Antibody data
- Antibody Data
- Antigen structure
- References [48]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [40]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-0739-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD73 Monoclonal Antibody (AD2), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This AD2 monoclonal antibody reacts with human CD73, a 5'-ectonucleotidase that converts 5'-adenosine monophosphate to adenosine. CD73 is expressed on the surface of endothelial cells, as well as B and T cells, including some CD4+Foxp3+ regulatory T cells. Adenosine production by these cells has been linked to the inhibition of CD4 T cell effector functions such as proliferation and cytokine secretion. Applications Reported: This AD2 antibody has been reported for use in flow cytometric analysis. Applications Tested: This AD2 antibody has been pre-titrated and tested by flow cytometric analysis on normal human peripheral blood cells. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- AD2
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Single-cell RNA landscape of the osteoimmunology microenvironment in periodontitis.
Exosomal CD73 from serum of patients with melanoma suppresses lymphocyte functions and is associated with therapy resistance to anti-PD-1 agents.
Mechanical Compression by Simulating Orthodontic Tooth Movement in an In Vitro Model Modulates Phosphorylation of AKT and MAPKs via TLR4 in Human Periodontal Ligament Cells.
Induced Neurodifferentiation of hBM-MSCs through Activation of the ERK/CREB Pathway via Pulsed Electromagnetic Fields and Physical Stimulation Promotes Neurogenesis in Cerebral Ischemic Models.
Mesenchymal stem cells transfer mitochondria to allogeneic Tregs in an HLA-dependent manner improving their immunosuppressive activity.
Reprogramming bone progenitor identity and potency through control of collagen density and oxygen tension.
In vitro differentiation of human embryonic stem cells to hemogenic endothelium and blood progenitors via embryoid body formation.
Bcl-xL mutant promotes cartilage differentiation of BMSCs by upregulating TGF-β/BMP expression levels.
Therapeutic potential of human umbilical cord mesenchymal stem cells on aortic atherosclerotic plaque in a high-fat diet rabbit model.
Matrix reverses immortalization-mediated stem cell fate determination.
In Vitro Anti-cancer Activity of Adipose-Derived Mesenchymal Stem Cells Increased after Infection with Oncolytic Reovirus.
VAP-PLGA microspheres (VAP-PLGA) promote adipose-derived stem cells (ADSCs)-induced wound healing in chronic skin ulcers in mice via PI3K/Akt/HIF-1α pathway.
Peptide 11R‑VIVIT promotes fracture healing in osteoporotic rats.
TRPA1 triggers hyperalgesia and inflammation after tooth bleaching.
Down-Regulated Exosomal MicroRNA-221 - 3p Derived From Senescent Mesenchymal Stem Cells Impairs Heart Repair.
Differentiation Potential of Early- and Late-Passage Adipose-Derived Mesenchymal Stem Cells Cultured under Hypoxia and Normoxia.
Vasculogenesis from Human Dental Pulp Stem Cells Grown in Matrigel with Fully Defined Serum-Free Culture Media.
Chondrogenic Differentiation from Induced Pluripotent Stem Cells Using Non-Viral Minicircle Vectors.
Prospective isolation of human fibroadipogenic progenitors with CD73.
Circulating Exosomes Inhibit B Cell Proliferation and Activity.
IGF-1 enhances BMSC viability, migration, and anti-apoptosis in myocardial infarction via secreted frizzled-related protein 2 pathway.
PF-127 hydrogel plus sodium ascorbyl phosphate improves Wharton's jelly mesenchymal stem cell-mediated skin wound healing in mice.
Impact of Fibronectin Knockout on Proliferation and Differentiation of Human Infrapatellar Fat Pad-Derived Stem Cells.
Therapeutic Effects of Human Urine-Derived Stem Cells in a Rat Model of Cisplatin-Induced Acute Kidney Injury In Vivo and In Vitro.
Sodium Selenite Improves The Therapeutic Effect Of BMSCs Via Promoting The Proliferation And Differentiation, Thereby Promoting The Hematopoietic Factors.
Isolation and Culture of Human Stem Cells from Apical Papilla under Low Oxygen Concentration Highlight Original Properties.
High-yield isolation of menstrual blood-derived endometrial stem cells by direct red blood cell lysis treatment.
Modulation of proliferation and differentiation of gingiva‑derived mesenchymal stem cells by concentrated growth factors: Potential implications in tissue engineering for dental regeneration and repair.
The influence of chemotherapy on adenosine-producing B cells in patients with head and neck squamous cell carcinoma.
Human umbilical cord-derived mesenchymal stem cells ameliorate the enteropathy of food allergies in mice.
BMP14 induces tenogenic differentiation of bone marrow mesenchymal stem cells in vitro.
A high-yield isolation and enrichment strategy for human lung microvascular endothelial cells.
Human umbilical cord mesenchymal stem cells improve the reserve function of perimenopausal ovary via a paracrine mechanism.
Low-dose interleukin-2 promotes STAT-5 phosphorylation, T(reg) survival and CTLA-4-dependent function in autoimmune liver diseases.
A Member of the Nuclear Receptor Superfamily, Designated as NR2F2, Supports the Self-Renewal Capacity and Pluripotency of Human Bone Marrow-Derived Mesenchymal Stem Cells.
Spontaneous Differentiation of Human Mesenchymal Stem Cells on Poly-Lactic-Co-Glycolic Acid Nano-Fiber Scaffold.
CRISPR/Cas9-based genetic correction for recessive dystrophic epidermolysis bullosa.
Transendothelial migration of human umbilical mesenchymal stem cells across uterine endothelial monolayers: Junctional dynamics and putative mechanisms.
NR2F2 regulates bone marrow-derived mesenchymal stem cell-promoted proliferation of Reh cells.
MIF Plays a Key Role in Regulating Tissue-Specific Chondro-Osteogenic Differentiation Fate of Human Cartilage Endplate Stem Cells under Hypoxia.
Human Gingival Integration-Free iPSCs; a Source for MSC-Like Cells.
FOXF1 mediates mesenchymal stem cell fusion-induced reprogramming of lung cancer cells.
Epithelial cell differentiation of human mesenchymal stromal cells in decellularized lung scaffolds.
Hypoxia-cultured human adipose-derived mesenchymal stem cells are non-oncogenic and have enhanced viability, motility, and tropism to brain cancer.
Myoblasts derived from normal hESCs and dystrophic hiPSCs efficiently fuse with existing muscle fibers following transplantation.
Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression.
T regulatory and primed uncommitted CD4 T cells express CD73, which suppresses effector CD4 T cells by converting 5'-adenosine monophosphate to adenosine.
Characterization of an IgM Fc-binding receptor on human T cells.
Chen Y, Wang H, Yang Q, Zhao W, Chen Y, Ni Q, Li W, Shi J, Zhang W, Li L, Xu Y, Zhang H, Miao D, Xing L, Sun W
Theranostics 2022;12(3):1074-1096
Theranostics 2022;12(3):1074-1096
Exosomal CD73 from serum of patients with melanoma suppresses lymphocyte functions and is associated with therapy resistance to anti-PD-1 agents.
Turiello R, Capone M, Morretta E, Monti MC, Madonna G, Azzaro R, Del Gaudio P, Simeone E, Sorrentino A, Ascierto PA, Morello S
Journal for immunotherapy of cancer 2022 Mar;10(3)
Journal for immunotherapy of cancer 2022 Mar;10(3)
Mechanical Compression by Simulating Orthodontic Tooth Movement in an In Vitro Model Modulates Phosphorylation of AKT and MAPKs via TLR4 in Human Periodontal Ligament Cells.
Roth CE, Craveiro RB, Niederau C, Malyaran H, Neuss S, Jankowski J, Wolf M
International journal of molecular sciences 2022 Jul 22;23(15)
International journal of molecular sciences 2022 Jul 22;23(15)
Induced Neurodifferentiation of hBM-MSCs through Activation of the ERK/CREB Pathway via Pulsed Electromagnetic Fields and Physical Stimulation Promotes Neurogenesis in Cerebral Ischemic Models.
Park HJ, Choi JH, Nam MH, Seo YK
International journal of molecular sciences 2022 Jan 21;23(3)
International journal of molecular sciences 2022 Jan 21;23(3)
Mesenchymal stem cells transfer mitochondria to allogeneic Tregs in an HLA-dependent manner improving their immunosuppressive activity.
Piekarska K, Urban-Wójciuk Z, Kurkowiak M, Pelikant-Małecka I, Schumacher A, Sakowska J, Spodnik JH, Arcimowicz Ł, Zielińska H, Tymoniuk B, Renkielska A, Siebert J, Słomińska E, Trzonkowski P, Hupp T, Marek-Trzonkowska NM
Nature communications 2022 Feb 14;13(1):856
Nature communications 2022 Feb 14;13(1):856
Reprogramming bone progenitor identity and potency through control of collagen density and oxygen tension.
Al Hosni R, Bozec L, Roberts SJ, Cheema U
iScience 2022 Apr 15;25(4):104059
iScience 2022 Apr 15;25(4):104059
In vitro differentiation of human embryonic stem cells to hemogenic endothelium and blood progenitors via embryoid body formation.
Garcia-Alegria E, Potts B, Menegatti S, Kouskoff V
STAR protocols 2021 Mar 19;2(1):100367
STAR protocols 2021 Mar 19;2(1):100367
Bcl-xL mutant promotes cartilage differentiation of BMSCs by upregulating TGF-β/BMP expression levels.
Xiao K, Yang L, Xie W, Gao X, Huang R, Xie M
Experimental and therapeutic medicine 2021 Jul;22(1):736
Experimental and therapeutic medicine 2021 Jul;22(1):736
Therapeutic potential of human umbilical cord mesenchymal stem cells on aortic atherosclerotic plaque in a high-fat diet rabbit model.
Li Y, Shi G, Han Y, Shang H, Li H, Liang W, Zhao W, Bai L, Qin C
Stem cell research & therapy 2021 Jul 15;12(1):407
Stem cell research & therapy 2021 Jul 15;12(1):407
Matrix reverses immortalization-mediated stem cell fate determination.
Wang Y, Hu G, Hill RC, Dzieciatkowska M, Hansen KC, Zhang XB, Yan Z, Pei M
Biomaterials 2021 Jan;265:120387
Biomaterials 2021 Jan;265:120387
In Vitro Anti-cancer Activity of Adipose-Derived Mesenchymal Stem Cells Increased after Infection with Oncolytic Reovirus.
Babaei A, Bannazadeh Baghi H, Nezhadi A, Jamalpoor Z
Advanced pharmaceutical bulletin 2021 Feb;11(2):361-370
Advanced pharmaceutical bulletin 2021 Feb;11(2):361-370
VAP-PLGA microspheres (VAP-PLGA) promote adipose-derived stem cells (ADSCs)-induced wound healing in chronic skin ulcers in mice via PI3K/Akt/HIF-1α pathway.
Jiang W, Zhang J, Zhang X, Fan C, Huang J
Bioengineered 2021 Dec;12(2):10264-10284
Bioengineered 2021 Dec;12(2):10264-10284
Peptide 11R‑VIVIT promotes fracture healing in osteoporotic rats.
Hou C, Wang X, Jiang W, Bian Z, Zhu L, Li M
International journal of molecular medicine 2021 Aug;48(2)
International journal of molecular medicine 2021 Aug;48(2)
TRPA1 triggers hyperalgesia and inflammation after tooth bleaching.
Chen C, Huang X, Zhu W, Ding C, Huang P, Li R
Scientific reports 2021 Aug 31;11(1):17418
Scientific reports 2021 Aug 31;11(1):17418
Down-Regulated Exosomal MicroRNA-221 - 3p Derived From Senescent Mesenchymal Stem Cells Impairs Heart Repair.
Sun L, Zhu W, Zhao P, Zhang J, Lu Y, Zhu Y, Zhao W, Liu Y, Chen Q, Zhang F
Frontiers in cell and developmental biology 2020;8:263
Frontiers in cell and developmental biology 2020;8:263
Differentiation Potential of Early- and Late-Passage Adipose-Derived Mesenchymal Stem Cells Cultured under Hypoxia and Normoxia.
Zhao AG, Shah K, Freitag J, Cromer B, Sumer H
Stem cells international 2020;2020:8898221
Stem cells international 2020;2020:8898221
Vasculogenesis from Human Dental Pulp Stem Cells Grown in Matrigel with Fully Defined Serum-Free Culture Media.
Luzuriaga J, Irurzun J, Irastorza I, Unda F, Ibarretxe G, Pineda JR
Biomedicines 2020 Nov 9;8(11)
Biomedicines 2020 Nov 9;8(11)
Chondrogenic Differentiation from Induced Pluripotent Stem Cells Using Non-Viral Minicircle Vectors.
Rim YA, Nam Y, Park N, Jung H, Lee K, Lee J, Ju JH
Cells 2020 Mar 1;9(3)
Cells 2020 Mar 1;9(3)
Prospective isolation of human fibroadipogenic progenitors with CD73.
Goloviznina NA, Xie N, Dandapat A, Iaizzo PA, Kyba M
Heliyon 2020 Jul;6(7):e04503
Heliyon 2020 Jul;6(7):e04503
Circulating Exosomes Inhibit B Cell Proliferation and Activity.
Schroeder JC, Puntigam L, Hofmann L, Jeske SS, Beccard IJ, Doescher J, Laban S, Hoffmann TK, Brunner C, Theodoraki MN, Schuler PJ
Cancers 2020 Jul 29;12(8)
Cancers 2020 Jul 29;12(8)
IGF-1 enhances BMSC viability, migration, and anti-apoptosis in myocardial infarction via secreted frizzled-related protein 2 pathway.
Lin M, Liu X, Zheng H, Huang X, Wu Y, Huang A, Zhu H, Hu Y, Mai W, Huang Y
Stem cell research & therapy 2020 Jan 9;11(1):22
Stem cell research & therapy 2020 Jan 9;11(1):22
PF-127 hydrogel plus sodium ascorbyl phosphate improves Wharton's jelly mesenchymal stem cell-mediated skin wound healing in mice.
Deng Q, Huang S, Wen J, Jiao Y, Su X, Shi G, Huang J
Stem cell research & therapy 2020 Apr 3;11(1):143
Stem cell research & therapy 2020 Apr 3;11(1):143
Impact of Fibronectin Knockout on Proliferation and Differentiation of Human Infrapatellar Fat Pad-Derived Stem Cells.
Wang Y, Fu Y, Yan Z, Zhang XB, Pei M
Frontiers in bioengineering and biotechnology 2019;7:321
Frontiers in bioengineering and biotechnology 2019;7:321
Therapeutic Effects of Human Urine-Derived Stem Cells in a Rat Model of Cisplatin-Induced Acute Kidney Injury In Vivo and In Vitro.
Sun B, Luo X, Yang C, Liu P, Yang Y, Dong X, Yang Z, Xu J, Zhang Y, Li L
Stem cells international 2019;2019:8035076
Stem cells international 2019;2019:8035076
Sodium Selenite Improves The Therapeutic Effect Of BMSCs Via Promoting The Proliferation And Differentiation, Thereby Promoting The Hematopoietic Factors.
Yan D, Tang B, Yan L, Zhang L, Miao M, Chen X, Sui G, Zhang Q, Liu D, Wang H
OncoTargets and therapy 2019;12:9685-9696
OncoTargets and therapy 2019;12:9685-9696
Isolation and Culture of Human Stem Cells from Apical Papilla under Low Oxygen Concentration Highlight Original Properties.
Rémy M, Ferraro F, Le Salver P, Rey S, Genot E, Djavaheri-Mergny M, Thébaud N, Boiziau C, Boeuf H
Cells 2019 Nov 21;8(12)
Cells 2019 Nov 21;8(12)
High-yield isolation of menstrual blood-derived endometrial stem cells by direct red blood cell lysis treatment.
Sun Y, Ren Y, Yang F, He Y, Liang S, Guan L, Cheng F, Liu Y, Lin J
Biology open 2019 May 2;8(5)
Biology open 2019 May 2;8(5)
Modulation of proliferation and differentiation of gingiva‑derived mesenchymal stem cells by concentrated growth factors: Potential implications in tissue engineering for dental regeneration and repair.
Chen X, Chen Y, Hou Y, Song P, Zhou M, Nie M, Liu X
International journal of molecular medicine 2019 Jul;44(1):37-46
International journal of molecular medicine 2019 Jul;44(1):37-46
The influence of chemotherapy on adenosine-producing B cells in patients with head and neck squamous cell carcinoma.
Ziebart A, Huber U, Jeske S, Laban S, Doescher J, Hoffmann TK, Brunner C, Jackson EK, Schuler PJ
Oncotarget 2018 Jan 19;9(5):5834-5847
Oncotarget 2018 Jan 19;9(5):5834-5847
Human umbilical cord-derived mesenchymal stem cells ameliorate the enteropathy of food allergies in mice.
Yan N, Xu J, Zhao C, Wu Y, Gao F, Li C, Zhou W, Xiao T, Zhou X, Shao Q, Xia S
Experimental and therapeutic medicine 2018 Dec;16(6):4445-4456
Experimental and therapeutic medicine 2018 Dec;16(6):4445-4456
BMP14 induces tenogenic differentiation of bone marrow mesenchymal stem cells in vitro.
Wang D, Jiang X, Lu A, Tu M, Huang W, Huang P
Experimental and therapeutic medicine 2018 Aug;16(2):1165-1174
Experimental and therapeutic medicine 2018 Aug;16(2):1165-1174
A high-yield isolation and enrichment strategy for human lung microvascular endothelial cells.
Gaskill C, Majka SM
Pulmonary circulation 2017 Mar;7(1):108-116
Pulmonary circulation 2017 Mar;7(1):108-116
Human umbilical cord mesenchymal stem cells improve the reserve function of perimenopausal ovary via a paracrine mechanism.
Li J, Mao Q, He J, She H, Zhang Z, Yin C
Stem cell research & therapy 2017 Mar 9;8(1):55
Stem cell research & therapy 2017 Mar 9;8(1):55
Low-dose interleukin-2 promotes STAT-5 phosphorylation, T(reg) survival and CTLA-4-dependent function in autoimmune liver diseases.
Jeffery HC, Jeffery LE, Lutz P, Corrigan M, Webb GJ, Hirschfield GM, Adams DH, Oo YH
Clinical and experimental immunology 2017 Jun;188(3):394-411
Clinical and experimental immunology 2017 Jun;188(3):394-411
A Member of the Nuclear Receptor Superfamily, Designated as NR2F2, Supports the Self-Renewal Capacity and Pluripotency of Human Bone Marrow-Derived Mesenchymal Stem Cells.
Zhu N, Wang H, Wang B, Wei J, Shan W, Feng J, Huang H
Stem cells international 2016;2016:5687589
Stem cells international 2016;2016:5687589
Spontaneous Differentiation of Human Mesenchymal Stem Cells on Poly-Lactic-Co-Glycolic Acid Nano-Fiber Scaffold.
Sonomoto K, Yamaoka K, Kaneko H, Yamagata K, Sakata K, Zhang X, Kondo M, Zenke Y, Sabanai K, Nakayamada S, Sakai A, Tanaka Y
PloS one 2016;11(4):e0153231
PloS one 2016;11(4):e0153231
CRISPR/Cas9-based genetic correction for recessive dystrophic epidermolysis bullosa.
Webber BR, Osborn MJ, McElroy AN, Twaroski K, Lonetree CL, DeFeo AP, Xia L, Eide C, Lees CJ, McElmurry RT, Riddle MJ, Kim CJ, Patel DD, Blazar BR, Tolar J
NPJ Regenerative medicine 2016;1:16014-
NPJ Regenerative medicine 2016;1:16014-
Transendothelial migration of human umbilical mesenchymal stem cells across uterine endothelial monolayers: Junctional dynamics and putative mechanisms.
Ebrahim NA, Leach L
Placenta 2016 Dec;48:87-98
Placenta 2016 Dec;48:87-98
NR2F2 regulates bone marrow-derived mesenchymal stem cell-promoted proliferation of Reh cells.
Zhu N, Wang H, Wei J, Wang B, Shan W, Lai X, Zhao Y, Yu J, Huang H
Molecular medicine reports 2016 Aug;14(2):1351-6
Molecular medicine reports 2016 Aug;14(2):1351-6
MIF Plays a Key Role in Regulating Tissue-Specific Chondro-Osteogenic Differentiation Fate of Human Cartilage Endplate Stem Cells under Hypoxia.
Yao Y, Deng Q, Song W, Zhang H, Li Y, Yang Y, Fan X, Liu M, Shang J, Sun C, Tang Y, Jin X, Liu H, Huang B, Zhou Y
Stem cell reports 2016 Aug 9;7(2):249-62
Stem cell reports 2016 Aug 9;7(2):249-62
Human Gingival Integration-Free iPSCs; a Source for MSC-Like Cells.
Umezaki Y, Hashimoto Y, Nishishita N, Kawamata S, Baba S
International journal of molecular sciences 2015 Jun 15;16(6):13633-48
International journal of molecular sciences 2015 Jun 15;16(6):13633-48
FOXF1 mediates mesenchymal stem cell fusion-induced reprogramming of lung cancer cells.
Wei HJ, Nickoloff JA, Chen WH, Liu HY, Lo WC, Chang YT, Yang PC, Wu CW, Williams DF, Gelovani JG, Deng WP
Oncotarget 2014 Oct 15;5(19):9514-29
Oncotarget 2014 Oct 15;5(19):9514-29
Epithelial cell differentiation of human mesenchymal stromal cells in decellularized lung scaffolds.
Mendez JJ, Ghaedi M, Steinbacher D, Niklason LE
Tissue engineering. Part A 2014 Jun;20(11-12):1735-46
Tissue engineering. Part A 2014 Jun;20(11-12):1735-46
Hypoxia-cultured human adipose-derived mesenchymal stem cells are non-oncogenic and have enhanced viability, motility, and tropism to brain cancer.
Feng Y, Zhu M, Dangelmajer S, Lee YM, Wijesekera O, Castellanos CX, Denduluri A, Chaichana KL, Li Q, Zhang H, Levchenko A, Guerrero-Cazares H, Quiñones-Hinojosa A
Cell death & disease 2014 Dec 11;5(12):e1567
Cell death & disease 2014 Dec 11;5(12):e1567
Myoblasts derived from normal hESCs and dystrophic hiPSCs efficiently fuse with existing muscle fibers following transplantation.
Goudenege S, Lebel C, Huot NB, Dufour C, Fujii I, Gekas J, Rousseau J, Tremblay JP
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Nov;20(11):2153-67
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Nov;20(11):2153-67
Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression.
Deaglio S, Dwyer KM, Gao W, Friedman D, Usheva A, Erat A, Chen JF, Enjyoji K, Linden J, Oukka M, Kuchroo VK, Strom TB, Robson SC
The Journal of experimental medicine 2007 Jun 11;204(6):1257-65
The Journal of experimental medicine 2007 Jun 11;204(6):1257-65
T regulatory and primed uncommitted CD4 T cells express CD73, which suppresses effector CD4 T cells by converting 5'-adenosine monophosphate to adenosine.
Kobie JJ, Shah PR, Yang L, Rebhahn JA, Fowell DJ, Mosmann TR
Journal of immunology (Baltimore, Md. : 1950) 2006 Nov 15;177(10):6780-6
Journal of immunology (Baltimore, Md. : 1950) 2006 Nov 15;177(10):6780-6
Characterization of an IgM Fc-binding receptor on human T cells.
Nakamura T, Kubagawa H, Ohno T, Cooper MD
Journal of immunology (Baltimore, Md. : 1950) 1993 Dec 15;151(12):6933-41
Journal of immunology (Baltimore, Md. : 1950) 1993 Dec 15;151(12):6933-41
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with Mouse IgG1 K Isotype Control APC (Product # 17-4714-81) (blue histogram) or Anti-Human CD73 APC (purple histogram). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Characterization of BMSCs. (A) Representative images show immunofluorescent staining of CD29 and CD44 on passage 2 BMSCs. Scale bars, 50 um. Pink, CD29; green, CD44; blue, DAPI. (B) Flow cytometry identified rat BMSC-positive markers CD44, CD90 and CD73, but not the marker CD45 on the surface of the cells. CD, cluster of differentiation; BMSC, bone marrow mesenchymal stem cell.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Characterization of MSC-like cells generated from iPSCs. ( A ) Morphology of HGF-iPSCs 1-1 at passage 45 cultured on GFRM with mTeSR1 ( left ). Morphology of MSC-like cells from HGF-iPSCs 1-1 differentiated (MSLCs 1-1) at passage 4 ( right ); ( B ) Flow cytometry analysis of MSC-related surface markers (CD44, CD73, CD90, and CD105), hematopoietic markers (CD34 and CD45), and pluripotent markers (SSEA3 and TRA1-60) on MSLCs 1-1 at passage 10. BMMSCs were used (passage 5) as positive control and HGF-iPSCs 1-1 (passage 45) as negative control; ( C ) MSC-like cells from HGF-iPSCs 1-2 (MSLCs 1-2) were differentiated at passage 23. Growth curve of BMMSCs, MSLCs 1-1 and 1-2. Scale bar = 400 mum. GFRM, Growth factor-reduced Matrigel.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 B cells were harvested after 2 days of co-culture with either NC or HNSCC exosomes or PBS and stained for FACS analysis. ( A ) Frequency of CD39 + CD73 + regulatory B cells. ( B ) The expression of CD39 on B cells was reduced after co-culture with NC or HNSCC exosomes. ( C ) Expression of CD73 on B cells. ( D ) Expression of CD86 on B cells. ( E ) The expression of CD19 on B cells was increased by stimulation with CD40L and IL-4. **: p < 0.01; *: p < 0.05, n = 8 (HNSCC), n = 6 (NC), n = 5 (Unstim). Unstim = Unstimulated B cells, NC = no cancer (exosomes from blood plasma of healthy volunteers), HNSCC, exosomes from blood plasma of HNSCC patients.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Flow cytometry analysis of phenotype characterization of hUCMSCs. Phenotype of CD73, CD90, CD105, CD14, CD34, CD45, CD79a and HLA-DR of hUCMSCs was detected by flow cytometry. Intensity >= 95% represented strong expression while
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
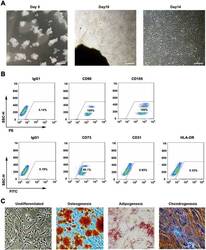
- Fig. 1 WJMSCs isolation and characterization. a Primary cell isolation procedure from Wharton''s jelly tissue. The migrated cells exhibited typical fibroblast-like morphology. Scale bar, 500 mum. b Flow cytometry analysis of P4 cells using mesenchymal stem cell markers (CD90, CD105, CD73), endothelial cell marker (CD31), and MHC class II protein HLA-DR. Isotypic antibodies (IgG1-PE and IgG1-FITC) were used as negative controls. c Representative stained images show that the fourth passage WJMSCs could differentiate into osteocytes (Alizarin Red S), adipocytes (Oil Red O), and chondrocytes (Alcian blue). Scale bar, 100 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
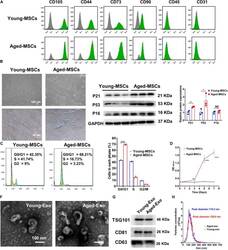
- FIGURE 1 Characterization of young and aged MSCs and exosomes. (A) Surface marker profiling of young-MSCs and aged-MSCs. (B) SA-beta-Gal staining showed that senescence increased significantly in aged MSCs. (C) Representative immunoblot images and quantitative analysis of p21, p53, and p16 protein level in young and aged-MSCs. ( n = 3). (D) Quantitation of cell cycle phases by propidium iodide staining. ( n = 3). (E) The CCK-8 assay showed that aged MSCs grew more slowly than young MSCs. ( n = 6). (F) Young and aged exosomes were observed using TEM. (G) The exosome surface markers were analyzed by Western blot. (H) Nanoparticle tracking analysis was used to analyze the particle size and concentration of Young-Exo and Aged-Exo. * p < 0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001; NS, not significant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Chondrogenesis using minicircle-transfected hiPSC-derived OG cells. ( a ) Scheme of chondrogenic differentiation process from hiPSCs. Minicircles were transfected after OG cells were induced. ( b ) Morphology of the hiPSC colony. ( c ) Morphology of the generated EBs. ( d ) Image of outgrowth cells derived from EBs attached to a gelatin-coated culture dish. ( e ) Morphology of OG cells before transfection. ( f ) Alizarin red-stained osteogenic cells differentiated from OG cells. ( g ) Oil red O staining image of adipogenic cells differentiated from OG cells. ( h ) Chondrogenic pellet generated from OG cells stained with alcian blue. Relative gene expression of ( i ) CD44, ( j ) CD73, ( k ) CD90, and ( l ) CD105 in OG cells. Percentage of ( m ) CD44, ( n ) CD73, ( o ) CD90, and ( p ) CD105 positive cells. ( q ) Fluorescence microscopy of mcMock-transfected OG cells. ( r ) Fluorescence microscopy of mcBMP2-transfected OG cells. ( s ) Fluorescence microscopy of mcTGFbeta3-transfected OG cells. ( t ) Percentage of OG cells transfected with each minicircle vectors. ( u ) Gel image of the PCR results against the insert of mcBMP2 and mcTGFbeta3 in transfected OG cells. Data are presented as mean +- standard deviation from three independent sets of experiments. Scale bars represents 200 mum. ** p < 0.01 and *** p < 0.001 indicate statistical significance. EB: embryonic body; OG: outgrowth; CDM: chondrogenic differentiation media; RFP: red fluorescence protein; MSC: mesenchym
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Identification of dental pulp stem cells (DPSCs). Human DPSCs were positive for the cell surface antigens CD73, CD90, and CD105, as well as negative for CD14, CD20, CD34, and CD45 demonstrated by flow cytometry ( A ). DPSCs were cultured under osteogenic ( B , 14 days) or adipogenic ( C , 21 days) conditions, and showed mineralized nodules and lipid clusters as revealed by alizarin red and oil red staining, respectively. Scale bar = 400 ( B ) or 100 ( C ) mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Expression of endothelial cell surface determinants by primary lung MVEC. Lung cells were incubated with primary antibodies directly conjugated to fluorophores and staining intensity analyzed by flow cytometry. Representative dot plots are presented. Human lung FB and MSC were used as known negative controls, and PAEC as a known positive control. DAPI was used to exclude dead cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 AD-MSCs characterization; (A) Flow cytometry assay to assess the CD markers in the surface of extracted AD-MSCs. Target cells were positive for CD73 (95.1%), and CD90 (90.6%) surface markers and were negative for CD44 and CD34 markers. (B) Morphology of AD-MSCs at passage 3. (C) Oil red staining to prove the adipogenic potential differentiation of AD-MSCs. (D) Alizarin-red staining to confirm the osteogenic potential differentiation of AD-MSCs. Scale bar = 100 mum. Abbreviations: AD-MSCs: adipose-derived mesenchymal stem cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 CD73 positive cells are fibrogenic and adipogenic but not myogenic in culture. A. FACS analysis showing CD73 staining of single cells from human muscle biopsy. Average fraction of CD73+ CD45- CD31- cells in wild type muscle is 4.5% (n = 4). B. CD73+ and CD73- cells were cultured in myogenic growth medium and stained with MyoD; or cultured in myogenic differentiation medium, and stained with MHC. C. CD73+ and CD73- cells were cultured in adipogenic differentiation medium, then stained with Oil red O. CD73+ cells were also cultured in fibrogenic differentiation medium, then stained with Sirius Red/Fast Green. Figure 1
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Culture and identification of bone marrow mesenchymal stem cells. (A) Light microscopy of BMSCs (scale bar=100 um for the left image and 200 um for the right image). (B) Percentage of CD34-, CD45-, CD73-, CD90- and CD105-positive BMSCs were detected by flow cytometry. BMSC, bone marrow mesenchymal stem cell.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 11R-VIVIT increases the osteogenic potential of osteoporotic BMSCs. (A) Flow cytometric analysis of BMSC surface antigens. (B) ALP staining results. (C) Alizarin Red S staining results. (D) Quantitative results of ALP staining. (E) Quantitative results of Alizarin Red S staining. (F) Semi-quantitative PCR detection of osteogenesis-related gens. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Characterization of UCSCs. A UCSCs display a spindle shaped and fibroblast-like morphology. B High UCSCs expression of CD90, CD29, CD73, and CD105, and low expression of HLA using flow cytometry
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Characterization of human dental pulp stem cells (hDPSCs) cultured in Neurocult(tm) proliferation medium by flow cytometry. ( A ) Data quantification as representative histograms at 3 days in vitro (DIV) (gray) and 7DIV (black) for CD90, CD105 and CD73 mesenchymal stromal cell (MSC) markers, CD45 hematopoietic and CD31 endothelial markers ( n = 3). Data are represented as the average percentage and standard error of the mean (SEM+-), with respect to the total cell population. ( B ) Representative cytometry of CD90, CD105 and CD73 mesenchymal, CD45 hematopoietic and CD31 endothelial markers in hDPSCs cultured in Neurocult proliferation medium at 3DIV (gray filling), with respect to negative controls (no filling). ( C ) Representative cytometry of CD90, CD105 and CD73 mesenchymal, CD45 hematopoietic and CD31 endothelial markers in hDPSCs cultured in Neurocult proliferation medium at 7DIV (gray filling), with respect to negative controls (no filling). *: p < 0.05. Kruskal-Wallis with Dunn's post hoc test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
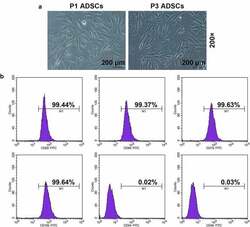
- Figure 1. Morphology and immune phenotype of adipose-derived stem cells (ADSCs) were identified by morphological observation and flow cytometry. (a) Morphology of the primary (P1) and third passage (P3) of ADSCs. Images were acquired at 200x magnification. (b) Immune phenotype of ADSCs. The average data from three independent experiments were shown as mean +- standard deviation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
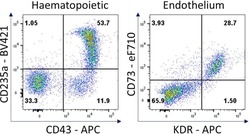
- Figure 7 Hematopoietic and endothelial cell characterization Flow cytometry analysis of day 4 hemogenic culture for hematopoietic (CD43, CD235a) and endothelial (KDR, CD73) cell surface markers.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 ( A ) Results of fluorescence-activated cell sorting (FACS) analysis on hBM-MSCs surface markers (CD73 and CD105) and IgG control after PEMF for three days. ( B ) Immunohistochemical analysis of MAP-2 antibodies on hBM-MSCs cultured after PEMF for three days (original magnification: 100x); and immunofluorescence staining of NF-L antibodies on hBM-MSCs after PEMF for three days (original magnification: 400x).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 CD73 expression and activity in serum-derived exosomes. (A) A representative western blot image for CD73 in purified exosomes (10 ug loaded protein). CD9 was used as specific exosomal marker and calregulin as negative marker. (B) A representative flow cytometry dot plot showing the expression of CD73 on isolated exosomes captured for CD63 and bound to magnetic beads, labeled with a specific antibody anti-CD73 (red line) or the isotype IgG control (green line). (C) Levels of CD73 expression as relative fluorescence intensity (RFI) values on exosomes isolated from healthy donors (HD) (n=9) and patients with melanoma (MP) at baseline (n=27). Comparison of the fold change in the 15 N adenosine production, measured by mass spectrometry, after incubating for 2 hours 5 ug of exosomes isolated from serum of HD (n=6) or MP (n=9) with 15 N AMP (10 uM) (D), with or without the anti-human CD73 mAb (5 ug/mL) or APCP (100 uM) (E), (n=5). Data are mean+-SD (C, D, E). P values are obtained from a repeated measures (RM) one-way analysis of variance, followed by Dunnet's multiple comparison test (E).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Baseline levels of exosomal CD73 expression in responders and non-responders to anti-PD-1 blockers nivolumab or pembrolizumab. CD73 expression was evaluated by beads-assisted flow cytometry on exosomes isolated from patients with melanoma prior to initiation of treatment. Responders n=11. Non-responders n=14. Data are mean+-SD. PD-1, programmed cell-death protein 1; RFI, relative fluorescence intensity.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Cell surface analysis of AMSCs within the transitional culture AMSCs were cultured in a 10% collagen type I gel for 7 days and embedded in a 0.2% collagen type I gel, making up the transitional culture for an additional 14 days. Flow cytometric analysis was conducted for PDPN, CD73, and CD146 (representative data presented).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Primary human adipose-derived cells cultured in hypoxia (hAMSCs-H) and normoxia (hAMSCs-N) are both MSCs but normoxia-cultured cells show increased signs of senescence, such as increased area and elongated morphology, compared with hypoxia-cultured cells. ( a ) hAMSCs were isolated from human fat tissue and cultured in hypoxic (1.5% oxygen) or normoxic (21% oxygen) conditions in vitro . The viability, mobility, tumor tropism, safety, and tumorigenic potential were subsequently compared in vitro and in vivo . ( b ) Differentiation assay. hAMSCs were cultured in control media and differentiation media for 3 weeks, 10 days after the second passage. Three different stains were performed to assess differentiation capabilities (scale bar, 100 mu m). ( c ) Flow cytometric analysis was performed to confirm the absence of CD31-, CD34-, and CD45-positive cells in both cell cultures. In addition, primary hAMSC cultures expressed high levels of CD73, CD90, and CD105, both in hypoxic and normoxic culture conditions at day 10 after passage 2. ( d ) Representative images of cell morphologies of hAMSCs on 2D surface (scale bar, 200 mu m). ( e ) Schematic of 3D-nanopatterned surface used to assess morphology and motility. ( f ) Images of cell morphologies of hAMSCs on 3D-nanopatterned surface (scale bar, 200 mu m). ( g - j ) The length, width, area, and length-to-width ratio were measured and compared after cell aligned on the nanopattern surface. Error bars represent S.E.M. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Hypoxia-cultured primary human adipose-derived mesenchymal stem cells (hAMSCs-H) retain a greater proliferation capacity compared with normoxia-cultured primary hAMSCs (hAMSCs-N) when exposed to GBM media. hAMSCs-H maintain stem cell characteristics when exposed to GBM media. ( a ) Representative MRI of GBM from a patient. ( b ) Schema showing the collection of GBM CM and culture of hAMSCs in filtered GBM CM for proliferation and migration assays. ( c ) MTT assay was used to determine the effects of hypoxic conditions on the proliferative capacity of primary hAMSCs in GBM CM. In GBM CM, hAMSCs-H showed greater proliferation at day 10 and 15 compared with hAMSCs-N. ( d ) Ki-67 immunostaining was performed to quantify the number of proliferating cells in GBM CM. Proliferative capacities of hAMSCs-H and hAMSCs-N are shown in GBM CM (normalized to hAMSC-N proliferative capacity in control media). In GBM CM, hAMSCs-H had a greater proportion of proliferating cells than hAMSCs-N. ( e) Differentiation assay. hAMSCs were cultured in control media, differentiation media, and GBM CM for 3 weeks, 10 days after the second passage. Three stainings were performed to assess the differentiation capabilities (scale bar, 100 mu m). Both hAMSCs-N and hAMSCs-H maintained tri-lineage differentiation capability in GBM CM. ( f ) Flow cytometric analysis for CD31, CD34, CD45, CD73, CD90, and CD105 in hAMSC-N and hAMSC-H cultures after exposure to GBM CM for 20 days. hAMSCs-H maintained MSC
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Characteristics of BM-MSCs. (a) Representative morphology of BM-MSCs. Scale bar = 500 mu m. (b) Representative flow cytometric characterization of cell surface markers expressed on BM-MSCs. Isotypic controls were represented by the gray filled histograms.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Characteristics of transfected BM-MSCs. (a) Representative morphology of transfected BM-MSCs in negative control group. (b) More than 90% of BM-MSCs expressed GFP in negative control group. (c) Representative morphology of transfected BM-MSCs in knock-down group. (d) More than 90% of BM-MSCs expressed GFP in knock-down group. (e) Representative flow cytometric characterization of cell surface markers expressed on transfected BM-MSCs. Isotypic controls were represented by black line. The red line represented the negative control group and the blue line represented the knock-down group. (f) The knock-down of NR2F2 was confirmed by western blot analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 CESCs Shared Features with BM-MSCs Regarding Morphology, Stem Cell Surface Markers, and Differentiation Ability (A) H&E staining of the tissue section. (B) Morphology of CESCs in agarose after seeding 6 weeks later. (C) Histologic section stained with Alcian blue of chondrified pellets in which CESCs formed in chondrogenic induction medium after 3 weeks. (D) Alizarin red staining of CESCs that underwent osteogenic induction for 3 weeks. (E) Immunophenotypic profile of stem cells in CESCs by flow cytometric analysis. The green lines indicate the fluorescence intensity of cells stained with the corresponding antibodies, and the red lines represent isotype-matched negative control cells. (F) Percentages of CESCs expressing different stem cell markers (n = 6 independent experiments). Data represent the mean +- SD.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Mesenchymal stem cell derivation. ( a ) MSC differentiation. Mono-layer iPSCs were subjected to bFGF, PDGFab and EGF resulting in differentiation to a cell population with spindle-shaped morphology (right). ( b ) FACS analysis. Passage 3 MSCs were analysed for cell surface expression of CD73, CD105, and CD90 ( n =3 experiments), and histogram analysis is shown in blue. Isotype antibody control FACS histograms are shown in pink. ( c , d ) Tri-lineage differentiation. ( c ) Oil red-O staining demonstrating the ability of iPSC-derived MSCs to form adipose cells. ( d ) Alizarin red staining of osteogenic progeny. ( e ) Toluidine blue staining of chondrogenic cells from MSCs. ( c - e ) Representative images of at least two different MSC pools and n =3-4 replicates. FACS, fluorescence-activated cell sorting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Cell proliferation capacity in human IPFSCs after FN1-KO. FN1-KO cells were compared with copGFP in cell increase (A) , percentage of cells in the S and G 2 phases (B) , and surface markers [SSEA4 (C) , CD73 (D) , CD90 (E) , CD105 (F) , and CD146 (G) ] by flow cytometry; stemness genes ( NANOG, SOX2, KLF4, BMI1, MYC, NOV, POU5F1 , and NES ) (H) , senescent genes ( CDKN1A, CDKN2A , and TP53 ) (I) , and the mesenchymal condensation gene ( CDH2 ) (J) by qPCR. GAPDH was used as an endogenous control. Data are shown as bar charts. * indicates a significant difference compared to the corresponding copGFP group ( P < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Proliferation capacity of human IPFSCs after expansion on the dECMs deposited by FN1-KO cells. Passage 15 human IPFSCs were compared after expansion on dECMs deposited by Cas9-sgFN1a/b transduced cells (sgFN1a ECM and sgFN1b ECM, respectively) with those deposited by copGFP (copGFP ECM) and those grown on TCP (TCP) as controls in cell increase (A) , percentage of cells in the S and G 2 phases (B) , and surface markers [SSEA4 (C) , CD73 (D) , CD90 (E) , and CD105 (F) ] by flow cytometry; stemness genes ( NANOG, SOX2, KLF4, BMI1, MYC, NOV, POU5F1 , and NES ) (G) , senescence genes ( CDKN1A, CDKN2A , and TP53 ) (H) , and the mesenchymal condensation gene ( CDH2 ) (I) by qPCR. GAPDH was used as an endogenous control. Data are shown as bar charts. *indicates a significant difference compared to the corresponding copGFP group ( P < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Growth characteristics of USCs. (a) The morphology of USCs by passage and differentiation. Single, small, compact rice grain-like cells were observed on the third day after initial seeding, and they formed a colony on the seventh day. The cells were considered to be at P0 when the confluence reached 70-80% and were passaged to the next generation. The USCs maintained the rice grain-like morphology after several passages, and USCs from the P3 generation were induced to differentiate into SMCs and UCs. The cells showed an elongated and spindle-shaped morphology after SMC differentiation and a cobblestone-shaped morphology after UC differentiation. Scale bar: 50 mu m, 100 mu m, and 200 mu m. (b) The growth curve of USCs from the P3 generation. (c) Detection of surface markers in USCs using flow cytometry. USCs did not express hematopoietic stem cell markers (CD31: 0.45%, CD34: 0.28%) but expressed MSC markers (CD44: 100%, CD73: 97.1%, and CD105: 96.3%) and pericyte markers (CD146: 95.7%). (d) Detection of surface markers in USCs using IF. USCs did not express hematopoietic stem cell markers (CD31, CD34, and CD45) but did express MSC markers (CD44 and CD133), the ESC marker SSEA4, and pericyte markers (CD146, PDGFRB, and NG2). NC: negative control; PDGFRB: platelet-derived growth factor beta-receptor; NG2: neural/glial antigen 2. Scale bar: 25 mu m.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Characterization of hUC-MSCs. (A) Morphological observations of hUC-MSCs. Umbilical cord tissues were cultured for >15 days and long spindle-shaded fibroblastic cells were observed around the tissue using Zeiss light microscopy (scale bar, 100 um). (B) Phenotyping of hUC-MSCs. hUC-MSCs were stained with a fluorescein-labeled antibodies (CD34, CD45, CD73, CD90, CD105, CD14, CD19 and HLA-DR) and analyzed with a flow cytometer. (C) Adipogenic and (D) osteogenic differentiation of hUC-MSCs. hUC-MSCs were cultured in adipogenic and osteogenic medium, respectively. Lipid droplets in the adipocytes are presented with Oil Red O staining (scale bar, 100 um). hUC-MSCs-derived osteoblasts were detected with Alizarin Red staining (scale bar, 200 um). (E) hUC-MSCs inhibit the proliferation of CFSE-labeled CD4 + T cells, which were activated by Con A stimulation. Experiments were repeated three times and representative graphs and images are presented. hUC-MSC, human umbilical cord-derived mesenchymal stem cell; MSC Sup, culture supernatant of hUC-MSCs; Con A, concanavalin A; CFSE, carboxyfluorescein succinimidyl ester.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3. The phenotype of MenSCs. (A,B) To determine the immunophenotype of MenSCs, P3 MenSCs-DGC ( n =5) and MenSCs-RLB ( n =5) were stained by corresponding conjugated antibodies and analyzed by FACS. Both MenSCs-DGC and MenSCs-RLB positively expressed classical ASCs' markers (CD29, CD44, CD73, CD90 and CD105) and HLA-ABC; they did not express hematopoietic stem cell markers (CD34 and CD45) and HLA-DR. (C) The quantification of flow cytometry results of A and B.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Effect of IGF-1 overexpression on BMSCs. a Identification of BMSCs by flow cytometry analysis. b Supernatants from cultured BMSCs-NC and BMSCs-IGF-1 were collected and subjected to ELISA to determine IGF-1 levels. c Cells were exposed to hypoxia for 48 h, and cell proliferation was determined by MTS assay. d Apoptosis was determined by TUNEL assay. e Cell migration was determined by Transwell assay. f Expression of OCT4, NANOG, cleaved caspase-3, BAX, and BCL-2 was determined by Western blotting. All assays were performed in triplicate (* P < 0.05, ** P < 0.01, *** P < 0.001)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Protein expression analysis. SCAPs from EXP III (early passages of UBx-SCAP) were grown at 3% or 21% O 2 . ( A ) Graphs showing flow cytometry analysis of different markers as indicated ((% of positive cells, left column) and mean of fluorescence intensity (MFI, in arbitrary units, right column)). Numbers 1, 2, 3 refer to the three individuals. For each group, three to six samples from different early passages were analyzed. Statistical analyses were done with a Mann Whitney test. * p < 0.05. ( B ) Western blot analysis of UBx-SCAP-1 (early passage) grown under 21% or 3% O 2 and of human iPSCs (induced pluripotent stem cells, IMR90 cell line) used as a positive control for expression of Oct4 and Nanog. ERK2 was used as the loading control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Characterization of GMSCs. (A) Flow cytometry analysis indicated that human GMSCs expressed CD73, CD105 and CD90, but not CD45. (B) Expression of stem cell markers in GMSCs. Cells cultured in a 24-well plate were fixed and stained with specific antibodies against human vimentin, CD90, CD73 and cytokeratin (CK). The cells were incubated with rhodamine- or fluorescein isothiocyanate-conjugated secondary antibodies and then observed under a fluorescence microscope (magnification, x200). (C) Immunofluorescence staining revealed that the cytoplasm of CD90- and vimentin-stained cells exhibited red fluorescence, and the cytoplasm of S100A4-stained cells exhibited green fluorescence. The nuclei of all cells exhibited blue fluorescence, whereas the cytokeratin-stained cells exhibited no fluorescence (magnification, x200). GMSCs, gingiva-derived mesenchymal stem cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Identification of BMSCs in mice. Morphological observations at different magnifications (40x, 100x, and 200x) showed that the bone marrow stem cells (BMSCs) isolated from mice were homogeneously elongated ( A ). Various cell surface markers of BMSCs were detected on the cells, including CD90, CD73, CD105, CD44, and CD45 ( B ). The values are from triplicate determinations. A P value less than 0.05 was considered to be statistically significant. ***Indicates a P value less than 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Flow cytometry of CD cell surface markers for cells cultured under hypoxia and normoxia. The positive CD markers for MSCs as detected by the fluorescent antibodies anti-CD73 FITC, anti-CD105 PE, and anti-CD90 PE Cy7. The negative markers of MSCs were detected using anti-CD14 FITC, anti-CD45 PerCP, anti-CD34-R-PE, and anti-CD19 PE-Cy7 antibodies. Unstained cell for each condition was used as negative controls.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 FACS analysis of hESC-derived mesenchymal-like precursors generated by culture in MB1 . ( a ) The culture of hESCs in the MB1 culture medium (MB1-hESCs) induced their differentiation in mesenchymal-like stem cells expressing CD73. ( b ) However, in the MB-1 medium,
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry