; ( d ). ATPase activity in DN-VCP TG and WT mouse heart tissues at young and older ages. **, p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
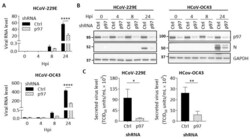
- Experimental details
- Figure 4 p97 knockdown suppresses HCoV replication in H1299 cells. H1299 cells with inducible control shRNA (Ctrl shRNA) or p97 shRNA were inoculated with Dox (0.5 mug/mL) for 72 h. Cells were infected with HCoV-229E (MOI 0.05) or HCoV-OC43 (MOI 0.01). After 1 h of infection, cells were washed and harvested at 0, 4, 8, or 24 hpi. Mock-infected cells were harvested at 24 hpi. ( A ) Quantification of viral RNA in cell lysates over time after infection. Data shows viral RNA levels relative to GAPDH. ( B ) Representative western blot images showing the expression of p97, viral N protein, and GAPDH (loading control) in cell lysates across the infection time course. Full-length blots can be found in Supplementary Figure S8 . ( C ) Viral titer in culture media collected from HCoV-infected cells at 24 hpi was quantified by determining TCID 50 in H1299 cells. Error bars represent SD ( n = 3). Statistical analysis was conducted with two-way ANOVA for Figure 4 A and Student's t test for Figure 4 C: *, p < 0.05; **, p < 0.01; ****, p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. Mitochondrial recruitment of VCP is not influenced by ectopic UBXN1 expression. (A) HeLa cells transfected with expression plasmid for FLAG-UBXN1 or control vector and mitoYFP-T2A-MYC-PRKN , treated with 25 uM CCCP for 90, 180, 360 min or left untreated, were fixed, stained using rabbit anti-FLAG and mouse anti-VCP antibodies and imaged by confocal microscopy. Shown are representative images from three independent experiments after 90 min of CCCP treatment. Scale bar: 20 um. (B) Mitochondrial translocation of VCP was quantified using line drawings across mitochondria. The plot presents the mean Pearson's correlation of 73 to 137 cells from three independent experiments. Statistical significance was tested using unbalanced one-way ANOVA.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
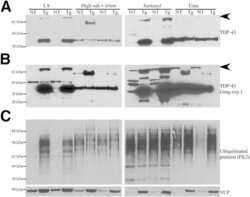
- 5 Fig Immunohistochemical findings in the striatonigral and pallidoluysian systems. p-TDP-43-immunoreactive NCIs and GCIs in the putamen (A, B), globus pallidus (C, D), subthalamic nucleus (E), and substantia nigra (F, G). (A, C, E, F) Right side. (B, D, G) Left side. NNIs in the putamen (H, I). p-TDP-43 (A-H) and VCP (I) immunostaining. Scale bars = 50 mum (A-G) and 5 mum (H, I).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure EV3 Parkin knockout mitigates the accumulation of mitophagy adaptors in G93A mitochondria at disease end stage A, B Representative Western blots of p62 (A), OPTN, and VCP (B) in spinal cord mitochondria from end-stage mice. C p62 quantification, using Complex V as normalizer, shows a strong accumulation of p62 in SOD1-G93A mitochondria at end stage. Results are expressed as mean +- SEM and as percent of Non Tg; n = 8 (four males and four females) mice per group. No statistically significant differences were found between G93A and PKO/G93A ( P = 0.078 by paired Wilcoxon's test); *** P = 0.0007 (Non Tg vs. G93A) and * P = 0.037 (PKO and PKO/G93A) both by paired Friedman's test with Dunn's correction. D Quantification of OPTN accumulation in end-stage mitochondria, with Complex V as protein loading control. Results are expressed as mean +- SEM and as percent of Non Tg; n = 8 (four males and four females) mice per group. ** P = 0.0078 (for G93A and PKO/G93A) by paired Wilcoxon's test; *** P = 0.0003 (Non Tg vs. G93A) by paired Friedman's test with Dunn's correction. No other statistically significant differences were found. E Quantification of VCP in mitochondria at disease end stage. Complex V was used as normalizer. Results are expressed as mean +- SEM and as percent of Non Tg; n = 8 (four males and four females) mice per group. * P = 0.039 (for G93A and PKO/G93A) by paired Wilcoxon's test; *** P = 0.0007 (Non Tg vs. G93A) and * P = 0.037 (PKO and PKO/G93A) both by pa
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
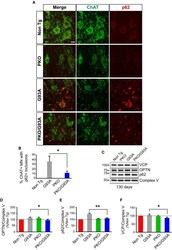
- Experimental details
- Figure 5 The content of mitochondrial p62, OPTN , and VCP in spinal cord of SOD 1-G93A mice is decreased by Parkin knockout Immunostaining of anterior horn of lumbar spinal cord for ChAT (in green) and p62 (in red) at 130 days. Scale bar, 10 mum. Quantification of the percentage of ChAT-positive motor neurons (MN) containing p62-positive inclusions at 130 days. PKO/G93A MN have less SOD1 aggregates than G93A, while Non Tg and PKO MN have none. Data were collected from n = 4 (two males and two females) mice for Non Tg and PKO and n = 6 (three males and three females) for G93A and PKO/G93A. Number of images recorded for each genotype: n = 9 for Non Tg and PKO, n = 16 for G93A, and n = 12 for PKO/G93A. Results are expressed as mean +- SEM; * P = 0.032, by unpaired one-tailed Student's t -test. Representative Western blots of OPTN, p62, and VCP in spinal cord mitochondrial fractions at 130 days. Protein levels are normalized by Complex V. The quantifications in panels (D-F) show that mitochondria of PKO/G93A mice have less mitophagy adaptor proteins than G93A relative to Complex V. Quantification of OPTN relative to Complex V at 130 days. Results are expressed as mean +- SEM and as percent of Non Tg; n = 8 (four males and four females); * P = 0.039 by paired Student's t -test. Quantification of p62, relative to Complex V at 130 days. Results are expressed as mean +- SEM and as percent of Non Tg; n = 8 (four males and four females); ** P = 0.0029 by paired Student's t -test. VC
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
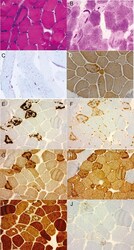
- Experimental details
- Figure 2 Vastus lateralis biopsy of P2 (A) Vastus lateralis muscle cryostat section hematoxylin and eosin (H&E) staining shows basophilic subsarcolemmal aggregate in one muscle fiber and one fiber with a small rimmed vacuole. H&E staining x100. (B) Periodic acid-Schiff (PAS) staining x100 reveals some otherwise pale fibers with prominent accumulates of PAS-positive polyglucosan material, evident also in (C) PAS semithin sections x200. (D) Immunohistochemical staining for phosphosphofructokinase shows no reduction in sarcolemmal or cytoplasmic expression. However, abnormal cytoplasmic phosphofructo-1-kinase-positive aggregates are observed in some fibers. These accumulations are unlike normal glycogen positive for (E) p62, (F) ubiquitin, (G) valosin-containing protein, and (H) desmin immunohistochemistry and negative for (I) myotilin, findings consistent with polyglucosan aggregates. (J) In these fiber regions, rimmed vacuolar pathology with accumulation of light chain 3b-positive material is also seen (original magnification x100 in panels E-J).
 Explore
Explore Validate
Validate Learn
Learn