Antibody data
- Antibody Data
- Antigen structure
- References [3]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [4]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-42049 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FAH Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- Peptide sequence: SFIPVAEDSD FPIHNLPYGV FSTRGDPRPR IGVAIGDQIL DLSIIKHLFT Sequence homology: Cow: 93%; Dog: 86%; Guinea Pig: 86%; Horse: 86%; Human: 100%; Mouse: 93%; Pig: 93%; Rat: 93%
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Concentration
- 0.5 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references A versatile transposon-based technology to generate loss- and gain-of-function phenotypes in the mouse liver.
Genetically blocking HPD via CRISPR-Cas9 protects against lethal liver injury in a pig model of tyrosinemia type I.
Identification and evaluation of fructose-bisphosphate aldolase B as a potential diagnostic biomarker in choledochal cysts patients: a quantitative proteomic analysis.
Kopasz AG, Pusztai DZ, Karkas R, Hudoba L, Abdullah KSA, Imre G, Pankotai-Bodó G, Migh E, Nagy A, Kriston A, Germán P, Drubi AB, Molnár A, Fekete I, Dani VÉ, Ocsovszki I, Puskás LG, Horváth P, Sükösd F, Mátés L
BMC biology 2022 Apr 1;20(1):74
BMC biology 2022 Apr 1;20(1):74
Genetically blocking HPD via CRISPR-Cas9 protects against lethal liver injury in a pig model of tyrosinemia type I.
Gu P, Yang Q, Chen B, Bie YN, Liu W, Tian Y, Luo H, Xu T, Liang C, Ye X, Liu Y, Tang X, Gu W
Molecular therapy. Methods & clinical development 2021 Jun 11;21:530-547
Molecular therapy. Methods & clinical development 2021 Jun 11;21:530-547
Identification and evaluation of fructose-bisphosphate aldolase B as a potential diagnostic biomarker in choledochal cysts patients: a quantitative proteomic analysis.
Ming G, Guo W, Cheng Y, Wang J
Translational pediatrics 2021 Aug;10(8):2083-2094
Translational pediatrics 2021 Aug;10(8):2083-2094
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
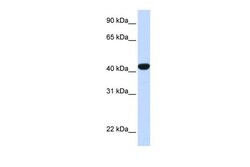
- Experimental details
- Western blot analysis of human liver cells using an anti-Fumarylacetoacetase polyclonal antibody (Product # PA5-42049).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
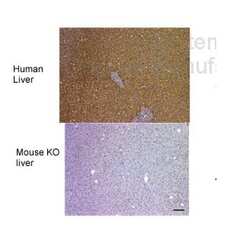
- Experimental details
- Immunohistochemistry analysis of human liver and mouse FAH KO liver tissue using an anti-Fumarylacetoacetase polyclonal antibody (Product # PA5-42049).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
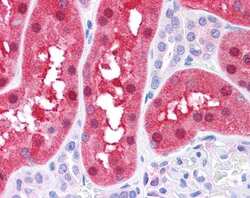
- Experimental details
- Immunohistochemistry analysis of human liver (top) and mouse KO liver tissue (bottom) using an anti-Fumarylacetoacetase polyclonal antibody (Product # PA5-42049).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
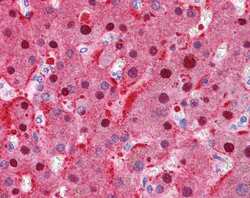
- Experimental details
- Immunohistochemistry analysis of human liver, mouse KO tissue using an anti-Fumarylacetoacetase polyclonal antibody (Product # PA5-42049).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
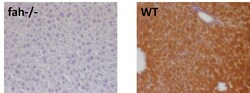
- Experimental details
- Immunohistochemistry analysis of FAH was performed on fah-/- (left) and wildtype (right) mouse liver paraffin sections. To expose target proteins, antigen retrieval was performed by microwaving tissues for 8-15 minutes in 10mM sodium citrate buffer (pH 6.0). Following antigen retrieval, endogenous peroxidases were blocked with 3% hydrogen peroxide-methanol for 15 min at room temperature. Tissue slides were washed with deionized water and PBS, and then blocked in 3% BSA-PBS for 30 min at room temperature. Tissues were probed with a FAH polyclonal antibody (Product # PA5-42049) diluted 1:400 in 3% BSA-PBS overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting. Images were taken at 20X magnification. Data courtesy of Antibody Data Exchange Program.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
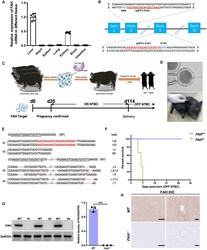
- Experimental details
- Figure 1 The generation of FAH -/- pigs via direct zygote injection (A) FAH mRNA levels were determined in the liver, heart, spleen, lung, kidney, brain, and intestine of healthy pigs (n = 6). (B) Schematic diagram of the target sequence at the FAH locus. The sgRNA sequences are underlined and highlight in red and the protospacer adjacent motif (PAM) sequences are highlighted in blue. (C) Schematic overview of the single-step generation of FAH mutant pigs. (D) Top panel: representative photograph of microinjection into the cytoplasm of one-cell-stage zygotes; bottom panel, representative photograph of newborn FAH - / - piglets. (E) Sanger sequencing of mutant sequences from four newborn piglets. The targeted sequences are underlined and shown at the top. The mutations are shown in red. Deletions and insertions are denoted as ""Delta"" and ""+"" plus the number of base pairs, respectively. (F) Kaplan-Meier survival curve for FAH - / - and FAH + / - pigs. (G) Western blotting and quantification of FAH and GAPDH protein levels in liver tissue of FAH - / - founders (#1, #3, #4). Data are presented as means +- SD. ***p < 0.001. (H) Immunohistochemical staining for FAH in liver sections derived from FAH - / - pigs showing the absence of FAH expression. Scale bar, 200 mum (left) and 50 mum (right).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
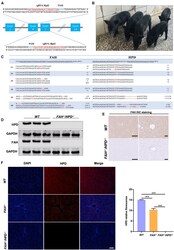
- Experimental details
- Figure 2 The generation of FAH -/- / HPD -/- double-knockout pigs (A) Schematic diagram of the target sequence at the HPD locus. The sgRNA sequences are underlined and highlighted in red and the PAM sequences are highlighted in blue. (B) Representative photographs of FAH - / - / HPD - / - founder piglets (2 months old). (C) Sanger sequencing results of the target regions at the FAH and HPD loci in FAH - / - / HPD - / - founders. The targeted sequences ( FAH and HPD ) are underlined and shown at the top. The mutations are shown in red. Deletions and insertions are denoted as ""Delta"" and ""+"" plus the number of base pairs, respectively. (D-F) FAH and HPD protein expression as detected by western blotting (D), immunohistochemistry (scale bar, 200 mum [left panel] and 50 mum [right panel]; E) and immunofluorescence (scale bar, 50 mum; F). The results show that the FAH - / - / HPD - / - founders were negative for FAH and HPD protein expression in liver tissue, whereas FAH - / - pigs expressed a reduced amount of HPD compared with that in wild-type (WT) pigs. Data are presented as means +- SD. ***p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
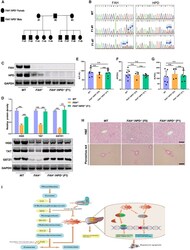
- Experimental details
- Figure 7 Characterization of the F1 double-mutant generation (A) Pedigree showing the intercrossing of the F0 FAH - / - / HPD - / - pigs to produce the F1 generation. Filled squares indicate FAH - / - / HPD - / - males, filled circles indicate FAH - / - / HPD - / - females. (B) Sanger sequencing chromatograms for sperm DNA from WT, F1- #1, and F1- #7 pigs. The blue arrow indicates the overlapping peaks in target sites. (C) Western blotting analysis of the FAH and HPD protein expression in liver tissues of WT, FAH - / - , and FAH - / - / HPD - / - F1 pigs. (D) Representative images and semiquantitative analysis of the protein expression levels of TAT, HGD, and GSTZ1 in the liver tissues of each group as detected by western blotting. (E-G) Serum ALT, AST, and ALP levels were determined in WT, FAH - / - / HPD - / - founders (F0), and FAH - / - / HPD - / - F1 generation pigs. (H) H&E and picrosirius red staining showing normal liver tissue architecture, without obvious hepatic fibrosis, of the FAH - / - / HPD - / - founders (F0) and F1 generations. Scale bar, 100 mum. (I) A schematic diagram depicting the mechanism involved in genetically blocking HPD to protect against liver injury in HT1. Data are reported as means +- SD; n.s., not significant; ***p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
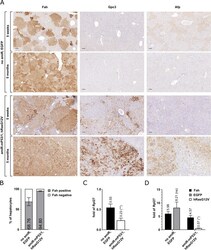
- Experimental details
- Induction of HCC using a predefined combination of drivers. a Immunohistochemical analysis of the Fah selection marker, Gpc3, and Afp HCC markers in liver sections from Fah -/- mice treated with either control (no amiR, EGFP) or driver (amiR-mP53/1, hRas G12V ) transposon constructs at 5 weeks and 5 months post-treatment. For the analysis of tumors emerging 5 months after treatment with the driver construct, a vector mixture containing 1% driver transposon vector and 99% transposon vector expressing only the Fah selection marker protein was used. Scale bars, 100 mum. b Determination of the percentage of Fah-positive hepatocytes 5 weeks after treatment by machine learning-based measurement. Data were presented as the mean +- SD ( n = 3) (see Additional file 2 for individual data values and statistics). c Monitoring of endogenous p53 mRNA levels in the liver of Fah -/- mice treated with driver and control transposon constructs. Liver RNA samples were collected from Fah -/- mice at 5 weeks post-treatment and tested using a p53 mRNA-specific RT-qPCR assay. Results were normalized to measurements of the ribosomal protein L27 ( Rpl27 ) transcript as input control and data were presented as the mean +- SD ( n = 3) (see Additional file 2 for individual data values and statistics). d Monitoring the amount of transcripts A and B in the liver of Fah -/- mice treated with driver and control transposon constructs. Liver RNA samples were collected from Fah -/- mice at 5 weeks post-treatmen
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot