Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [1]
- Immunohistochemistry [2]
- Flow cytometry [1]
- Other assay [20]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 13-8000 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- alpha Tubulin Monoclonal Antibody (TU-01)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- TU-01
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Alpha Tubulin was performed by loading 30 µg of HeLa (lane1), Jurkat (lane2), HepG2 (lane3), A549 (lane4) and Daudi (lane5) lysate using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX), XCell SureLock™ Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (Product # LC5800), and iBlot® Dry Blotting System (Product # IB21001). Proteins were transferred to a nitrocellulose membrane and blocked with 5 % skim milk for 1 hour at room temperature. Alpha Tubulin was detected at 52 kDa using Alpha Tubulin Mouse Monoclonal Antibody (Product # 13-8000) at 1:250 dilution in 5 % skim milk at 4°C overnight on a rocking platform. Goat Anti-Mouse IgG - HRP Secondary Antibody (Product # 62-6520) at 1:4000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Alpha-Tubulin was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Alpha-Tubulin Mouse monoclonal Antibody (Product # 13-8000) at 1:250 dilution in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Rabbit Anti-Mouse IgG Secondary Antibody (Product # A-11059) at a dilution of 1:400 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Alpha-Tubulin showing staining in the cytoplasm of paraffin-embedded human breast carcinoma tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Alpha-Tubulin monoclonal antibody (Product # 13-8000) diluted in 3% BSA-PBS at a dilution of 1:50 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Alpha-Tubulin showing staining in the cytoplasm of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Alpha-Tubulin monoclonal antibody (Product # 13-8000) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Flow cytometry analysis of Alpha-Tubulin was done on A549 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with Alpha-Tubulin Mouse Monoclonal Antibody (138000, red histogram) or with mouse isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
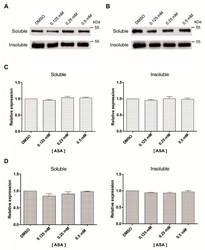
- Experimental details
- Figure 3 Soluble and insoluble tubulin fractions of Giardia trophozoites exposed to ASA. The amount of soluble and insoluble alpha-tubulin was analyzed by Western blotting after 24 h ( A ) and 48 h ( B ). This experiment was repeated three times. Densitometric analysis of soluble and insoluble tubulin levels after 24 h ( C ) and 48 h ( D ).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Proteomics analysis shows that the nuclear abundance of the proteasome is reduced in association with oncogenic transformation. ( a ) Strategy for cell transformation and subcellular fractionation. TIG-3(WT) cells were transformed by the introduction of hTert and the early region of SV40. Cellular proteins of both TIG-3(WT) cells and the transformed cells, designated TIG-3(T + SV40), were separated into three fractions corresponding to the cytoplasm (S), the nucleoplasm and proteins loosely associated with chromatin (P1), and proteins tightly associated with chromatin (P2). Each fraction was analyzed by liquid chromatography and tandem mass spectrometry (LC-MS/MS). ( b ) Validation of subcellular fractionation. Whole cell extract (WCE) and subcellular fractions of TIG-3(WT) cells were subjected to immunoblot (IB) analysis with antibodies to LDHA, HSP90, alpha-tubulin, and calnexin as cytoplasmic marker proteins; to c-Jun and E2F1 as nucleoplasmic marker proteins; to histone H1 as a chromatin marker protein; and to the proteasome subunits PSMA2 and PSMD1. ( c ) Label-free proteomics analysis of P1 and P2 fractions. The log 2 [fold change] for protein abundance in TIG-3(T + SV40) cells relative to TIG-3(WT) cells and the -log 10 [q-value] are shown as Volcano plots. The threshold for determining differential expression is indicated by the dashed lines (q-value of
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Immunofluorescence and immunoblot analyses confirming that the nuclear localization of the proteasome is suppressed by oncogenic transformation. ( a ) Immunofluorescence staining of alpha-proteasomal subunits (red) in TIG-3(WT), TIG-3(T), and TIG-3(T + SV40) cells fixed after cytoplasm removal. DNA (blue) was counterstained with DAPI (upper panel). Scale bars, 10 um. Histograms show the frequency distribution for normalized immunofluorescence signal intensity (A.U., arbitrary units) in the nucleus, with the median value for TIG-3(WT) cells being indicated with a dashed red line (lower panel). ( b ) Box plot of the percentage of cells with proteasome fluorescence intensity greater than the median of the distribution for TIG-3(WT) cells determined as in ( a ). Data are for four independent biological replicates. * P < 0.05 (one-way ANOVA followed by Bonferroni''s post hoc test). ( c ) Immunoblot analysis of PSMA2 and PSMD1 in the S and P1 fractions of TIG-3(WT), TIG-3(T), and TIG-3(T + SV40) cells. HSP90 and alpha-tubulin were examined as markers for the S fraction, and c-Jun as a marker for the P1 fraction. ( d ) Quantitative analysis of band intensities for PSMA2 and PSMD1 in experiments similar to that in ( c ). Data are from six independent biological replicates. ** P < 0.01 (one-way ANOVA followed by Bonferroni''s post hoc test). Boxes and whiskers show the median, the lower and upper quartiles, and the minimum and maximum values. R software version 3.6.2 ( https:
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 SV40 LT, but not ST, alters the nuclear abundance of the proteasome. ( a ) Immunoblot analysis of SV40 early region expression in whole cell extracts of TIG-3(T), TIG-3(T + SV40), TIG-3(T + SV40LT), and TIG-3(T + SV40ST) cells. The antibodies detect the NH 2 -terminal region of both LT and ST. ( b ) Immunofluorescence staining of alpha-proteasomal subunits (red) in cells as in ( a ). The cells were fixed after cytoplasm removal and before staining. DNA (blue) was counterstained with DAPI (upper panel). Scale bars, 20 um. Histograms show the frequency distribution for normalized immunofluorescence signal intensity in the nucleus, with the median value for TIG-3(T) cells being indicated with the dashed red line (lower panel). ( c ) Box plot for the percentage of cells with a proteasome fluorescence intensity greater than the median of the distribution for TIG-3(T) cells determined as in ( b ). Data are for three independent biological replicates. * P < 0.05 (one-way ANOVA followed by Bonferroni''s post hoc test). ( d ) Immunoblot analysis of PSMA2 and PSMD1 in the S and P1 fractions of TIG-3(T), TIG-3(T + SV40), TIG-3(T + SV40LT), and TIG-3(T + SV40ST) cells. HSP90 and alpha-tubulin were examined as controls for the S fraction, and c-Jun as a control for the P1 fraction. ( e ) Quantitative analysis of band intensities for PSMA2 and PSMD1 in experiments similar to that in ( d ). Data are for five independent biological replicates. ** P < 0.01 (one-way ANOVA followed by
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
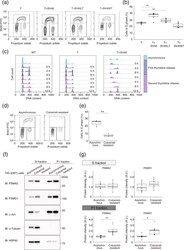
- Experimental details
- Figure 5 Nuclear proteasome abundance is increased in response to cell cycle arrest at G 0 -G 1 . ( a ) Cell cycle analysis in TIG-3(T), TIG-3(T + SV40), TIG-3(T + SV40LT), and TIG-3(T + SV40ST) cells. Cells were labeled with bromodeoxyuridine (BrdU) for 90 min, fixed, and stained with fluorescein isothiocyanate (FITC)-conjugated antibodies to BrdU. Nuclear DNA was stained with propidium iodide, and the cells were then analyzed by flow cytometry. ( b ) Box plot of the proportion of proliferative cells (cells in S phase) in experiments similar to that in ( a ). Data are for six independent biological replicates. ** P < 0.01 (one-way ANOVA followed by Bonferroni''s post hoc test). ( c ) Analysis of cell cycle progression by double thymidine block. TIG-3(WT), TIG-3(T), and TIG-3(T + SV40) cells were arrested at the G 1 -S transition by exposure to excess thymidine and then released for the indicated times. They were then stained with propidium iodide for flow cytometric analysis of DNA content. Data from the asynchronous cells are also shown at the top. ( d ) Cell cycle analysis by the colcemid-challenge test. Asynchronous TIG-3(WT) cells and TIG-3(WT) cells remaining (colcemid-resistant cells) after long-term treatment with colcemid and shake-off of those arrested at G 2 -M phase were analyzed for cell cycle profile as in ( a ). ( e ) Box plot of the proportion of BrdU-positive cells in experiments similar to that in ( d ). Data are for three independent biological replicates.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
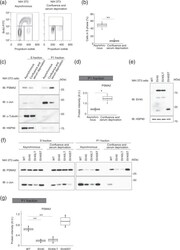
- Experimental details
- Figure 8 Nuclear proteasome abundance is regulated in a cell cycle-dependent manner in NIH 3T3 cells. ( a ) Cell cycle analysis of NIH 3T3 cells in asynchronous culture or after induction of quiescence by confluent culture and serum deprivation. Cells were labeled with BrdU for 30 min, fixed, and stained with FITC-conjugated antibodies to BrdU. Nuclear DNA was stained with propidium iodide, and the cells were then analyzed by flow cytometry. ( b ) Box plot of the proportion of proliferative cells (cells in S phase) for experiments similar to that in ( a ). Data are for three independent biological replicates. ** P < 0.01 (Student''s t test). ( c ) Immunoblot analysis of PSMA2 in the S and P1 fractions of NIH 3T3 cells maintained as in ( a ). HSP90 a nd alpha-tubulin were examined as controls for the S fraction, and c-Jun as a control for the P1 fraction. ( d ) Quantitative analysis of PSMA2 in the P1 fraction for experiments similar to that in ( c ). Data are for three independent biological replicates. * P < 0.05 (Student''s t test). ( e ) Immunoblo t analysis of SV40 early region expression in NIH 3T3(WT), NIH 3T3(SV40), NIH 3T3(SV40LT), and NIH 3T3(SV40ST) cells. ( f ) Immunoblot analysis of PSMA2 in the S and P1 fractions of NIH 3T3(WT), NIH 3T3(SV40), NIH 3T3(SV40LT), and NIH 3T3(SV40ST) cells maintained as in ( a ). ( g ) Quantitative a nalysis of band intensities for PSMA2 in the P1 fraction of cells after induction of quiescence by confluent culture and serum deprivat
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
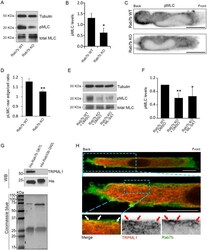
- Experimental details
- Fig. 6. Rab7b regulates myosin II activation and interacts with the lysosomal Ca 2+ channel TRPML1. (A) Lysates from BMDCs from WT and Rab7b KO mice pulsed with 100 ng/ml LPS for 30 min were subjected to immunoblotting analysis with the indicated antibodies. Tubulin was used as loading control. (B) The graph shows the quantification of phosphorylated myosin light chain (pMLC) levels in WT and Rab7b KO DCs normalized to the tubulin levels. Data represents the mean+-s.d. of four independent experiments. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. MENE(2R)-E B6 is a mutant of Drosophila Snap29 . ( A ) Schematic view of the Snap29 locus. Df(2R)egl2 (black) complements the B6-21 mutation, while Df(2R)3-659 and Df(2R)106 (red) fail to complement it, indicating that B6-21 maps to the genetic interval 60A3-A5 on the right arm of the Drosophila chromosome 2. The coding sequence of Snap29 is shown in orange, while the domains of Snap29 are indicated in yellow and blue. A black triangle marks the approximate position of the B6 mutation. ( B ) Sequencing of the B6 allele in heterozygosity with the parental chromosome on which the mutation was induced. A C-to-T change creates a premature stop codon that truncates the protein right after the first SNARE domain. ( C ) Expression of Snap29 mRNA is only 25% reduced in mutant eye-antennal and wing discs, relative to WT. ( D ) Analysis of Snap29 expression by protein gel blot in WT disc extracts and in extracts of discs containing Snap29 B6 mutant cells indicates that Snap29 B6 , a truncated form of Snap29, is present in mutant cells. ( E ) Ubiquitous expression of CFP-Snap29 under tubulin-Gal4 (tub>) rescues lethality of homozygous Snap29 flies. Rescued flies (right) are indistinguishable from heterozygous animals (left). ( F ) Adult eyes of flies with the indicated genetic background. Eye-specific ectopic expression of CFP-Snap29, or of a Snap29 form with a mutated NPF motif (Snap29 AAA ) rescue defects of Snap29 B6 mutant eye discs and yield adults wit
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. Snap29 mutant cells fail to complete autophagy. ( A to C ) Clones of Snap29 B6 ( A and B ) or Vps25 A3 ( C ) mutant cells in mosaic eye-antennal discs accumulate high levels of ref(2)p and ubiquitin, compared to surrounding WT cells. ( B and C ) show a high magnification image of an anterior portion of an eye discs. Single ref(2)P and Ubiquitin channels are shown. ( D to F ) Immunoblots of protein extracts from eye-antennal discs of the indicated genotypes to detect ref(2)P ( D ), ubiquitin ( E ) and pS6k ( F ). Compared to protein extracts of WT discs, discs mutant for Snap29 and for the autophagy and trafficking regulator Vps25 accumulate ref(2)P, ubiquitin and pS6k. Loading controls are shown below each blot. ( G ) Relative expression of Atg8a or Atg18b by Q-PCR analysis of mRNA extracts from WT and mutant discs. Mutant discs do not show induction of expression of Atg genes. ( H and I ) A single medial confocal cross-section of the Drosophila FE of a stage 9 egg chamber. FE cells overexpressing GFP-LAMP1 are stained for Snap29 and Atg8a. H ' to H' show respectively the LAMP1 and Snap29, the LAMP1 and Atg8a, and the Snap29 and Atg8a merged channels. A high magnification of a typical cluster formed by GFP-LAMP1, Snap29 and Atg8a-positive vesicles is shown in ( I ). The 3 proteins are in close proximity.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7. Snap29 mutant cells display altered N trafficking. ( A and B ) Clones of Snap29 B6 mutant cells in mosaic eye-antennal discs accumulate high levels of N, compared to surrounding WT cells. A ' and B'' are single channels. ( C and D ) Clones of Snap29 B6 mutant cells in the anterior portion of a mosaic eye discs stained as indicated. N and GM130 do not colocalize in both WT and mutant cells, while a fraction of N colocalizes with Syx7 in both WT and mutant cells, excluding accumulation in these compartments. ( C ' and D'' ) are single channels. ( E ) Labeling of nonpermeabilized Snap29 B6 mosaic eye-antennal discs with an anti-N NECD. Compared to WT cells marked by expression of GFP, clones of mutant cells in the eye disc display higher N surface levels (Surf. N). E' shows the single confocal channel for anti-N. ( F and G ) Z-sections of eye disc epithelia subjected to 15' ( F ) and 210' ( G ) internalization of anti-NECD and staining as indicated. Similar to WT cells, N is present on the apical plasma membrane of mutant cells ( F , arrow) and is able to access early endosomes ( F , arrowhead). N is efficiently internalized over time in both WT and mutant cells ( G , arrow); however, in mutant cells it fails to be degraded and accumulates in a Syx7-negative compartment ( G , arrowhead). ( F ' -G ') show the single confocal channel for anti-N. ( H ) 210' min internalization of anti-NECD. Compared to WT cells, mutant cells display intracellular N accumulations. The acti
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA