Antibody data
- Antibody Data
- Antigen structure
- References [183]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [18]
- Other assay [51]
Submit
Validation data
Reference
Comment
Report error
- Product number
- A-21270 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Anti-Golgin-97 Monoclonal Antibody (CDF4)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Anti-human golgin-97 antibodies were originally isolated from the serum of a patient with the autoimmune disease known as Sjögren's syndrome. These antibodies recognize a 97 kDa protein called golgin-97, a member of the granin family of proteins and a peripheral membrane protein localized on the cytoplasmic face of the Golgi apparatus. Because the antibody recognizes a protein unique to the Golgi apparatus of most vertebrate species, it is useful for immunodetection and identification of the Golgi apparatus in cells. To prepare a stock solution, reconstitute the antibody in 0.5-1 mL of phosphate-buffered saline, pH 7.4, containing 1% BSA. Store the solution for up to two weeks at 4°C with the addition of 2 mM sodium azide. For longer storage, divide the solution into single-use aliquots and freeze at -20°C.
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- CDF4
- Vial size
- 100 µg
- Storage
- -20°C
Submitted references Biallelic loss-of-function variants in WDR11 are associated with microcephaly and intellectual disability.
Decreased GLUT2 and glucose uptake contribute to insulin secretion defects in MODY3/HNF1A hiPSC-derived mutant β cells.
TFG binds LC3C to regulate ULK1 localization and autophagosome formation.
Hijacking of the host cell Golgi by Plasmodium berghei liver stage parasites.
Iron control of erythroid microtubule cytoskeleton as a potential target in treatment of iron-restricted anemia.
Sterol-resistant SCAP Overexpression in Vascular Smooth Muscle Cells Accelerates Atherosclerosis by Increasing Local Vascular Inflammation through Activation of the NLRP3 Inflammasome in Mice.
Rapid degradation of GRASP55 and GRASP65 reveals their immediate impact on the Golgi structure.
Diacylglycerol kinase and phospholipase D inhibitors alter the cellular lipidome and endosomal sorting towards the Golgi apparatus.
TMEM98 is a negative regulator of FRAT mediated Wnt/ß-catenin signalling.
The golgin family exhibits a propensity to form condensates in living cells.
Evaluation of active Rac1 levels in cancer cells: A case of misleading conclusions from immunofluorescence analysis.
Chromogranin A preferential interaction with Golgi phosphatidic acid induces membrane deformation and contributes to secretory granule biogenesis.
G protein-regulated endocytic trafficking of adenylyl cyclase type 9.
The GTPase Rab27b regulates the release, autophagic clearance, and toxicity of α-synuclein.
Human mastadenovirus-B (HAdV-B)-specific E3-CR1β and E3-CR1γ glycoproteins interact with each other and localize at the plasma membrane of non-polarized airway epithelial cells.
Retromer stabilization results in neuroprotection in a model of Amyotrophic Lateral Sclerosis.
TMEM16K is an interorganelle regulator of endosomal sorting.
Phenome-based approach identifies RIC1-linked Mendelian syndrome through zebrafish models, biobank associations and clinical studies.
Distinct anterograde trafficking pathways of BACE1 and amyloid precursor protein from the TGN and the regulation of amyloid-β production.
Acute inactivation of retromer and ESCPE-1 leads to time-resolved defects in endosomal cargo sorting.
Caveolin-1 suppresses tumor formation through the inhibition of the unfolded protein response.
Visualization of Protein Sorting at the Trans-Golgi Network and Endosomes Through Super-Resolution Imaging.
Restoration of F508-del Function by Transcomplementation: The Partners Meet in the Endoplasmic Reticulum.
A Novel Role for Triglyceride Metabolism in Foxp3 Expression.
Effects of Stuffer DNA on the Suppression of Choroidal Neovascularization by a rAAV Expressing a mTOR-Inhibiting shRNA.
ARFRP1 functions upstream of ARL1 and ARL5 to coordinate recruitment of distinct tethering factors to the trans-Golgi network.
Effects of Long-Term Citrate Treatment in the PC3 Prostate Cancer Cell Line.
FANCM limits ALT activity by restricting telomeric replication stress induced by deregulated BLM and R-loops.
Overexpression-mediated activation of MET in the Golgi promotes HER3/ERBB3 phosphorylation.
Biosynthesis of O-N-acetylgalactosamine glycans in the human cell nucleus.
HNF4A Haploinsufficiency in MODY1 Abrogates Liver and Pancreas Differentiation from Patient-Derived Induced Pluripotent Stem Cells.
Correction: Regulation of E-cadherin localization by microtubule targeting agents: rapid promotion of cortical E-cadherin through p130CAS/Src inhibition by eribulin.
Biallelic VARS variants cause developmental encephalopathy with microcephaly that is recapitulated in vars knockout zebrafish.
SNAREs define targeting specificity of trafficking vesicles by combinatorial interaction with tethering factors.
Golgin-97 Targets Ectopically Expressed Inward Rectifying Potassium Channel, Kir2.1, to the trans-Golgi Network in COS-7 Cells.
Reinterpretation of the localization of the ATP binding cassette transporter ABCG1 in insulin-secreting cells and insights regarding its trafficking and function.
PAQR3 Regulates Endoplasmic Reticulum-to-Golgi Trafficking of COPII Vesicle via Interaction with Sec13/Sec31 Coat Proteins.
Intravitreal Injection of AAV Expressing Soluble VEGF Receptor-1 Variant Induces Anti-VEGF Activity and Suppresses Choroidal Neovascularization.
Endoplasmic reticulum, Golgi, and lysosomes are disorganized in lung fibroblasts from chronic obstructive pulmonary disease patients.
Upon Infection, Cellular WD Repeat-Containing Protein 5 (WDR5) Localizes to Cytoplasmic Inclusion Bodies and Enhances Measles Virus Replication.
ArfGAP1 restricts Mycobacterium tuberculosis entry by controlling the actin cytoskeleton.
Parkinson disease-associated mutations in LRRK2 cause centrosomal defects via Rab8a phosphorylation.
Amnionless-mediated glycosylation is crucial for cell surface targeting of cubilin in renal and intestinal cells.
Dynamic changes in copper homeostasis and post-transcriptional regulation of Atp7a during myogenic differentiation.
Organelle membrane derived patches: reshaping classical methods for new targets.
High levels of the AR-V7 Splice Variant and Co-Amplification of the Golgi Protein Coding YIPF6 in AR Amplified Prostate Cancer Bone Metastases.
Rab33B Controls Hepatitis B Virus Assembly by Regulating Core Membrane Association and Nucleocapsid Processing.
Ablation of SNX6 leads to defects in synaptic function of CA1 pyramidal neurons and spatial memory.
Ror2 signaling regulates Golgi structure and transport through IFT20 for tumor invasiveness.
COPI-TRAPPII activates Rab18 and regulates its lipid droplet association.
Human cactin interacts with DHX8 and SRRM2 to assure efficient pre-mRNA splicing and sister chromatid cohesion.
Inhibitors of GLUT/SLC2A Enhance the Action of BCNU and Temozolomide against High-Grade Gliomas.
Lysosomal putative RNA transporter SIDT2 mediates direct uptake of RNA by lysosomes.
Two-Step Mechanism of Cellular Uptake of Cationic Gold Nanoparticles Modified by (16-Mercaptohexadecyl)trimethylammonium Bromide.
Palmitoylation of Desmoglein 2 Is a Regulator of Assembly Dynamics and Protein Turnover.
Aurora-A recruitment and centrosomal maturation are regulated by a Golgi-activated pool of Src during G2.
Excess sphingomyelin disturbs ATG9A trafficking and autophagosome closure.
Disruption of Golgi morphology and altered protein glycosylation in PLA2G6-associated neurodegeneration.
The Endoplasmic Reticulum Chaperone Calnexin Is a NADPH Oxidase NOX4 Interacting Protein.
A large Rab GTPase encoded by CRACR2A is a component of subsynaptic vesicles that transmit T cell activation signals.
Golgi membrane fission requires the CtBP1-S/BARS-induced activation of lysophosphatidic acid acyltransferase δ.
Protein Kinase D2 Assembles a Multiprotein Complex at the Trans-Golgi Network to Regulate Matrix Metalloproteinase Secretion.
Caveolin-1 is an aggresome-inducing protein.
The CD63-Syntenin-1 Complex Controls Post-Endocytic Trafficking of Oncogenic Human Papillomaviruses.
Simplified protocol for flow cytometry analysis of fluorescently labeled exosomes and microvesicles using dedicated flow cytometer.
Simplified protocol for flow cytometry analysis of fluorescently labeled exosomes and microvesicles using dedicated flow cytometer.
Sensitive detection of lysosomal membrane permeabilization by lysosomal galectin puncta assay.
Post-Golgi anterograde transport requires GARP-dependent endosome-to-TGN retrograde transport.
IGF-1 drives chromogranin A secretion via activation of Arf1 in human neuroendocrine tumour cells.
Memory and PTPIP51--a new protein in hippocampus and cerebellum.
Beclin 1 regulates neuronal transforming growth factor-β signaling by mediating recycling of the type I receptor ALK5.
Auxiliary KCNE subunits modulate both homotetrameric Kv2.1 and heterotetrameric Kv2.1/Kv6.4 channels.
RC/BTB2 is essential for formation of primary cilia in mammalian cells.
HSP47 and FKBP65 cooperate in the synthesis of type I procollagen.
Characterization of Rabaptin-5 γ isoform.
CCHCR1 interacts with EDC4, suggesting its localization in P-bodies.
Nile Tilapia Neu3 sialidases: molecular cloning, functional characterization and expression in Oreochromis niloticus.
Mutation in VPS35 associated with Parkinson's disease impairs WASH complex association and inhibits autophagy.
LAPTM5 promotes lysosomal degradation of intracellular CD3ζ but not of cell surface CD3ζ.
Arf4 is required for Mammalian development but dispensable for ciliary assembly.
An L2 SUMO interacting motif is important for PML localization and infection of human papillomavirus type 16.
Alternative transcripts and 3'UTR elements govern the incorporation of selenocysteine into selenoprotein S.
A novel domain regulating degradation of the glomerular slit diaphragm protein podocin in cell culture systems.
Intracellular itinerary of internalised β-secretase, BACE1, and its potential impact on β-amyloid peptide biogenesis.
Partial IFN-γR2 deficiency is due to protein misfolding and can be rescued by inhibitors of glycosylation.
Expression of conventional and novel glucose transporters, GLUT1, -9, -10, and -12, in vascular smooth muscle cells.
Involvement of lysosomal exocytosis in the excretion of mesoporous silica nanoparticles and enhancement of the drug delivery effect by exocytosis inhibition.
Trafficking of the Menkes copper transporter ATP7A is regulated by clathrin-, AP-2-, AP-1-, and Rab22-dependent steps.
Versican is upregulated in circulating monocytes in patients with systemic sclerosis and amplifies a CCL2-mediated pathogenic loop.
Molecular cloning and biochemical characterization of two novel Neu3 sialidases, neu3a and neu3b, from medaka (Oryzias latipes).
Regulation of WASH-dependent actin polymerization and protein trafficking by ubiquitination.
Regulation of cathepsins S and L by cystatin F during maturation of dendritic cells.
Chemical structure requirements and cellular targeting of microRNA-122 by peptide nucleic acids anti-miRs.
Spatiotemporal behavior of nuclear cyclophilin B indicates a role in RNA transcription.
Amyloid precursor protein (APP) traffics from the cell surface via endosomes for amyloid β (Aβ) production in the trans-Golgi network.
Differential regulation of myosin X movements by its cargos, DCC and neogenin.
An optimized isolation of biotinylated cell surface proteins reveals novel players in cancer metastasis.
GM130 gain-of-function induces cell pathology in a model of lysosomal storage disease.
Mre11 is expressed in mammalian mitochondria where it binds to mitochondrial DNA.
Cytomegalovirus UL103 controls virion and dense body egress.
Down-regulation of oestrogen receptor-β associates with transcriptional co-regulator PATZ1 delocalization in human testicular seminomas.
Chemical chaperone therapy: chaperone effect on mutant enzyme and cellular pathophysiology in β-galactosidase deficiency.
Dysfunction of autophagy participates in vacuole formation and cell death in cells replicating hepatitis C virus.
Splice variant-specific cellular function of the formin INF2 in maintenance of Golgi architecture.
A method to separate nuclear, cytosolic, and membrane-associated signaling molecules in cultured cells.
Tumor-associated calcium signal transducer 2 is required for the proper subcellular localization of claudin 1 and 7: implications in the pathogenesis of gelatinous drop-like corneal dystrophy.
PIST regulates the intracellular trafficking and plasma membrane expression of cadherin 23.
Open reading frame E3-10.9K of subspecies B1 human adenoviruses encodes a family of late orthologous proteins that vary in their predicted structural features and subcellular localization.
Phospholipase D family member 4, a transmembrane glycoprotein with no phospholipase D activity, expression in spleen and early postnatal microglia.
Tumor protein D52 expression and Ca2+-dependent phosphorylation modulates lysosomal membrane protein trafficking to the plasma membrane.
HSV usurps eukaryotic initiation factor 3 subunit M for viral protein translation: novel prevention target.
Geoditin A induces oxidative stress and apoptosis on human colon HT29 cells.
The cytoplasmic tail of fibrocystin contains a ciliary targeting sequence.
Storage vesicles in neurons are related to Golgi complex alterations in mucopolysaccharidosis IIIB.
LG186: An inhibitor of GBF1 function that causes Golgi disassembly in human and canine cells.
Optimization of immunoprecipitation-western blot analysis in detecting GW182-associated components of GW/P bodies.
Hepatitis B virus and hepatitis C virus interaction in Huh-7 cells.
Akt and 14-3-3 control a PACS-2 homeostatic switch that integrates membrane traffic with TRAIL-induced apoptosis.
Microenvironmental pH is a key factor for exosome traffic in tumor cells.
Primary ciliogenesis defects are associated with human astrocytoma/glioblastoma cells.
Thioesterase activity and subcellular localization of acylprotein thioesterase 1/lysophospholipase 1.
Subcellular localization of Nox4 and regulation in diabetes.
IFAP syndrome is caused by deficiency in MBTPS2, an intramembrane zinc metalloprotease essential for cholesterol homeostasis and ER stress response.
A FRET-based fluorogenic phosphine for live-cell imaging with the Staudinger ligation.
Identification and characterization of an alternatively spliced variant of the MHC class I-related porcine neonatal Fc receptor for IgG.
Altered cellular distribution and sub-cellular sorting of doppel (Dpl) protein in human astrocytoma cell lines.
Caveolin-1-dependent infectious entry of human papillomavirus type 31 in human keratinocytes proceeds to the endosomal pathway for pH-dependent uncoating.
The cytoplasmic domain of proEGF negatively regulates motility and elastinolytic activity in thyroid carcinoma cells.
A dynamic view of hepatitis C virus replication complexes.
HtrA2/Omi terminates cytomegalovirus infection and is controlled by the viral mitochondrial inhibitor of apoptosis (vMIA).
Envelope protein palmitoylations are crucial for murine coronavirus assembly.
Clathrin-dependent trafficking of subtilase cytotoxin, a novel AB5 toxin that targets the endoplasmic reticulum chaperone BiP.
Cellular trafficking of lipoteichoic acid and Toll-like receptor 2 in relation to signaling: role of CD14 and CD36.
Genome-wide and functional annotation of human E3 ubiquitin ligases identifies MULAN, a mitochondrial E3 that regulates the organelle's dynamics and signaling.
The Golgin GMAP210/TRIP11 anchors IFT20 to the Golgi complex.
A fluorimetry-based ssYFP secretion assay to monitor vasopressin-induced exocytosis in LLC-PK1 cells expressing aquaporin-2.
beta-dystrobrevin, a kinesin-binding receptor, interacts with the extracellular matrix components pancortins.
Rab11-FIP2 regulates differentiable steps in transcytosis.
Ankyrin-G is a molecular partner of E-cadherin in epithelial cells and early embryos.
Requirements for the localization of nesprin-3 at the nuclear envelope and its interaction with plectin.
Human ClC-6 is a late endosomal glycoprotein that associates with detergent-resistant lipid domains.
Secretory cytotoxic granule maturation and exocytosis require the effector protein hMunc13-4.
Missense mutations of dual oxidase 2 (DUOX2) implicated in congenital hypothyroidism have impaired trafficking in cells reconstituted with DUOX2 maturation factor.
Redox-signaling transmitted in trans to neighboring cells by melanoma-derived TNF-containing exosomes.
NGEP, a prostate-specific plasma membrane protein that promotes the association of LNCaP cells.
Multiple receptor interactions trigger release of membrane and intracellular calcium stores critical for herpes simplex virus entry.
Expression and release of soluble HLA-E is an immunoregulatory feature of endothelial cell activation.
The intraflagellar transport protein IFT20 is associated with the Golgi complex and is required for cilia assembly.
Molecular characterization of pannexins in the lens.
Leukocyte adhesion deficiency II patients with a dual defect of the GDP-fucose transporter.
Ghrelin and the growth hormone secretagogue receptor constitute a novel autocrine pathway in astrocytoma motility.
Identification of the expressed form of human cytosolic phospholipase A2beta (cPLA2beta): cPLA2beta3 is a novel variant localized to mitochondria and early endosomes.
A trans-Golgi network resident protein, golgin-97, accumulates in viral factories and incorporates into virions during poxvirus infection.
The mTOR pathway is regulated by polycystin-1, and its inhibition reverses renal cystogenesis in polycystic kidney disease.
Functional analysis of Arl1 and golgin-97 in endosome-to-TGN transport using recombinant Shiga toxin B fragment.
The transmembrane domain is essential for the microtubular trafficking of membrane type-1 matrix metalloproteinase (MT1-MMP).
Proteasome inhibition activates the transport and the ectodomain shedding of TNF-alpha receptors in human endothelial cells.
Differentiation- and maturation-dependent content, localization, and secretion of cystatin C in human dendritic cells.
Coenzyme Q distribution in HL-60 human cells depends on the endomembrane system.
Proteolytic maturation and activation of autotaxin (NPP2), a secreted metastasis-enhancing lysophospholipase D.
Intracellular localization and trafficking of fluorescein-labeled cisplatin in human ovarian carcinoma cells.
Proinflammatory mediators elicit secretion of the intracellular B-lymphocyte stimulator pool (BLyS) that is stored in activated neutrophils: implications for inflammatory diseases.
Dynamic fluorescent imaging of human immunodeficiency virus type 1 gag in live cells by biarsenical labeling.
The caveolae-mediated sv40 entry pathway bypasses the golgi complex en route to the endoplasmic reticulum.
Proteasome-dependent degradation of cytochromes P450 2E1 and 2B1 expressed in tetracycline-regulated HeLa cells.
Activation of endogenous Cdc42 visualized in living cells.
Cross-presentation of a human tumor antigen delivered to dendritic cells by HSV VP22-mediated protein translocation.
Autoantigen Golgin-97, an effector of Arl1 GTPase, participates in traffic from the endosome to the trans-golgi network.
PIBF (progesterone induced blocking factor) is overexpressed in highly proliferating cells and associated with the centrosome.
Translocation of the inhibitor of apoptosis protein c-IAP1 from the nucleus to the Golgi in hematopoietic cells undergoing differentiation: a nuclear export signal-mediated event.
Intracellular accumulation of amyloidogenic fragments of amyloid-beta precursor protein in neurons with Niemann-Pick type C defects is associated with endosomal abnormalities.
Lipid-dependent bidirectional traffic of apolipoprotein B in polarized enterocytes.
An MDR-EGFP gene fusion allows for direct cellular localization, function and stability assessment of P-glycoprotein.
Vimentin is secreted by activated macrophages.
Interaction of Hsp90 with the nascent form of the mutant epidermal growth factor receptor EGFRvIII.
The human phosphatidylinositol phosphatase SAC1 interacts with the coatomer I complex.
The melanocytic protein Melan-A/MART-1 has a subcellular localization distinct from typical melanosomal proteins.
Biologically active APRIL is secreted following intracellular processing in the Golgi apparatus by furin convertase.
Subcellular localization of the melanoma-associated protein Melan-AMART-1 influences the processing of its HLA-A2-restricted epitope.
Intracellular calcium signals regulating cytosolic phospholipase A2 translocation to internal membranes.
Intracellular redirection of plasma membrane trafficking after loss of epithelial cell polarity.
Intracellular redirection of plasma membrane trafficking after loss of epithelial cell polarity.
A novel Rab6-interacting domain defines a family of Golgi-targeted coiled-coil proteins.
Haag N, Tan EC, Begemann M, Buschmann L, Kraft F, Holschbach P, Lai AHM, Brett M, Mochida GH, DiTroia S, Pais L, Neil JE, Al-Saffar M, Bastaki L, Walsh CA, Kurth I, Knopp C
European journal of human genetics : EJHG 2021 Nov;29(11):1663-1668
European journal of human genetics : EJHG 2021 Nov;29(11):1663-1668
Decreased GLUT2 and glucose uptake contribute to insulin secretion defects in MODY3/HNF1A hiPSC-derived mutant β cells.
Low BSJ, Lim CS, Ding SSL, Tan YS, Ng NHJ, Krishnan VG, Ang SF, Neo CWY, Verma CS, Hoon S, Lim SC, Tai ES, Teo AKK
Nature communications 2021 May 25;12(1):3133
Nature communications 2021 May 25;12(1):3133
TFG binds LC3C to regulate ULK1 localization and autophagosome formation.
Carinci M, Testa B, Bordi M, Milletti G, Bonora M, Antonucci L, Ferraina C, Carro M, Kumar M, Ceglie D, Eck F, Nardacci R, le Guerroué F, Petrini S, Soriano ME, Caruana I, Doria V, Manifava M, Peron C, Lambrughi M, Tiranti V, Behrends C, Papaleo E, Pinton P, Giorgi C, Ktistakis NT, Locatelli F, Nazio F, Cecconi F
The EMBO journal 2021 May 17;40(10):e103563
The EMBO journal 2021 May 17;40(10):e103563
Hijacking of the host cell Golgi by Plasmodium berghei liver stage parasites.
De Niz M, Caldelari R, Kaiser G, Zuber B, Heo WD, Heussler VT, Agop-Nersesian C
Journal of cell science 2021 May 15;134(10)
Journal of cell science 2021 May 15;134(10)
Iron control of erythroid microtubule cytoskeleton as a potential target in treatment of iron-restricted anemia.
Goldfarb AN, Freeman KC, Sahu RK, Elagib KE, Holy M, Arneja A, Polanowska-Grabowska R, Gru AA, White Z 3rd, Khalil S, Kerins MJ, Ooi A, Leitinger N, Luckey CJ, Delehanty LL
Nature communications 2021 Mar 12;12(1):1645
Nature communications 2021 Mar 12;12(1):1645
Sterol-resistant SCAP Overexpression in Vascular Smooth Muscle Cells Accelerates Atherosclerosis by Increasing Local Vascular Inflammation through Activation of the NLRP3 Inflammasome in Mice.
Li D, Liu M, Li Z, Zheng G, Chen A, Zhao L, Yang P, Wei L, Chen Y, Ruan XZ
Aging and disease 2021 Jun;12(3):747-763
Aging and disease 2021 Jun;12(3):747-763
Rapid degradation of GRASP55 and GRASP65 reveals their immediate impact on the Golgi structure.
Zhang Y, Seemann J
The Journal of cell biology 2021 Jan 4;220(1)
The Journal of cell biology 2021 Jan 4;220(1)
Diacylglycerol kinase and phospholipase D inhibitors alter the cellular lipidome and endosomal sorting towards the Golgi apparatus.
Lingelem ABD, Kavaliauskiene S, Halsne R, Klokk TI, Surma MA, Klose C, Skotland T, Sandvig K
Cellular and molecular life sciences : CMLS 2021 Feb;78(3):985-1009
Cellular and molecular life sciences : CMLS 2021 Feb;78(3):985-1009
TMEM98 is a negative regulator of FRAT mediated Wnt/ß-catenin signalling.
van der Wal T, Lambooij JP, van Amerongen R
PloS one 2020;15(1):e0227435
PloS one 2020;15(1):e0227435
The golgin family exhibits a propensity to form condensates in living cells.
Ziltener P, Rebane AA, Graham M, Ernst AM, Rothman JE
FEBS letters 2020 Oct;594(19):3086-3094
FEBS letters 2020 Oct;594(19):3086-3094
Evaluation of active Rac1 levels in cancer cells: A case of misleading conclusions from immunofluorescence analysis.
Baker MJ, Cooke M, Kreider-Letterman G, Garcia-Mata R, Janmey PA, Kazanietz MG
The Journal of biological chemistry 2020 Oct 2;295(40):13698-13710
The Journal of biological chemistry 2020 Oct 2;295(40):13698-13710
Chromogranin A preferential interaction with Golgi phosphatidic acid induces membrane deformation and contributes to secretory granule biogenesis.
Carmon O, Laguerre F, Riachy L, Delestre-Delacour C, Wang Q, Tanguy E, Jeandel L, Cartier D, Thahouly T, Haeberlé AM, Fouillen L, Rezazgui O, Schapman D, Haefelé A, Goumon Y, Galas L, Renard PY, Alexandre S, Vitale N, Anouar Y, Montero-Hadjadje M
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 May;34(5):6769-6790
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 May;34(5):6769-6790
G protein-regulated endocytic trafficking of adenylyl cyclase type 9.
Lazar AM, Irannejad R, Baldwin TA, Sundaram AB, Gutkind JS, Inoue A, Dessauer CW, Von Zastrow M
eLife 2020 Jun 9;9
eLife 2020 Jun 9;9
The GTPase Rab27b regulates the release, autophagic clearance, and toxicity of α-synuclein.
Underwood R, Wang B, Carico C, Whitaker RH, Placzek WJ, Yacoubian TA
The Journal of biological chemistry 2020 Jun 5;295(23):8005-8016
The Journal of biological chemistry 2020 Jun 5;295(23):8005-8016
Human mastadenovirus-B (HAdV-B)-specific E3-CR1β and E3-CR1γ glycoproteins interact with each other and localize at the plasma membrane of non-polarized airway epithelial cells.
Lakshmi Narayan PK, Kajon AE
Virology 2020 Jul;546:67-78
Virology 2020 Jul;546:67-78
Retromer stabilization results in neuroprotection in a model of Amyotrophic Lateral Sclerosis.
Muzio L, Sirtori R, Gornati D, Eleuteri S, Fossaghi A, Brancaccio D, Manzoni L, Ottoboni L, Feo L, Quattrini A, Mastrangelo E, Sorrentino L, Scalone E, Comi G, Marinelli L, Riva N, Milani M, Seneci P, Martino G
Nature communications 2020 Jul 31;11(1):3848
Nature communications 2020 Jul 31;11(1):3848
TMEM16K is an interorganelle regulator of endosomal sorting.
Petkovic M, Oses-Prieto J, Burlingame A, Jan LY, Jan YN
Nature communications 2020 Jul 3;11(1):3298
Nature communications 2020 Jul 3;11(1):3298
Phenome-based approach identifies RIC1-linked Mendelian syndrome through zebrafish models, biobank associations and clinical studies.
Unlu G, Qi X, Gamazon ER, Melville DB, Patel N, Rushing AR, Hashem M, Al-Faifi A, Chen R, Li B, Cox NJ, Alkuraya FS, Knapik EW
Nature medicine 2020 Jan;26(1):98-109
Nature medicine 2020 Jan;26(1):98-109
Distinct anterograde trafficking pathways of BACE1 and amyloid precursor protein from the TGN and the regulation of amyloid-β production.
Tan JZA, Fourriere L, Wang J, Perez F, Boncompain G, Gleeson PA
Molecular biology of the cell 2020 Jan 1;31(1):27-44
Molecular biology of the cell 2020 Jan 1;31(1):27-44
Acute inactivation of retromer and ESCPE-1 leads to time-resolved defects in endosomal cargo sorting.
Evans AJ, Daly JL, Anuar ANK, Simonetti B, Cullen PJ
Journal of cell science 2020 Aug 3;133(15)
Journal of cell science 2020 Aug 3;133(15)
Caveolin-1 suppresses tumor formation through the inhibition of the unfolded protein response.
Díaz MI, Díaz P, Bennett JC, Urra H, Ortiz R, Orellana PC, Hetz C, Quest AFG
Cell death & disease 2020 Aug 3;11(8):648
Cell death & disease 2020 Aug 3;11(8):648
Visualization of Protein Sorting at the Trans-Golgi Network and Endosomes Through Super-Resolution Imaging.
Huang Y, Ma T, Lau PK, Wang J, Zhao T, Du S, Loy MMT, Guo Y
Frontiers in cell and developmental biology 2019;7:181
Frontiers in cell and developmental biology 2019;7:181
Restoration of F508-del Function by Transcomplementation: The Partners Meet in the Endoplasmic Reticulum.
Bergbower EAS, Sabirzhanova I, Boinot C, Guggino WB, Cebotaru L
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2019;52(6):1267-1279
Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 2019;52(6):1267-1279
A Novel Role for Triglyceride Metabolism in Foxp3 Expression.
Howie D, Ten Bokum A, Cobbold SP, Yu Z, Kessler BM, Waldmann H
Frontiers in immunology 2019;10:1860
Frontiers in immunology 2019;10:1860
Effects of Stuffer DNA on the Suppression of Choroidal Neovascularization by a rAAV Expressing a mTOR-Inhibiting shRNA.
Lee SHS, Chang H, Kim HJ, Choi JS, Kim J, Kim JH, Woo HN, Nah SK, Jung SJ, Lee JY, Park K, Park TK, Lee H
Molecular therapy. Methods & clinical development 2019 Sep 13;14:171-179
Molecular therapy. Methods & clinical development 2019 Sep 13;14:171-179
ARFRP1 functions upstream of ARL1 and ARL5 to coordinate recruitment of distinct tethering factors to the trans-Golgi network.
Ishida M, Bonifacino JS
The Journal of cell biology 2019 Nov 4;218(11):3681-3696
The Journal of cell biology 2019 Nov 4;218(11):3681-3696
Effects of Long-Term Citrate Treatment in the PC3 Prostate Cancer Cell Line.
Caiazza C, D'Agostino M, Passaro F, Faicchia D, Mallardo M, Paladino S, Pierantoni GM, Tramontano D
International journal of molecular sciences 2019 May 28;20(11)
International journal of molecular sciences 2019 May 28;20(11)
FANCM limits ALT activity by restricting telomeric replication stress induced by deregulated BLM and R-loops.
Silva B, Pentz R, Figueira AM, Arora R, Lee YW, Hodson C, Wischnewski H, Deans AJ, Azzalin CM
Nature communications 2019 May 28;10(1):2253
Nature communications 2019 May 28;10(1):2253
Overexpression-mediated activation of MET in the Golgi promotes HER3/ERBB3 phosphorylation.
Frazier NM, Brand T, Gordan JD, Grandis J, Jura N
Oncogene 2019 Mar;38(11):1936-1950
Oncogene 2019 Mar;38(11):1936-1950
Biosynthesis of O-N-acetylgalactosamine glycans in the human cell nucleus.
Cejas RB, Lorenz V, Garay YC, Irazoqui FJ
The Journal of biological chemistry 2019 Mar 1;294(9):2997-3011
The Journal of biological chemistry 2019 Mar 1;294(9):2997-3011
HNF4A Haploinsufficiency in MODY1 Abrogates Liver and Pancreas Differentiation from Patient-Derived Induced Pluripotent Stem Cells.
Ng NHJ, Jasmen JB, Lim CS, Lau HH, Krishnan VG, Kadiwala J, Kulkarni RN, Ræder H, Vallier L, Hoon S, Teo AKK
iScience 2019 Jun 28;16:192-205
iScience 2019 Jun 28;16:192-205
Correction: Regulation of E-cadherin localization by microtubule targeting agents: rapid promotion of cortical E-cadherin through p130CAS/Src inhibition by eribulin.
Dybdal-Hargreaves NF, Risinger AL, Mooberry SL
Oncotarget 2019 Jul 23;10(45):4719-4720
Oncotarget 2019 Jul 23;10(45):4719-4720
Biallelic VARS variants cause developmental encephalopathy with microcephaly that is recapitulated in vars knockout zebrafish.
Siekierska A, Stamberger H, Deconinck T, Oprescu SN, Partoens M, Zhang Y, Sourbron J, Adriaenssens E, Mullen P, Wiencek P, Hardies K, Lee JS, Giong HK, Distelmaier F, Elpeleg O, Helbig KL, Hersh J, Isikay S, Jordan E, Karaca E, Kecskes A, Lupski JR, Kovacs-Nagy R, May P, Narayanan V, Pendziwiat M, Ramsey K, Rangasamy S, Shinde DN, Spiegel R, Timmerman V, von Spiczak S, Helbig I, C4RCD Research Group., AR working group of the EuroEPINOMICS RES Consortium., Weckhuysen S, Francklyn C, Antonellis A, de Witte P, De Jonghe P
Nature communications 2019 Feb 12;10(1):708
Nature communications 2019 Feb 12;10(1):708
SNAREs define targeting specificity of trafficking vesicles by combinatorial interaction with tethering factors.
Koike S, Jahn R
Nature communications 2019 Apr 8;10(1):1608
Nature communications 2019 Apr 8;10(1):1608
Golgin-97 Targets Ectopically Expressed Inward Rectifying Potassium Channel, Kir2.1, to the trans-Golgi Network in COS-7 Cells.
Taneja TK, Ma D, Kim BY, Welling PA
Frontiers in physiology 2018;9:1070
Frontiers in physiology 2018;9:1070
Reinterpretation of the localization of the ATP binding cassette transporter ABCG1 in insulin-secreting cells and insights regarding its trafficking and function.
Harris MT, Hussain SS, Inouye CM, Castle AM, Castle JD
PloS one 2018;13(9):e0198383
PloS one 2018;13(9):e0198383
PAQR3 Regulates Endoplasmic Reticulum-to-Golgi Trafficking of COPII Vesicle via Interaction with Sec13/Sec31 Coat Proteins.
Cao Q, Wang Z, Wan H, Xu L, You X, Liao L, Chen Y
iScience 2018 Nov 30;9:382-398
iScience 2018 Nov 30;9:382-398
Intravitreal Injection of AAV Expressing Soluble VEGF Receptor-1 Variant Induces Anti-VEGF Activity and Suppresses Choroidal Neovascularization.
Lee SHS, Kim HJ, Shin OK, Choi JS, Kim J, Cho YH, Ha J, Park TK, Lee JY, Park K, Lee H
Investigative ophthalmology & visual science 2018 Nov 1;59(13):5398-5407
Investigative ophthalmology & visual science 2018 Nov 1;59(13):5398-5407
Endoplasmic reticulum, Golgi, and lysosomes are disorganized in lung fibroblasts from chronic obstructive pulmonary disease patients.
Weidner J, Jarenbäck L, Åberg I, Westergren-Thorsson G, Ankerst J, Bjermer L, Tufvesson E
Physiological reports 2018 Mar;6(5)
Physiological reports 2018 Mar;6(5)
Upon Infection, Cellular WD Repeat-Containing Protein 5 (WDR5) Localizes to Cytoplasmic Inclusion Bodies and Enhances Measles Virus Replication.
Ma D, George CX, Nomburg JL, Pfaller CK, Cattaneo R, Samuel CE
Journal of virology 2018 Mar 1;92(5)
Journal of virology 2018 Mar 1;92(5)
ArfGAP1 restricts Mycobacterium tuberculosis entry by controlling the actin cytoskeleton.
Song OR, Queval CJ, Iantomasi R, Delorme V, Marion S, Veyron-Churlet R, Werkmeister E, Popoff M, Ricard I, Jouny S, Deboosere N, Lafont F, Baulard A, Yeramian E, Marsollier L, Hoffmann E, Brodin P
EMBO reports 2018 Jan;19(1):29-42
EMBO reports 2018 Jan;19(1):29-42
Parkinson disease-associated mutations in LRRK2 cause centrosomal defects via Rab8a phosphorylation.
Madero-Pérez J, Fdez E, Fernández B, Lara Ordóñez AJ, Blanca Ramírez M, Gómez-Suaga P, Waschbüsch D, Lobbestael E, Baekelandt V, Nairn AC, Ruiz-Martínez J, Aiastui A, López de Munain A, Lis P, Comptdaer T, Taymans JM, Chartier-Harlin MC, Beilina A, Gonnelli A, Cookson MR, Greggio E, Hilfiker S
Molecular neurodegeneration 2018 Jan 23;13(1):3
Molecular neurodegeneration 2018 Jan 23;13(1):3
Amnionless-mediated glycosylation is crucial for cell surface targeting of cubilin in renal and intestinal cells.
Udagawa T, Harita Y, Miura K, Mitsui J, Ode KL, Morishita S, Urae S, Kanda S, Kajiho Y, Tsurumi H, Ueda HR, Tsuji S, Saito A, Oka A
Scientific reports 2018 Feb 5;8(1):2351
Scientific reports 2018 Feb 5;8(1):2351
Dynamic changes in copper homeostasis and post-transcriptional regulation of Atp7a during myogenic differentiation.
Vest KE, Paskavitz AL, Lee JB, Padilla-Benavides T
Metallomics : integrated biometal science 2018 Feb 21;10(2):309-322
Metallomics : integrated biometal science 2018 Feb 21;10(2):309-322
Organelle membrane derived patches: reshaping classical methods for new targets.
Shapovalov G, Ritaine A, Bidaux G, Slomianny C, Borowiec AS, Gordienko D, Bultynck G, Skryma R, Prevarskaya N
Scientific reports 2017 Oct 26;7(1):14082
Scientific reports 2017 Oct 26;7(1):14082
High levels of the AR-V7 Splice Variant and Co-Amplification of the Golgi Protein Coding YIPF6 in AR Amplified Prostate Cancer Bone Metastases.
Djusberg E, Jernberg E, Thysell E, Golovleva I, Lundberg P, Crnalic S, Widmark A, Bergh A, Brattsand M, Wikström P
The Prostate 2017 May;77(6):625-638
The Prostate 2017 May;77(6):625-638
Rab33B Controls Hepatitis B Virus Assembly by Regulating Core Membrane Association and Nucleocapsid Processing.
Bartusch C, Döring T, Prange R
Viruses 2017 Jun 21;9(6)
Viruses 2017 Jun 21;9(6)
Ablation of SNX6 leads to defects in synaptic function of CA1 pyramidal neurons and spatial memory.
Niu Y, Dai Z, Liu W, Zhang C, Yang Y, Guo Z, Li X, Xu C, Huang X, Wang Y, Shi YS, Liu JJ
eLife 2017 Jan 30;6
eLife 2017 Jan 30;6
Ror2 signaling regulates Golgi structure and transport through IFT20 for tumor invasiveness.
Nishita M, Park SY, Nishio T, Kamizaki K, Wang Z, Tamada K, Takumi T, Hashimoto R, Otani H, Pazour GJ, Hsu VW, Minami Y
Scientific reports 2017 Jan 26;7(1):1
Scientific reports 2017 Jan 26;7(1):1
COPI-TRAPPII activates Rab18 and regulates its lipid droplet association.
Li C, Luo X, Zhao S, Siu GK, Liang Y, Chan HC, Satoh A, Yu SS
The EMBO journal 2017 Feb 15;36(4):441-457
The EMBO journal 2017 Feb 15;36(4):441-457
Human cactin interacts with DHX8 and SRRM2 to assure efficient pre-mRNA splicing and sister chromatid cohesion.
Zanini IM, Soneson C, Lorenzi LE, Azzalin CM
Journal of cell science 2017 Feb 15;130(4):767-778
Journal of cell science 2017 Feb 15;130(4):767-778
Inhibitors of GLUT/SLC2A Enhance the Action of BCNU and Temozolomide against High-Grade Gliomas.
Azzalin A, Nato G, Parmigiani E, Garello F, Buffo A, Magrassi L
Neoplasia (New York, N.Y.) 2017 Apr;19(4):364-373
Neoplasia (New York, N.Y.) 2017 Apr;19(4):364-373
Lysosomal putative RNA transporter SIDT2 mediates direct uptake of RNA by lysosomes.
Aizawa S, Fujiwara Y, Contu VR, Hase K, Takahashi M, Kikuchi H, Kabuta C, Wada K, Kabuta T
Autophagy 2016;12(3):565-78
Autophagy 2016;12(3):565-78
Two-Step Mechanism of Cellular Uptake of Cationic Gold Nanoparticles Modified by (16-Mercaptohexadecyl)trimethylammonium Bromide.
Zarska M, Novotny F, Havel F, Sramek M, Babelova A, Benada O, Novotny M, Saran H, Kuca K, Musilek K, Hvezdova Z, Dzijak R, Vancurova M, Krejcikova K, Gabajova B, Hanzlikova H, Kyjacova L, Bartek J, Proska J, Hodny Z
Bioconjugate chemistry 2016 Oct 19;27(10):2558-2574
Bioconjugate chemistry 2016 Oct 19;27(10):2558-2574
Palmitoylation of Desmoglein 2 Is a Regulator of Assembly Dynamics and Protein Turnover.
Roberts BJ, Svoboda RA, Overmiller AM, Lewis JD, Kowalczyk AP, Mahoney MG, Johnson KR, Wahl JK 3rd
The Journal of biological chemistry 2016 Nov 25;291(48):24857-24865
The Journal of biological chemistry 2016 Nov 25;291(48):24857-24865
Aurora-A recruitment and centrosomal maturation are regulated by a Golgi-activated pool of Src during G2.
Barretta ML, Spano D, D'Ambrosio C, Cervigni RI, Scaloni A, Corda D, Colanzi A
Nature communications 2016 May 31;7:11727
Nature communications 2016 May 31;7:11727
Excess sphingomyelin disturbs ATG9A trafficking and autophagosome closure.
Corcelle-Termeau E, Vindeløv SD, Hämälistö S, Mograbi B, Keldsbo A, Bräsen JH, Favaro E, Adam D, Szyniarowski P, Hofman P, Krautwald S, Farkas T, Petersen NH, Rohde M, Linkermann A, Jäättelä M
Autophagy 2016 May 3;12(5):833-49
Autophagy 2016 May 3;12(5):833-49
Disruption of Golgi morphology and altered protein glycosylation in PLA2G6-associated neurodegeneration.
Davids M, Kane MS, He M, Wolfe LA, Li X, Raihan MA, Chao KR, Bone WP, Boerkoel CF, Gahl WA, Toro C
Journal of medical genetics 2016 Mar;53(3):180-9
Journal of medical genetics 2016 Mar;53(3):180-9
The Endoplasmic Reticulum Chaperone Calnexin Is a NADPH Oxidase NOX4 Interacting Protein.
Prior KK, Wittig I, Leisegang MS, Groenendyk J, Weissmann N, Michalak M, Jansen-Dürr P, Shah AM, Brandes RP
The Journal of biological chemistry 2016 Mar 25;291(13):7045-59
The Journal of biological chemistry 2016 Mar 25;291(13):7045-59
A large Rab GTPase encoded by CRACR2A is a component of subsynaptic vesicles that transmit T cell activation signals.
Srikanth S, Kim KD, Gao Y, Woo JS, Ghosh S, Calmettes G, Paz A, Abramson J, Jiang M, Gwack Y
Science signaling 2016 Mar 22;9(420):ra31
Science signaling 2016 Mar 22;9(420):ra31
Golgi membrane fission requires the CtBP1-S/BARS-induced activation of lysophosphatidic acid acyltransferase δ.
Pagliuso A, Valente C, Giordano LL, Filograna A, Li G, Circolo D, Turacchio G, Marzullo VM, Mandrich L, Zhukovsky MA, Formiggini F, Polishchuk RS, Corda D, Luini A
Nature communications 2016 Jul 12;7:12148
Nature communications 2016 Jul 12;7:12148
Protein Kinase D2 Assembles a Multiprotein Complex at the Trans-Golgi Network to Regulate Matrix Metalloproteinase Secretion.
Eiseler T, Wille C, Koehler C, Illing A, Seufferlein T
The Journal of biological chemistry 2016 Jan 1;291(1):462-77
The Journal of biological chemistry 2016 Jan 1;291(1):462-77
Caveolin-1 is an aggresome-inducing protein.
Tiwari A, Copeland CA, Han B, Hanson CA, Raghunathan K, Kenworthy AK
Scientific reports 2016 Dec 8;6:38681
Scientific reports 2016 Dec 8;6:38681
The CD63-Syntenin-1 Complex Controls Post-Endocytic Trafficking of Oncogenic Human Papillomaviruses.
Gräßel L, Fast LA, Scheffer KD, Boukhallouk F, Spoden GA, Tenzer S, Boller K, Bago R, Rajesh S, Overduin M, Berditchevski F, Florin L
Scientific reports 2016 Aug 31;6:32337
Scientific reports 2016 Aug 31;6:32337
Simplified protocol for flow cytometry analysis of fluorescently labeled exosomes and microvesicles using dedicated flow cytometer.
Pospichalova V, Svoboda J, Dave Z, Kotrbova A, Kaiser K, Klemova D, Ilkovics L, Hampl A, Crha I, Jandakova E, Minar L, Weinberger V, Bryja V
Journal of extracellular vesicles 2015;4:25530
Journal of extracellular vesicles 2015;4:25530
Simplified protocol for flow cytometry analysis of fluorescently labeled exosomes and microvesicles using dedicated flow cytometer.
Pospichalova V, Svoboda J, Dave Z, Kotrbova A, Kaiser K, Klemova D, Ilkovics L, Hampl A, Crha I, Jandakova E, Minar L, Weinberger V, Bryja V
Journal of extracellular vesicles 2015;4:25530
Journal of extracellular vesicles 2015;4:25530
Sensitive detection of lysosomal membrane permeabilization by lysosomal galectin puncta assay.
Aits S, Kricker J, Liu B, Ellegaard AM, Hämälistö S, Tvingsholm S, Corcelle-Termeau E, Høgh S, Farkas T, Holm Jonassen A, Gromova I, Mortensen M, Jäättelä M
Autophagy 2015;11(8):1408-24
Autophagy 2015;11(8):1408-24
Post-Golgi anterograde transport requires GARP-dependent endosome-to-TGN retrograde transport.
Hirata T, Fujita M, Nakamura S, Gotoh K, Motooka D, Murakami Y, Maeda Y, Kinoshita T
Molecular biology of the cell 2015 Sep 1;26(17):3071-84
Molecular biology of the cell 2015 Sep 1;26(17):3071-84
IGF-1 drives chromogranin A secretion via activation of Arf1 in human neuroendocrine tumour cells.
Münzberg C, Höhn K, Krndija D, Maaß U, Bartsch DK, Slater EP, Oswald F, Walther P, Seufferlein T, von Wichert G
Journal of cellular and molecular medicine 2015 May;19(5):948-59
Journal of cellular and molecular medicine 2015 May;19(5):948-59
Memory and PTPIP51--a new protein in hippocampus and cerebellum.
Brobeil A, Viard M, Petri MK, Steger K, Tag C, Wimmer M
Molecular and cellular neurosciences 2015 Jan;64:61-73
Molecular and cellular neurosciences 2015 Jan;64:61-73
Beclin 1 regulates neuronal transforming growth factor-β signaling by mediating recycling of the type I receptor ALK5.
O'Brien CE, Bonanno L, Zhang H, Wyss-Coray T
Molecular neurodegeneration 2015 Dec 21;10:69
Molecular neurodegeneration 2015 Dec 21;10:69
Auxiliary KCNE subunits modulate both homotetrameric Kv2.1 and heterotetrameric Kv2.1/Kv6.4 channels.
David JP, Stas JI, Schmitt N, Bocksteins E
Scientific reports 2015 Aug 5;5:12813
Scientific reports 2015 Aug 5;5:12813
RC/BTB2 is essential for formation of primary cilia in mammalian cells.
Zhang L, Li W, Ni J, Wu J, Liu J, Zhang Z, Zhang Y, Li H, Shi Y, Teves ME, Song S, Strauss JF 3rd, Zhang Z
Cytoskeleton (Hoboken, N.J.) 2015 Apr;72(4):171-81
Cytoskeleton (Hoboken, N.J.) 2015 Apr;72(4):171-81
HSP47 and FKBP65 cooperate in the synthesis of type I procollagen.
Duran I, Nevarez L, Sarukhanov A, Wu S, Lee K, Krejci P, Weis M, Eyre D, Krakow D, Cohn DH
Human molecular genetics 2015 Apr 1;24(7):1918-28
Human molecular genetics 2015 Apr 1;24(7):1918-28
Characterization of Rabaptin-5 γ isoform.
Korobko EV, Kiselev SL, Korobko IV
Biochemistry. Biokhimiia 2014 Sep;79(9):856-64
Biochemistry. Biokhimiia 2014 Sep;79(9):856-64
CCHCR1 interacts with EDC4, suggesting its localization in P-bodies.
Ling YH, Wong CC, Li KW, Chan KM, Boukamp P, Liu WK
Experimental cell research 2014 Sep 10;327(1):12-23
Experimental cell research 2014 Sep 10;327(1):12-23
Nile Tilapia Neu3 sialidases: molecular cloning, functional characterization and expression in Oreochromis niloticus.
Chigwechokha PK, Komatsu M, Itakura T, Shiozaki K
Gene 2014 Nov 15;552(1):155-64
Gene 2014 Nov 15;552(1):155-64
Mutation in VPS35 associated with Parkinson's disease impairs WASH complex association and inhibits autophagy.
Zavodszky E, Seaman MN, Moreau K, Jimenez-Sanchez M, Breusegem SY, Harbour ME, Rubinsztein DC
Nature communications 2014 May 13;5:3828
Nature communications 2014 May 13;5:3828
LAPTM5 promotes lysosomal degradation of intracellular CD3ζ but not of cell surface CD3ζ.
Kawai Y, Ouchida R, Yamasaki S, Dragone L, Tsubata T, Wang JY
Immunology and cell biology 2014 Jul;92(6):527-34
Immunology and cell biology 2014 Jul;92(6):527-34
Arf4 is required for Mammalian development but dispensable for ciliary assembly.
Follit JA, San Agustin JT, Jonassen JA, Huang T, Rivera-Perez JA, Tremblay KD, Pazour GJ
PLoS genetics 2014 Feb;10(2):e1004170
PLoS genetics 2014 Feb;10(2):e1004170
An L2 SUMO interacting motif is important for PML localization and infection of human papillomavirus type 16.
Bund T, Spoden GA, Koynov K, Hellmann N, Boukhallouk F, Arnold P, Hinderberger D, Florin L
Cellular microbiology 2014 Aug;16(8):1179-200
Cellular microbiology 2014 Aug;16(8):1179-200
Alternative transcripts and 3'UTR elements govern the incorporation of selenocysteine into selenoprotein S.
Bubenik JL, Miniard AC, Driscoll DM
PloS one 2013;8(4):e62102
PloS one 2013;8(4):e62102
A novel domain regulating degradation of the glomerular slit diaphragm protein podocin in cell culture systems.
Gödel M, Ostendorf BN, Baumer J, Weber K, Huber TB
PloS one 2013;8(2):e57078
PloS one 2013;8(2):e57078
Intracellular itinerary of internalised β-secretase, BACE1, and its potential impact on β-amyloid peptide biogenesis.
Chia PZ, Toh WH, Sharples R, Gasnereau I, Hill AF, Gleeson PA
Traffic (Copenhagen, Denmark) 2013 Sep;14(9):997-1013
Traffic (Copenhagen, Denmark) 2013 Sep;14(9):997-1013
Partial IFN-γR2 deficiency is due to protein misfolding and can be rescued by inhibitors of glycosylation.
Moncada-Vélez M, Martinez-Barricarte R, Bogunovic D, Kong XF, Blancas-Galicia L, Tirpan C, Aksu G, Vincent QB, Boisson B, Itan Y, Ramírez-Alejo N, Okada S, Kreins AY, Bryant VL, Franco JL, Migaud M, Espinosa-Padilla S, Yamazaki-Nakashimada M, Espinosa-Rosales F, Kutukculer N, Abel L, Bustamante J, Vogt G, Casanova JL, Boisson-Dupuis S
Blood 2013 Oct 3;122(14):2390-401
Blood 2013 Oct 3;122(14):2390-401
Expression of conventional and novel glucose transporters, GLUT1, -9, -10, and -12, in vascular smooth muscle cells.
Pyla R, Poulose N, Jun JY, Segar L
American journal of physiology. Cell physiology 2013 Mar;304(6):C574-89
American journal of physiology. Cell physiology 2013 Mar;304(6):C574-89
Involvement of lysosomal exocytosis in the excretion of mesoporous silica nanoparticles and enhancement of the drug delivery effect by exocytosis inhibition.
Yanes RE, Tarn D, Hwang AA, Ferris DP, Sherman SP, Thomas CR, Lu J, Pyle AD, Zink JI, Tamanoi F
Small (Weinheim an der Bergstrasse, Germany) 2013 Mar 11;9(5):697-704
Small (Weinheim an der Bergstrasse, Germany) 2013 Mar 11;9(5):697-704
Trafficking of the Menkes copper transporter ATP7A is regulated by clathrin-, AP-2-, AP-1-, and Rab22-dependent steps.
Holloway ZG, Velayos-Baeza A, Howell GJ, Levecque C, Ponnambalam S, Sztul E, Monaco AP
Molecular biology of the cell 2013 Jun;24(11):1735-48, S1-8
Molecular biology of the cell 2013 Jun;24(11):1735-48, S1-8
Versican is upregulated in circulating monocytes in patients with systemic sclerosis and amplifies a CCL2-mediated pathogenic loop.
Masuda A, Yasuoka H, Satoh T, Okazaki Y, Yamaguchi Y, Kuwana M
Arthritis research & therapy 2013 Jul 11;15(4):R74
Arthritis research & therapy 2013 Jul 11;15(4):R74
Molecular cloning and biochemical characterization of two novel Neu3 sialidases, neu3a and neu3b, from medaka (Oryzias latipes).
Shiozaki K, Takeshita K, Ikeda M, Ikeda A, Harasaki Y, Komatsu M, Yamada S, Yamaguchi K, Miyagi T
Biochimie 2013 Feb;95(2):280-9
Biochimie 2013 Feb;95(2):280-9
Regulation of WASH-dependent actin polymerization and protein trafficking by ubiquitination.
Hao YH, Doyle JM, Ramanathan S, Gomez TS, Jia D, Xu M, Chen ZJ, Billadeau DD, Rosen MK, Potts PR
Cell 2013 Feb 28;152(5):1051-64
Cell 2013 Feb 28;152(5):1051-64
Regulation of cathepsins S and L by cystatin F during maturation of dendritic cells.
Magister S, Obermajer N, Mirković B, Svajger U, Renko M, Softić A, Romih R, Colbert JD, Watts C, Kos J
European journal of cell biology 2012 May;91(5):391-401
European journal of cell biology 2012 May;91(5):391-401
Chemical structure requirements and cellular targeting of microRNA-122 by peptide nucleic acids anti-miRs.
Torres AG, Fabani MM, Vigorito E, Williams D, Al-Obaidi N, Wojciechowski F, Hudson RH, Seitz O, Gait MJ
Nucleic acids research 2012 Mar;40(5):2152-67
Nucleic acids research 2012 Mar;40(5):2152-67
Spatiotemporal behavior of nuclear cyclophilin B indicates a role in RNA transcription.
Dieriks B, Van Oostveldt P
International journal of molecular medicine 2012 Jun;29(6):1031-8
International journal of molecular medicine 2012 Jun;29(6):1031-8
Amyloid precursor protein (APP) traffics from the cell surface via endosomes for amyloid β (Aβ) production in the trans-Golgi network.
Choy RW, Cheng Z, Schekman R
Proceedings of the National Academy of Sciences of the United States of America 2012 Jul 24;109(30):E2077-82
Proceedings of the National Academy of Sciences of the United States of America 2012 Jul 24;109(30):E2077-82
Differential regulation of myosin X movements by its cargos, DCC and neogenin.
Liu Y, Peng Y, Dai PG, Du QS, Mei L, Xiong WC
Journal of cell science 2012 Feb 1;125(Pt 3):751-62
Journal of cell science 2012 Feb 1;125(Pt 3):751-62
An optimized isolation of biotinylated cell surface proteins reveals novel players in cancer metastasis.
Karhemo PR, Ravela S, Laakso M, Ritamo I, Tatti O, Mäkinen S, Goodison S, Stenman UH, Hölttä E, Hautaniemi S, Valmu L, Lehti K, Laakkonen P
Journal of proteomics 2012 Dec 21;77:87-100
Journal of proteomics 2012 Dec 21;77:87-100
GM130 gain-of-function induces cell pathology in a model of lysosomal storage disease.
Roy E, Bruyère J, Flamant P, Bigou S, Ausseil J, Vitry S, Heard JM
Human molecular genetics 2012 Apr 1;21(7):1481-95
Human molecular genetics 2012 Apr 1;21(7):1481-95
Mre11 is expressed in mammalian mitochondria where it binds to mitochondrial DNA.
Dmitrieva NI, Malide D, Burg MB
American journal of physiology. Regulatory, integrative and comparative physiology 2011 Sep;301(3):R632-40
American journal of physiology. Regulatory, integrative and comparative physiology 2011 Sep;301(3):R632-40
Cytomegalovirus UL103 controls virion and dense body egress.
Ahlqvist J, Mocarski E
Journal of virology 2011 May;85(10):5125-35
Journal of virology 2011 May;85(10):5125-35
Down-regulation of oestrogen receptor-β associates with transcriptional co-regulator PATZ1 delocalization in human testicular seminomas.
Esposito F, Boscia F, Franco R, Tornincasa M, Fusco A, Kitazawa S, Looijenga LH, Chieffi P
The Journal of pathology 2011 May;224(1):110-20
The Journal of pathology 2011 May;224(1):110-20
Chemical chaperone therapy: chaperone effect on mutant enzyme and cellular pathophysiology in β-galactosidase deficiency.
Higaki K, Li L, Bahrudin U, Okuzawa S, Takamuram A, Yamamoto K, Adachi K, Paraguison RC, Takai T, Ikehata H, Tominaga L, Hisatome I, Iida M, Ogawa S, Matsuda J, Ninomiya H, Sakakibara Y, Ohno K, Suzuki Y, Nanba E
Human mutation 2011 Jul;32(7):843-52
Human mutation 2011 Jul;32(7):843-52
Dysfunction of autophagy participates in vacuole formation and cell death in cells replicating hepatitis C virus.
Taguwa S, Kambara H, Fujita N, Noda T, Yoshimori T, Koike K, Moriishi K, Matsuura Y
Journal of virology 2011 Dec;85(24):13185-94
Journal of virology 2011 Dec;85(24):13185-94
Splice variant-specific cellular function of the formin INF2 in maintenance of Golgi architecture.
Ramabhadran V, Korobova F, Rahme GJ, Higgs HN
Molecular biology of the cell 2011 Dec;22(24):4822-33
Molecular biology of the cell 2011 Dec;22(24):4822-33
A method to separate nuclear, cytosolic, and membrane-associated signaling molecules in cultured cells.
Liu X, Fagotto F
Science signaling 2011 Dec 13;4(203):pl2
Science signaling 2011 Dec 13;4(203):pl2
Tumor-associated calcium signal transducer 2 is required for the proper subcellular localization of claudin 1 and 7: implications in the pathogenesis of gelatinous drop-like corneal dystrophy.
Nakatsukasa M, Kawasaki S, Yamasaki K, Fukuoka H, Matsuda A, Tsujikawa M, Tanioka H, Nagata-Takaoka M, Hamuro J, Kinoshita S
The American journal of pathology 2010 Sep;177(3):1344-55
The American journal of pathology 2010 Sep;177(3):1344-55
PIST regulates the intracellular trafficking and plasma membrane expression of cadherin 23.
Xu Z, Oshima K, Heller S
BMC cell biology 2010 Oct 19;11:80
BMC cell biology 2010 Oct 19;11:80
Open reading frame E3-10.9K of subspecies B1 human adenoviruses encodes a family of late orthologous proteins that vary in their predicted structural features and subcellular localization.
Frietze KM, Campos SK, Kajon AE
Journal of virology 2010 Nov;84(21):11310-22
Journal of virology 2010 Nov;84(21):11310-22
Phospholipase D family member 4, a transmembrane glycoprotein with no phospholipase D activity, expression in spleen and early postnatal microglia.
Yoshikawa F, Banno Y, Otani Y, Yamaguchi Y, Nagakura-Takagi Y, Morita N, Sato Y, Saruta C, Nishibe H, Sadakata T, Shinoda Y, Hayashi K, Mishima Y, Baba H, Furuichi T
PloS one 2010 Nov 11;5(11):e13932
PloS one 2010 Nov 11;5(11):e13932
Tumor protein D52 expression and Ca2+-dependent phosphorylation modulates lysosomal membrane protein trafficking to the plasma membrane.
Thomas DD, Martin CL, Weng N, Byrne JA, Groblewski GE
American journal of physiology. Cell physiology 2010 Mar;298(3):C725-39
American journal of physiology. Cell physiology 2010 Mar;298(3):C725-39
HSV usurps eukaryotic initiation factor 3 subunit M for viral protein translation: novel prevention target.
Cheshenko N, Trepanier JB, Segarra TJ, Fuller AO, Herold BC
PloS one 2010 Jul 27;5(7):e11829
PloS one 2010 Jul 27;5(7):e11829
Geoditin A induces oxidative stress and apoptosis on human colon HT29 cells.
Cheung FW, Li C, Che CT, Liu BP, Wang L, Liu WK
Marine drugs 2010 Jan 19;8(1):80-90
Marine drugs 2010 Jan 19;8(1):80-90
The cytoplasmic tail of fibrocystin contains a ciliary targeting sequence.
Follit JA, Li L, Vucica Y, Pazour GJ
The Journal of cell biology 2010 Jan 11;188(1):21-8
The Journal of cell biology 2010 Jan 11;188(1):21-8
Storage vesicles in neurons are related to Golgi complex alterations in mucopolysaccharidosis IIIB.
Vitry S, Bruyère J, Hocquemiller M, Bigou S, Ausseil J, Colle MA, Prévost MC, Heard JM
The American journal of pathology 2010 Dec;177(6):2984-99
The American journal of pathology 2010 Dec;177(6):2984-99
LG186: An inhibitor of GBF1 function that causes Golgi disassembly in human and canine cells.
Boal F, Guetzoyan L, Sessions RB, Zeghouf M, Spooner RA, Lord JM, Cherfils J, Clarkson GJ, Roberts LM, Stephens DJ
Traffic (Copenhagen, Denmark) 2010 Dec;11(12):1537-51
Traffic (Copenhagen, Denmark) 2010 Dec;11(12):1537-51
Optimization of immunoprecipitation-western blot analysis in detecting GW182-associated components of GW/P bodies.
Moser JJ, Chan EK, Fritzler MJ
Nature protocols 2009;4(5):674-85
Nature protocols 2009;4(5):674-85
Hepatitis B virus and hepatitis C virus interaction in Huh-7 cells.
Eyre NS, Phillips RJ, Bowden S, Yip E, Dewar B, Locarnini SA, Beard MR
Journal of hepatology 2009 Sep;51(3):446-57
Journal of hepatology 2009 Sep;51(3):446-57
Akt and 14-3-3 control a PACS-2 homeostatic switch that integrates membrane traffic with TRAIL-induced apoptosis.
Aslan JE, You H, Williamson DM, Endig J, Youker RT, Thomas L, Shu H, Du Y, Milewski RL, Brush MH, Possemato A, Sprott K, Fu H, Greis KD, Runckel DN, Vogel A, Thomas G
Molecular cell 2009 May 14;34(4):497-509
Molecular cell 2009 May 14;34(4):497-509
Microenvironmental pH is a key factor for exosome traffic in tumor cells.
Parolini I, Federici C, Raggi C, Lugini L, Palleschi S, De Milito A, Coscia C, Iessi E, Logozzi M, Molinari A, Colone M, Tatti M, Sargiacomo M, Fais S
The Journal of biological chemistry 2009 Dec 4;284(49):34211-22
The Journal of biological chemistry 2009 Dec 4;284(49):34211-22
Primary ciliogenesis defects are associated with human astrocytoma/glioblastoma cells.
Moser JJ, Fritzler MJ, Rattner JB
BMC cancer 2009 Dec 17;9:448
BMC cancer 2009 Dec 17;9:448
Thioesterase activity and subcellular localization of acylprotein thioesterase 1/lysophospholipase 1.
Hirano T, Kishi M, Sugimoto H, Taguchi R, Obinata H, Ohshima N, Tatei K, Izumi T
Biochimica et biophysica acta 2009 Aug;1791(8):797-805
Biochimica et biophysica acta 2009 Aug;1791(8):797-805
Subcellular localization of Nox4 and regulation in diabetes.
Block K, Gorin Y, Abboud HE
Proceedings of the National Academy of Sciences of the United States of America 2009 Aug 25;106(34):14385-90
Proceedings of the National Academy of Sciences of the United States of America 2009 Aug 25;106(34):14385-90
IFAP syndrome is caused by deficiency in MBTPS2, an intramembrane zinc metalloprotease essential for cholesterol homeostasis and ER stress response.
Oeffner F, Fischer G, Happle R, König A, Betz RC, Bornholdt D, Neidel U, Boente Mdel C, Redler S, Romero-Gomez J, Salhi A, Vera-Casaño A, Weirich C, Grzeschik KH
American journal of human genetics 2009 Apr;84(4):459-67
American journal of human genetics 2009 Apr;84(4):459-67
A FRET-based fluorogenic phosphine for live-cell imaging with the Staudinger ligation.
Hangauer MJ, Bertozzi CR
Angewandte Chemie (International ed. in English) 2008;47(13):2394-7
Angewandte Chemie (International ed. in English) 2008;47(13):2394-7
Identification and characterization of an alternatively spliced variant of the MHC class I-related porcine neonatal Fc receptor for IgG.
Ye L, Tuo W, Liu X, Simister NE, Zhu X
Developmental and comparative immunology 2008;32(8):966-79
Developmental and comparative immunology 2008;32(8):966-79
Altered cellular distribution and sub-cellular sorting of doppel (Dpl) protein in human astrocytoma cell lines.
Sbalchiero E, Azzalin A, Palumbo S, Barbieri G, Arias A, Simonelli L, Ferretti L, Comincini S
Cellular oncology : the official journal of the International Society for Cellular Oncology 2008;30(4):337-47
Cellular oncology : the official journal of the International Society for Cellular Oncology 2008;30(4):337-47
Caveolin-1-dependent infectious entry of human papillomavirus type 31 in human keratinocytes proceeds to the endosomal pathway for pH-dependent uncoating.
Smith JL, Campos SK, Wandinger-Ness A, Ozbun MA
Journal of virology 2008 Oct;82(19):9505-12
Journal of virology 2008 Oct;82(19):9505-12
The cytoplasmic domain of proEGF negatively regulates motility and elastinolytic activity in thyroid carcinoma cells.
Glogowska A, Pyka J, Kehlen A, Los M, Perumal P, Weber E, Cheng SY, Hoang-Vu C, Klonisch T
Neoplasia (New York, N.Y.) 2008 Oct;10(10):1120-30
Neoplasia (New York, N.Y.) 2008 Oct;10(10):1120-30
A dynamic view of hepatitis C virus replication complexes.
Wölk B, Büchele B, Moradpour D, Rice CM
Journal of virology 2008 Nov;82(21):10519-31
Journal of virology 2008 Nov;82(21):10519-31
HtrA2/Omi terminates cytomegalovirus infection and is controlled by the viral mitochondrial inhibitor of apoptosis (vMIA).
McCormick AL, Roback L, Mocarski ES
PLoS pathogens 2008 May 9;4(5):e1000063
PLoS pathogens 2008 May 9;4(5):e1000063
Envelope protein palmitoylations are crucial for murine coronavirus assembly.
Boscarino JA, Logan HL, Lacny JJ, Gallagher TM
Journal of virology 2008 Mar;82(6):2989-99
Journal of virology 2008 Mar;82(6):2989-99
Clathrin-dependent trafficking of subtilase cytotoxin, a novel AB5 toxin that targets the endoplasmic reticulum chaperone BiP.
Chong DC, Paton JC, Thorpe CM, Paton AW
Cellular microbiology 2008 Mar;10(3):795-806
Cellular microbiology 2008 Mar;10(3):795-806
Cellular trafficking of lipoteichoic acid and Toll-like receptor 2 in relation to signaling: role of CD14 and CD36.
Nilsen NJ, Deininger S, Nonstad U, Skjeldal F, Husebye H, Rodionov D, von Aulock S, Hartung T, Lien E, Bakke O, Espevik T
Journal of leukocyte biology 2008 Jul;84(1):280-91
Journal of leukocyte biology 2008 Jul;84(1):280-91
Genome-wide and functional annotation of human E3 ubiquitin ligases identifies MULAN, a mitochondrial E3 that regulates the organelle's dynamics and signaling.
Li W, Bengtson MH, Ulbrich A, Matsuda A, Reddy VA, Orth A, Chanda SK, Batalov S, Joazeiro CA
PloS one 2008 Jan 23;3(1):e1487
PloS one 2008 Jan 23;3(1):e1487
The Golgin GMAP210/TRIP11 anchors IFT20 to the Golgi complex.
Follit JA, San Agustin JT, Xu F, Jonassen JA, Samtani R, Lo CW, Pazour GJ
PLoS genetics 2008 Dec;4(12):e1000315
PLoS genetics 2008 Dec;4(12):e1000315
A fluorimetry-based ssYFP secretion assay to monitor vasopressin-induced exocytosis in LLC-PK1 cells expressing aquaporin-2.
Nunes P, Hasler U, McKee M, Lu HA, Bouley R, Brown D
American journal of physiology. Cell physiology 2008 Dec;295(6):C1476-87
American journal of physiology. Cell physiology 2008 Dec;295(6):C1476-87
beta-dystrobrevin, a kinesin-binding receptor, interacts with the extracellular matrix components pancortins.
Veroni C, Grasso M, Macchia G, Ramoni C, Ceccarini M, Petrucci TC, Macioce P
Journal of neuroscience research 2007 Sep;85(12):2631-9
Journal of neuroscience research 2007 Sep;85(12):2631-9
Rab11-FIP2 regulates differentiable steps in transcytosis.
Ducharme NA, Williams JA, Oztan A, Apodaca G, Lapierre LA, Goldenring JR
American journal of physiology. Cell physiology 2007 Sep;293(3):C1059-72
American journal of physiology. Cell physiology 2007 Sep;293(3):C1059-72
Ankyrin-G is a molecular partner of E-cadherin in epithelial cells and early embryos.
Kizhatil K, Davis JQ, Davis L, Hoffman J, Hogan BL, Bennett V
The Journal of biological chemistry 2007 Sep 7;282(36):26552-61
The Journal of biological chemistry 2007 Sep 7;282(36):26552-61
Requirements for the localization of nesprin-3 at the nuclear envelope and its interaction with plectin.
Ketema M, Wilhelmsen K, Kuikman I, Janssen H, Hodzic D, Sonnenberg A
Journal of cell science 2007 Oct 1;120(Pt 19):3384-94
Journal of cell science 2007 Oct 1;120(Pt 19):3384-94
Human ClC-6 is a late endosomal glycoprotein that associates with detergent-resistant lipid domains.
Ignoul S, Simaels J, Hermans D, Annaert W, Eggermont J
PloS one 2007 May 30;2(5):e474
PloS one 2007 May 30;2(5):e474
Secretory cytotoxic granule maturation and exocytosis require the effector protein hMunc13-4.
Ménager MM, Ménasché G, Romao M, Knapnougel P, Ho CH, Garfa M, Raposo G, Feldmann J, Fischer A, de Saint Basile G
Nature immunology 2007 Mar;8(3):257-67
Nature immunology 2007 Mar;8(3):257-67
Missense mutations of dual oxidase 2 (DUOX2) implicated in congenital hypothyroidism have impaired trafficking in cells reconstituted with DUOX2 maturation factor.
Grasberger H, De Deken X, Miot F, Pohlenz J, Refetoff S
Molecular endocrinology (Baltimore, Md.) 2007 Jun;21(6):1408-21
Molecular endocrinology (Baltimore, Md.) 2007 Jun;21(6):1408-21
Redox-signaling transmitted in trans to neighboring cells by melanoma-derived TNF-containing exosomes.
Söderberg A, Barral AM, Söderström M, Sander B, Rosén A
Free radical biology & medicine 2007 Jul 1;43(1):90-9
Free radical biology & medicine 2007 Jul 1;43(1):90-9
NGEP, a prostate-specific plasma membrane protein that promotes the association of LNCaP cells.
Das S, Hahn Y, Nagata S, Willingham MC, Bera TK, Lee B, Pastan I
Cancer research 2007 Feb 15;67(4):1594-601
Cancer research 2007 Feb 15;67(4):1594-601
Multiple receptor interactions trigger release of membrane and intracellular calcium stores critical for herpes simplex virus entry.
Cheshenko N, Liu W, Satlin LM, Herold BC
Molecular biology of the cell 2007 Aug;18(8):3119-30
Molecular biology of the cell 2007 Aug;18(8):3119-30
Expression and release of soluble HLA-E is an immunoregulatory feature of endothelial cell activation.
Coupel S, Moreau A, Hamidou M, Horejsi V, Soulillou JP, Charreau B
Blood 2007 Apr 1;109(7):2806-14
Blood 2007 Apr 1;109(7):2806-14
The intraflagellar transport protein IFT20 is associated with the Golgi complex and is required for cilia assembly.
Follit JA, Tuft RA, Fogarty KE, Pazour GJ
Molecular biology of the cell 2006 Sep;17(9):3781-92
Molecular biology of the cell 2006 Sep;17(9):3781-92
Molecular characterization of pannexins in the lens.
Dvoriantchikova G, Ivanov D, Pestova A, Shestopalov V
Molecular vision 2006 Nov 17;12:1417-26
Molecular vision 2006 Nov 17;12:1417-26
Leukocyte adhesion deficiency II patients with a dual defect of the GDP-fucose transporter.
Helmus Y, Denecke J, Yakubenia S, Robinson P, Lühn K, Watson DL, McGrogan PJ, Vestweber D, Marquardt T, Wild MK
Blood 2006 May 15;107(10):3959-66
Blood 2006 May 15;107(10):3959-66
Ghrelin and the growth hormone secretagogue receptor constitute a novel autocrine pathway in astrocytoma motility.
Dixit VD, Weeraratna AT, Yang H, Bertak D, Cooper-Jenkins A, Riggins GJ, Eberhart CG, Taub DD
The Journal of biological chemistry 2006 Jun 16;281(24):16681-90
The Journal of biological chemistry 2006 Jun 16;281(24):16681-90
Identification of the expressed form of human cytosolic phospholipase A2beta (cPLA2beta): cPLA2beta3 is a novel variant localized to mitochondria and early endosomes.
Ghosh M, Loper R, Gelb MH, Leslie CC
The Journal of biological chemistry 2006 Jun 16;281(24):16615-24
The Journal of biological chemistry 2006 Jun 16;281(24):16615-24
A trans-Golgi network resident protein, golgin-97, accumulates in viral factories and incorporates into virions during poxvirus infection.
Alzhanova D, Hruby DE
Journal of virology 2006 Dec;80(23):11520-7
Journal of virology 2006 Dec;80(23):11520-7
The mTOR pathway is regulated by polycystin-1, and its inhibition reverses renal cystogenesis in polycystic kidney disease.
Shillingford JM, Murcia NS, Larson CH, Low SH, Hedgepeth R, Brown N, Flask CA, Novick AC, Goldfarb DA, Kramer-Zucker A, Walz G, Piontek KB, Germino GG, Weimbs T
Proceedings of the National Academy of Sciences of the United States of America 2006 Apr 4;103(14):5466-71
Proceedings of the National Academy of Sciences of the United States of America 2006 Apr 4;103(14):5466-71
Functional analysis of Arl1 and golgin-97 in endosome-to-TGN transport using recombinant Shiga toxin B fragment.
Tai G, Lu L, Johannes L, Hong W
Methods in enzymology 2005;404:442-53
Methods in enzymology 2005;404:442-53
The transmembrane domain is essential for the microtubular trafficking of membrane type-1 matrix metalloproteinase (MT1-MMP).
Remacle AG, Rozanov DV, Baciu PC, Chekanov AV, Golubkov VS, Strongin AY
Journal of cell science 2005 Nov 1;118(Pt 21):4975-84
Journal of cell science 2005 Nov 1;118(Pt 21):4975-84
Proteasome inhibition activates the transport and the ectodomain shedding of TNF-alpha receptors in human endothelial cells.
Peiretti F, Canault M, Bernot D, Bonardo B, Deprez-Beauclair P, Juhan-Vague I, Nalbone G
Journal of cell science 2005 Mar 1;118(Pt 5):1061-70
Journal of cell science 2005 Mar 1;118(Pt 5):1061-70
Differentiation- and maturation-dependent content, localization, and secretion of cystatin C in human dendritic cells.
Zavasnik-Bergant T, Repnik U, Schweiger A, Romih R, Jeras M, Turk V, Kos J
Journal of leukocyte biology 2005 Jul;78(1):122-34
Journal of leukocyte biology 2005 Jul;78(1):122-34
Coenzyme Q distribution in HL-60 human cells depends on the endomembrane system.
Fernández-Ayala DJ, Brea-Calvo G, López-Lluch G, Navas P
Biochimica et biophysica acta 2005 Jul 30;1713(2):129-37
Biochimica et biophysica acta 2005 Jul 30;1713(2):129-37
Proteolytic maturation and activation of autotaxin (NPP2), a secreted metastasis-enhancing lysophospholipase D.
Jansen S, Stefan C, Creemers JW, Waelkens E, Van Eynde A, Stalmans W, Bollen M
Journal of cell science 2005 Jul 15;118(Pt 14):3081-9
Journal of cell science 2005 Jul 15;118(Pt 14):3081-9
Intracellular localization and trafficking of fluorescein-labeled cisplatin in human ovarian carcinoma cells.
Safaei R, Katano K, Larson BJ, Samimi G, Holzer AK, Naerdemann W, Tomioka M, Goodman M, Howell SB
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Jan 15;11(2 Pt 1):756-67
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Jan 15;11(2 Pt 1):756-67
Proinflammatory mediators elicit secretion of the intracellular B-lymphocyte stimulator pool (BLyS) that is stored in activated neutrophils: implications for inflammatory diseases.
Scapini P, Carletto A, Nardelli B, Calzetti F, Roschke V, Merigo F, Tamassia N, Pieropan S, Biasi D, Sbarbati A, Sozzani S, Bambara L, Cassatella MA
Blood 2005 Jan 15;105(2):830-7
Blood 2005 Jan 15;105(2):830-7
Dynamic fluorescent imaging of human immunodeficiency virus type 1 gag in live cells by biarsenical labeling.
Rudner L, Nydegger S, Coren LV, Nagashima K, Thali M, Ott DE
Journal of virology 2005 Apr;79(7):4055-65
Journal of virology 2005 Apr;79(7):4055-65
The caveolae-mediated sv40 entry pathway bypasses the golgi complex en route to the endoplasmic reticulum.
Norkin LC, Kuksin D
Virology journal 2005 Apr 19;2:38
Virology journal 2005 Apr 19;2:38
Proteasome-dependent degradation of cytochromes P450 2E1 and 2B1 expressed in tetracycline-regulated HeLa cells.
Huan JY, Streicher JM, Bleyle LA, Koop DR
Toxicology and applied pharmacology 2004 Sep 15;199(3):332-43
Toxicology and applied pharmacology 2004 Sep 15;199(3):332-43
Activation of endogenous Cdc42 visualized in living cells.
Nalbant P, Hodgson L, Kraynov V, Toutchkine A, Hahn KM
Science (New York, N.Y.) 2004 Sep 10;305(5690):1615-9
Science (New York, N.Y.) 2004 Sep 10;305(5690):1615-9
Cross-presentation of a human tumor antigen delivered to dendritic cells by HSV VP22-mediated protein translocation.
Chhabra A, Mehrotra S, Chakraborty NG, Mukherji B, Dorsky DI
European journal of immunology 2004 Oct;34(10):2824-33
European journal of immunology 2004 Oct;34(10):2824-33
Autoantigen Golgin-97, an effector of Arl1 GTPase, participates in traffic from the endosome to the trans-golgi network.
Lu L, Tai G, Hong W
Molecular biology of the cell 2004 Oct;15(10):4426-43
Molecular biology of the cell 2004 Oct;15(10):4426-43
PIBF (progesterone induced blocking factor) is overexpressed in highly proliferating cells and associated with the centrosome.
Lachmann M, Gelbmann D, Kálmán E, Polgár B, Buschle M, Von Gabain A, Szekeres-Barthó J, Nagy E
International journal of cancer 2004 Oct 20;112(1):51-60
International journal of cancer 2004 Oct 20;112(1):51-60
Translocation of the inhibitor of apoptosis protein c-IAP1 from the nucleus to the Golgi in hematopoietic cells undergoing differentiation: a nuclear export signal-mediated event.
Plenchette S, Cathelin S, Rébé C, Launay S, Ladoire S, Sordet O, Ponnelle T, Debili N, Phan TH, Padua RA, Dubrez-Daloz L, Solary E
Blood 2004 Oct 1;104(7):2035-43
Blood 2004 Oct 1;104(7):2035-43
Intracellular accumulation of amyloidogenic fragments of amyloid-beta precursor protein in neurons with Niemann-Pick type C defects is associated with endosomal abnormalities.
Jin LW, Shie FS, Maezawa I, Vincent I, Bird T
The American journal of pathology 2004 Mar;164(3):975-85
The American journal of pathology 2004 Mar;164(3):975-85
Lipid-dependent bidirectional traffic of apolipoprotein B in polarized enterocytes.
Morel E, Demignot S, Chateau D, Chambaz J, Rousset M, Delers F
Molecular biology of the cell 2004 Jan;15(1):132-41
Molecular biology of the cell 2004 Jan;15(1):132-41
An MDR-EGFP gene fusion allows for direct cellular localization, function and stability assessment of P-glycoprotein.
Pétriz J, Gottesman MM, Aran JM
Current drug delivery 2004 Jan;1(1):43-56
Current drug delivery 2004 Jan;1(1):43-56
Vimentin is secreted by activated macrophages.
Mor-Vaknin N, Punturieri A, Sitwala K, Markovitz DM
Nature cell biology 2003 Jan;5(1):59-63
Nature cell biology 2003 Jan;5(1):59-63
Interaction of Hsp90 with the nascent form of the mutant epidermal growth factor receptor EGFRvIII.
Lavictoire SJ, Parolin DA, Klimowicz AC, Kelly JF, Lorimer IA
The Journal of biological chemistry 2003 Feb 14;278(7):5292-9
The Journal of biological chemistry 2003 Feb 14;278(7):5292-9
The human phosphatidylinositol phosphatase SAC1 interacts with the coatomer I complex.
Rohde HM, Cheong FY, Konrad G, Paiha K, Mayinger P, Boehmelt G
The Journal of biological chemistry 2003 Dec 26;278(52):52689-99
The Journal of biological chemistry 2003 Dec 26;278(52):52689-99
The melanocytic protein Melan-A/MART-1 has a subcellular localization distinct from typical melanosomal proteins.
De Mazière AM, Muehlethaler K, van Donselaar E, Salvi S, Davoust J, Cerottini JC, Lévy F, Slot JW, Rimoldi D
Traffic (Copenhagen, Denmark) 2002 Sep;3(9):678-93
Traffic (Copenhagen, Denmark) 2002 Sep;3(9):678-93
Biologically active APRIL is secreted following intracellular processing in the Golgi apparatus by furin convertase.
López-Fraga M, Fernández R, Albar JP, Hahne M
EMBO reports 2001 Oct;2(10):945-51
EMBO reports 2001 Oct;2(10):945-51
Subcellular localization of the melanoma-associated protein Melan-AMART-1 influences the processing of its HLA-A2-restricted epitope.
Rimoldi D, Muehlethaler K, Salvi S, Valmori D, Romero P, Cerottini JC, Levy F
The Journal of biological chemistry 2001 Nov 16;276(46):43189-96
The Journal of biological chemistry 2001 Nov 16;276(46):43189-96
Intracellular calcium signals regulating cytosolic phospholipase A2 translocation to internal membranes.
Evans JH, Spencer DM, Zweifach A, Leslie CC
The Journal of biological chemistry 2001 Aug 10;276(32):30150-60
The Journal of biological chemistry 2001 Aug 10;276(32):30150-60
Intracellular redirection of plasma membrane trafficking after loss of epithelial cell polarity.
Low SH, Miura M, Roche PA, Valdez AC, Mostov KE, Weimbs T
Molecular biology of the cell 2000 Sep;11(9):3045-60
Molecular biology of the cell 2000 Sep;11(9):3045-60
Intracellular redirection of plasma membrane trafficking after loss of epithelial cell polarity.
Low SH, Miura M, Roche PA, Valdez AC, Mostov KE, Weimbs T
Molecular biology of the cell 2000 Sep;11(9):3045-60
Molecular biology of the cell 2000 Sep;11(9):3045-60
A novel Rab6-interacting domain defines a family of Golgi-targeted coiled-coil proteins.
Barr FA
Current biology : CB 1999 Apr 8;9(7):381-4
Current biology : CB 1999 Apr 8;9(7):381-4
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Knockdown of Golgin-97 was achieved by transfecting T-47D with Golgin-97 specific siRNAs (Silencer® select Product # S5940, S5939). Western blot analysis (Fig. a) was performed using Nuclear enriched extracts from the Golgin-97 knockdown cells (lane 3), non-targeting scrambled siRNA transfected cells (lane 2) and untransfected cells (lane 1). The blot was probed with Golgin-97 Monoclonal Antibody (CDF4) (Product # A21270, 1:1000 dilution) and Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution). Densitometric analysis of this western blot is shown in histogram (Fig. b). Decrease in signal upon siRNA mediated knock down confirms that antibody is specific to Golgin-97.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
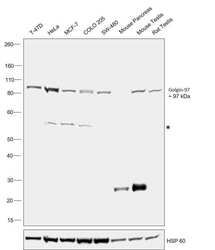
- Experimental details
- Western blot was performed using Anti-Golgin-97 Monoclonal Antibody (CDF4) (Product # A21270) and a 97kDa band corresponding to Golgin-97 was observed along with uncharacterized bands (*) at 55 kDa across cell lines and tissues tested. Whole cell extracts (30 µg lysate) of T-47D (Lane 1), HeLa (Lane 2), MCF7 (Lane 3), COLO 205 (Lane 4), SW480 (Lane 5), Mouse Pancreas (Lane 6), Mouse Testis (Lane 7) and Rat Testis (Lane 8) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177,1:20000 dilution) using the iBright FL 1000 (Product # A32752). Relative expression of Golgin-97 was observed to be high in Mouse Testis in comparison to no expression in Mouse Pancreas. Chemiluminescent detection was performed using SuperSignal™ West Pico PLUS Chemiluminescent Substrate (Product # 34580).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
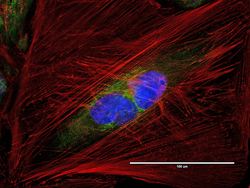
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 1 µg/mL anti-golgin-97 antibody (Product # A-21270) for labeling of Golgi for 30 min at room temperature and washed three times with dPBS. Cells were then incubated with a 1:1000 dilution of FITC-labeled secondary antibody (Product # A16079) for 30 min and washed three times in dPBS. Cells were labeled with NucBlue® Live (Product # R37605) and ActinRed™ 555 ReadyProbes® reagent (Product # R37112) according to protocol. Coverslips were then mounted using ProLong® Gold antifade reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
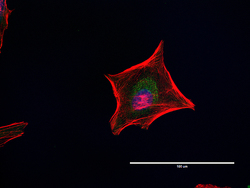
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 1 µg/mL anti-golgin-97 antibody (Product # A-21270) for labeling of Golgi for 30 min at room temperature and washed three times with dPBS. Cells were then incubated with a 1:1000 dilution of FITC-labeled secondary antibody (Product # A16079) for 30 min and washed three times in dPBS. Cells were labeled with NucBlue® Live (Product # R37605) and ActinRed™ 555 ReadyProbes® reagent (Product # R37112) according to protocol. Coverslips were then mounted using ProLong® Gold antifade reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
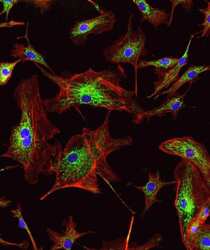
- Experimental details
- BPAE cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602). The mitochondrial ox-phos complex V inhibitor protein was stained using primary antibody: Mouse anti ATP Synthase Subunit IF1 Monoclonal Antibody, (clone 5E2D7) (Product # A-21355) detected with a goat anti-mouse Alexa Fluor® 488 secondary antibody (Product # A-11029). Actin was stained using Alexa Fluor® 594 phalloidin (Product # A12381) and nuclei were stained using NucBlue Live Cell Stain (Product # R37605). Slides were mounted using ProLong® Gold Antifade Kit (P7481) and images were acquired on the FLoid™ Cell Imaging Station (Product # 4471136).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
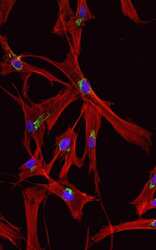
- Experimental details
- Human dermal fibroblasts, neonatal (HDFn) (C-004-5C) were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602). Golgi staining was done using an anti-Golgin-97 primary antibody (Product # A-21270) and a goat anti-mouse Alexa Fluor® 488 secondary antibody (Product # A-11029). Actin was stained using Alexa Fluor® 594 phalloidin (Product # A12381) and nuclei were stained using NucBlue™ Live Cell Stain (Product # R37605). Slides were mounted using ProLong® Gold antifade kit (P7481) and images were acquired on the FLoid™ Cell Imaging Station (Product # 4471136).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
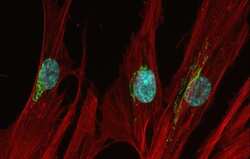
- Experimental details
- HDFn cells were fixed, permeabilized, and blocked using the Image-iT® Fixation/Permeabilization Kit (Product # R37602). Golgi were labeled with an anti-golgin 97 antibody (Product # A-21270) followed by detection with an Alexa Fluor® 488 secondary (Product # A-11029). Cells were also stained with Alexa Fluor® 594 phalloidin (Product # A12381) to label actin and NucBlue Fixed (Product # R37606) to label nuclei. Finally, cells were mounted in ProLong® Gold antifade reagent.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
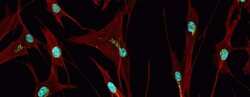
- Experimental details
- HDFn cells were fixed, permeabilized, and blocked using the Image-iT® Fixation/Permeabilization Kit (Product # R37602). Golgi were labeled with an anti-golgin 97 antibody (Product # A-21270) followed by detection with an Alexa Fluor® 488 secondary (Product # A-11029). Cells were also stained with Alexa Fluor® 594 phalloidin (Product # A12381) to label actin and NucBlue Fixed (Product # R37606) to label nuclei. Finally, cells were mounted in ProLong® Gold antifade reagent.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
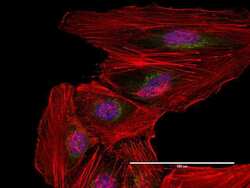
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 1 µg/mL anti-golgin-97 antibody (Product # A-21270) for labeling of Golgi for 30 min at room temperature and washed three times with dPBS. Cells were then incubated with a 1:1000 dilution of FITC-labeled secondary antibody (Product # A16079) for 30 min and washed three times in dPBS. Cells were labeled with NucBlue® Live (Product # R37605) and ActinRed™ 555 ReadyProbes® reagent (Product # R37112) according to protocol. Coverslips were then mounted using ProLong® Gold antifade reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
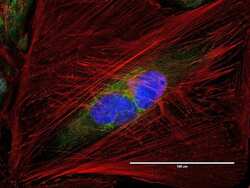
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 1 µg/mL anti-golgin-97 antibody (Product # A-21270) for labeling of Golgi for 30 min at room temperature and washed three times with dPBS. Cells were then incubated with a 1:1000 dilution of FITC-labeled secondary antibody (Product # A16079) for 30 min and washed three times in dPBS. Cells were labeled with NucBlue® Live (Product # R37605) and ActinRed™ 555 ReadyProbes® reagent (Product # R37112) according to protocol. Coverslips were then mounted using ProLong® Gold antifade reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
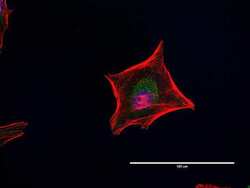
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 1 µg/mL anti-golgin-97 antibody (Product # A-21270) for labeling of Golgi for 30 min at room temperature and washed three times with dPBS. Cells were then incubated with a 1:1000 dilution of FITC-labeled secondary antibody (Product # A16079) for 30 min and washed three times in dPBS. Cells were labeled with NucBlue® Live (Product # R37605) and ActinRed™ 555 ReadyProbes® reagent (Product # R37112) according to protocol. Coverslips were then mounted using ProLong® Gold antifade reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
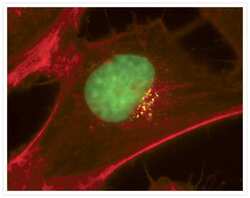
- Experimental details
- HeLa cells. Anti-human Golgin-97, mouse monoclonal CDF4, Alexa Fluor® 430 goat anti-mouse IgG, Alexa Fluor® 594 phalloidin, and SYTOX® Green nucleic acid stain. Fixed, permeabilized and RNase-treated HeLa cells were labeled with mouse monoclonal anti-human Golgin-97 monoclonal antibody (Product # A-21270) and visualized with the yellow-fluorescent Alexa Fluor® 430 goat anti-mouse IgG antibody (Product # A-11063). The F-actin was labeled with red-fluorescent Alexa Fluor® 594 phalloidin (Product # A12381) and the nuclei were stained with the SYTOX® Green nucleic acid stain (Product # S7020). This multiple-exposure image was acquired using a Nikon Diaphot fluorescent microscope equipped with a Quantix CCD camera.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
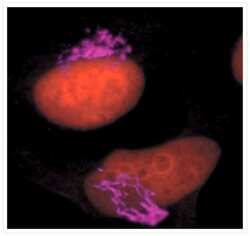
- Experimental details
- HeLa cells labeled with SYTOX® Orange nucleic acid stain, anti-Golgin-97 antibody and detected using Alexa Fluor® 647 goat anti-mouse IgG antibody. Fixed, permeabilized HeLa cells were labeled with anti-Golgin-97 polyclonal antibody (Product # A-21270) and detected using Alexa Fluor® 647 goat anti-mouse IgG antibody (Product # A-21235). The cells were counterstained with SYTOX® Orange nucleic acid stain (Product # S11368).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
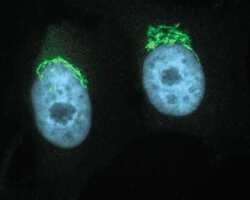
- Experimental details
- HeLa cells labeled with anti-golgin-97 antibody and detected using Alexa Fluor® 488 Goat Anti-Mouse IgG antibody. Fixed, permeabilized HeLa cells were labeled with anti-golgin-97 monoclonal antibody (Product # A-21270) and detected using Alexa Fluor® 488 Goat Anti-Mouse IgG antibody (Product # A-11001). The cells were counterstained with DAPI (D1306, D3571, D21490). (Product # A-21270).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
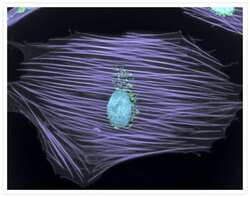
- Experimental details
- Muntjac skin cell labeled in three different colors. Golgi bodies in a muntjac skin cell were labeled with anti-Golgin-97 monoclonal antibody (Product # A-21270) and visualized with green-fluorescent Alexa Fluor® 488 Goat Anti-Mouse IgG1 antibody (Product # A-21121). Filamentous actin was labeled with Alexa Fluor® 680 phalloidin (Product # A22286, pseudocolored purple). Nuclei were stained with blue-fluorescent DAPI (Product # D1306, D3571, D21490). (Product # A-21270).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
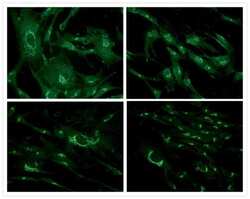
- Experimental details
- Increased label specificity and resolution afforded by Image-iT® FX signal enhancer. Fixed and permeabilized MRC-5 human lung fibroblast cells were labeled with mouse anti-human Golgin-97 primary antibody (Product # A-21270), then visualized with either FITC-conjugated goat anti-mouse IgG (F2761, top row) or Alexa Fluor® 488 Goat Anti-Mouse IgG (Product # A-11001, bottom row). Cells on the right were treated with Image-iT® FX signal enhancer (Product # I36933) prior to primary antibody incubation (Product # A-21270).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
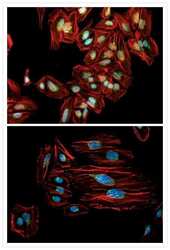
- Experimental details
- Golgi and actin staining in HeLa cells. Golgi in fixed and permeabilized HeLa cells labeled with anti-golgin-97 monoclonal antibody (Product # A-21270) and visualized with green-fluorescent Alexa Fluor® 488 Goat-Anti-Mouse IgG (Product # A-11001). Actin was stained with red-fluorescent Alexa Fluor® 594 phalloidin (Product # A12381); nuclei were stained with blue-fluorescent DAPI (Product # D1306, D3571, D21490). Treatment with Image-iT® FX signal enhancer (Product # I36933) largely eliminates nonspecific dye binding (bottom) as compared to untreated slide (top).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
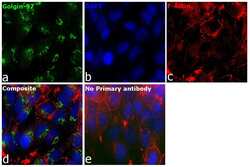
- Experimental details
- Immunofluorescence analysis of Golgin-97 was performed using 70% confluent log phase T-47D cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with Golgin-97 Monoclonal Antibody (CDF4) (Product # A21270) at 1:100 dilution in 0.1% BSA, incubated at 4 °C overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing golgi localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification in EVOS™ M7000 Imaging System (Product # AMF7000) and externally deconvoluted (D.Sage et al. / Methods 115 (2017) 28-41).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Human dermal fibroblasts, neonatal (HDFn) (C-004-5C) were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602). Golgi staining was done using an anti-Golgin-97 primary antibody (Product # A-21270) and a goat anti-mouse Alexa Fluor® 488 secondary antibody (Product # A-11029). Actin was stained using Alexa Fluor® 594 phalloidin (Product # A12381) and nuclei were stained using NucBlue Live Cell Stain (Product # R37605). Slides were mounted using ProLong® Gold antifade kit (P7481) and images were acquired on the FLoid Cell Imaging Station (Product # 4471136).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Golgi in HeLa cells detected with Alexa Fluor® 546 tyramide. Golgi in HeLa cells detected with Alexa Fluor® 546 tyramide. Cells were fixed and permeabilized, then labeled with anti-human Golgin-97 polyclonal antibody (Product # A-21270) and detected using HRP-conjugated goat anti-mouse IgG antibody and Alexa Fluor® 546 tyramide, which are components of the TSA Kit #3 (Product # T-20913). The nuclei were counterstained using DAPI (Product # D1306, D3571, D21490). The images were acquired using filters appropriate for DAPI and Alexa Fluor® 546 and processed using MetaMorph software from Universal Imaging Corp.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
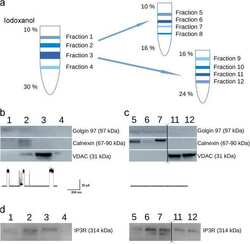
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
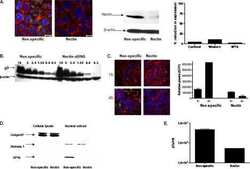
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
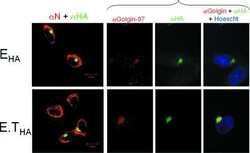
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
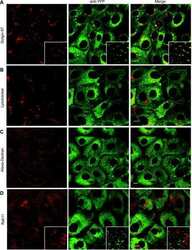
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
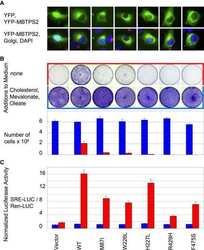
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
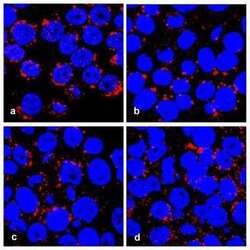
- Experimental details
- Figure 3 Golgi fragmentation in apoptotic HT29 cells. Cells were treated with serial concentrations of geoditin A for 24 h, fixed in 3% paraformaldehyde (PFA) and incubated with antibody against Golgi marker protein, golgin-97 and visualized by secondary antibody IgG conjugated to Alexa-647. Nuclei were counterstained with DAPI. Typical Golgi stack is shown at the juxtanuclear region of the untreated HT29 cells (a), while Golgi fragmentation becomes prominent in geoditin A-treated in a dose-dependent manner (b, c, and d for 10, 20 and 40 mug/mL, respectively).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
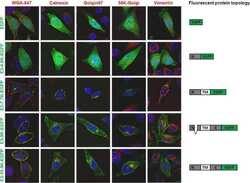
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
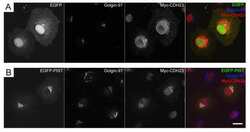
- Experimental details
- Figure 5 Retention of cadherin 23 by PIST in trans-golgi network in transfected COS-7 cells . A. Myc-tagged cadherin 23 localizes in the cytoplasm. B. In the presence of EGFP-tagged PIST, Myc-tagged cadherin 23 co-localizes with EGFP-PIST and a trans-golgi network marker golgin-97. Myc-cadherin 23 was stained with rabbit anti-cadherin 23 antibody and golgin-97 was stained with mouse anti-golgin-97 antibody. Scale bar, 20 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
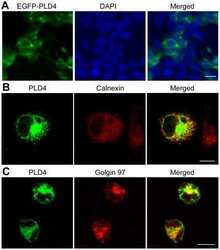
- Experimental details
- Figure 7 Intracellular localization of PLD4 protein exogenously expressed in cultured cell lines. A, EGFP fluorescent imaging (green) and DAPI nuclear staining (blue) of HEK293 cells transfected with pEGFP-PLD4. PLD4 is localized in a meshwork-like structure and around the periphery of nuclei. B and C, immunocytochemical localization of PLD4 protein (green) and either calnexin, an ER marker (red) (B) or golgin 97, a trans -Golgi marker (red) (C), in HeLa cells transfected with pcDNA3-PLD4. PLD4 is colocalized with calnexin in the ER and with golgin 97 in the Golgi complex. Scale bars = 10 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
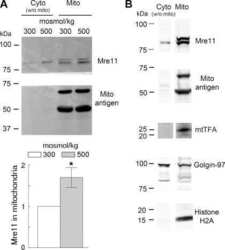
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
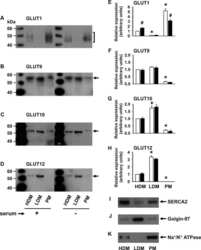
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1 Podocin localizes to endosomal vesicles. Immunofluorescence for transiently expressed, V5- (a-c, e) or Flag-tagged (d) podocin using anti-V5 or anti-Flag antibody in HeLa cells. Costainings of endogenous calnexin (a), endogenous golgin-97 (b), endogenous EEA1 (c), Lysotracker Red (d) and eGfp-tagged and transiently expressed CD63 (e) as markers for the endoplasmatic reticulum, Golgi apparatus, early endosomes, acidic organelles and late endosomes respectively, displayed that podocin localizes to the endosomal compartment. Analogously to HeLa cells, transiently expressed V5-tagged podocin colocalizes with eGFP tagged CD63/LAMP3 in cultured human podocytes (f).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
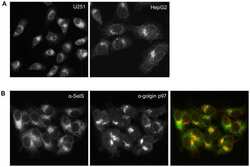
- Experimental details
- Figure 8 Endogenous localization of SelS includes a perinuclear accumulation. A , U251 cells or HepG2 cells were examined for the localization of endogenous SelS protein by immunofluorescence as described in Materials and Methods . Images were taken at 40x magnification. B , Costaining of SelS (green) and the Golgi (red) was performed using alpha-SelS and alpha-golgin p97 antibodies in HepG2 cells. Regions of colocalization are indicated in yellow (40x magnification).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
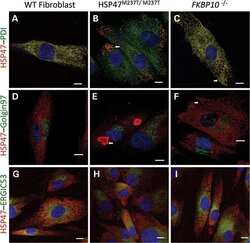
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
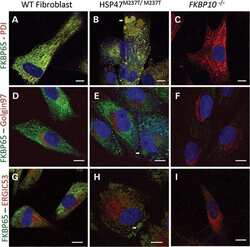
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
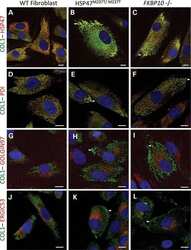
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
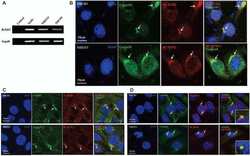
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 6 Characterization of exs derived from ascites using antibodies. (a) Presence of characteristic exs markers Alix, Hsp70 and Tsg101 as well as CD63, and absence of ""antimarker"" Golgin A1 (present in the cells of origin but absent from exs) was verified by western blotting (WB) in cells (P 0.2 fraction) and exs from ovarian cancer patient #2. (b) TEM of exs isolated from ascites of ovarian cancer patient #2. Exs marker CD63 is detected at the surface of exs by secondary antibodies conjugated to 10-nm gold particles. (c) CD63 + , exs derived from ascites of ovarian cancer patient #2 can be detected by FC analysis of CD63-PE antibody-stained exs (right panel). Left panel, exs stained with isotype control antibody-PE. (d) Double labeling of exs from ascites of patient #1 by CFSE and EpCAM-APC antibody reveals feasibility of double labeling and demonstrates that only 11.5% of CFSE + exs are EpCAM + . (e) Summary of timing of the presented protocol. Each step is marked in the time scale by different color. For collection of EVs from conditioned media, cells are plated a day ahead. Isolation of exs can be achieved in 1 day. Setting up the instrument and FC data acquisition can be completed within the morning of the second day. - F, 0.22 um filtered; - NF, 0.22 um non-filtered.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
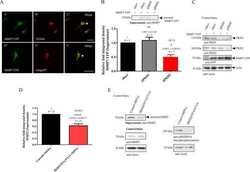
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
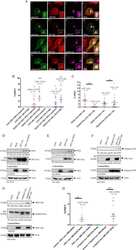
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
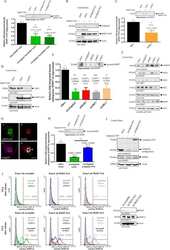
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
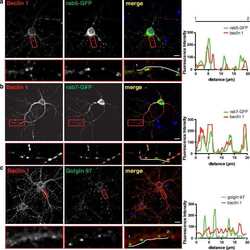
- Experimental details
- Fig. 2 Beclin 1 localizes to endosomes in neurons. Representative images showing colocalization of beclin 1 with ( a ) Rab5-GFP, colocalization coefficient 0.49 +- 0.03, ( b ) Rab7-GFP, colocalization coefficient 0.50 +- 0.03 or ( c ) Golgin 97, colocalization coefficient 0.13 +- 0.01 in primary hippocampal neurons. Regions in the red boxes are enlarged below. Intensity traces (offset white lines) are plotted at right. Scale bars are 10 mum. Colocalization coefficients are the combined results of 2-3 independent experiments ( n = 28-30 total fields/group).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1. Characterization of SIDT2. (A) Lysosomes (Lys) were isolated from mouse brain homogenates (Hom), and analyzed by immunoblotting using polyclonal goat anti-SIDT2 antibody and antibodies against LAMP2 (lysosomal marker), RAB7A (late endosome and lysosome), RAB5A (early endosome), CANX (endoplasmic reticulum), COX4I1 (mitochondria), GOLGA1 (Golgi apparatus), GAPDH (cytosol), LMNA/lamin A (nuclei), and MAP1LC3A/B (autophagosome). (B) Neuro2a cells expressing GFP-tagged SIDT2 were incubated with LysoTracker Red. Fluorescence images were visualized using a confocal laser-scanning microscope. Scale bar: 10 mum. Colocalization rate was quantified using ImageJ software (right panel, n = 3). ( C ) Neuro2a cells expressing GFP-tagged SIDT2 were fixed, and immunostained using anti-RAB7A, anti-EEA1 (early endosomal marker) or anti-MAP1LC3A/B antibodies. Fluorescent images were obtained using confocal microscopy. Scale bars: 5 mum. Colocalization rate was quantified (right panels, n = 3). (D) Lysosomes were isolated from HeLa cells expressing SIDT2-FLAG or CTSB-FLAG. Isolated lysosomes (4 mug protein) were incubated with the indicated concentrations of trypsin at 37degC for 5 min. Proteins in the samples were analyzed by immunoblotting using an anti-FLAG antibody. (E) LAMP2C and SIDT2 or SIDT2-FLAG were overexpressed in HeLa cells. Cell lysates were prepared and immunoprecipitated with an anti-FLAG antibody. Cell lysates and the resulting immunoprecipitant were analyzed by immuno
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 Altered trafficking of ATG9A in SMPD1 -deficient cells. (A) NAGLU activities ( top ), indicated immunoblots ( middle ) and ATG9A distribution profiles of density gradient fractions of postnuclear supernatant fractions from HC-3 and NPA-3 fibroblasts ( bottom ). When indicated, HC-3 fibroblasts were starved in HBSS plus 1 g/L glucose and treated with 2 nM ConA for 2 h (S/C). The migration of MAP1LC3B-I and MAP1LC3B-II are marked with a line and greater-than symbol, respectively. ATG9A distribution in fractions is expressed as percentage of the total ATG9A in all fractions as quantified by densitometry and ImageJ software. See figures S2A and B for additional data. (B and C) Representative confocal images of HC-1 and NPA-3 fibroblasts and CRISPr-Cas-9 wild-type (WT) and SMPD1 -KO (KO) MCF7 cells stained as indicated. When indicated, cells were starved in HBSS plus 1 g/L glucose for 2 h. Step size, 300 nm. See figures S2C and D for additional staining. (D) Manders coefficients for colocalization of TFRC with ATG9A and vice versa ( left ), and percentages of ATG9A and colocalized ATG9A-TFRC in the juxtanuclear area (4 um from nucleus) in WT and KO cells. Bars, SEM for 20 randomly chosen cells. (E) Representative immunoblots of the indicated proteins in lysates of WT and KO cells. (F) Quantification of the number of WIPI2 puncta/cell ( top ) and the percentage of ATG9A-positive WIPI2 puncta of all WIPI2 puncta ( bottom ) in WT and KO cells. Bars, SEM for 20 randomly chose
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
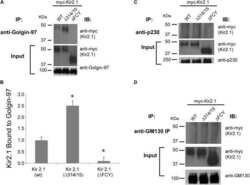
- Experimental details
- FIGURE 1 Kir2.1 specifically binds to Golgin-97. Immunoprecipitation (IP) performed with (A) anti-Golgin-97; (C) anti-p230; (D) anti-GM130 antibodies from COS cells transfected with myc-Kir2.1 wild type (WT), the TGN-export mutant (Delta314-315), and the ER-export mutant (DeltaFCY) and then immunoblotted (IB) with myc-antibodies to detect Kir2.1 in the Golgin immunoprecipitates. 5% input is shown below; (B) Quantification of the relative amounts of Kir2.1 immunoprecipatated with Golgin-97. * P < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
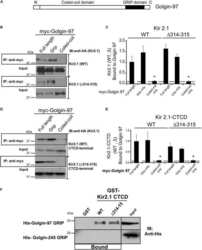
- Experimental details
- FIGURE 2 Golgin-97 GRIP domain directly interacts with Kir2.1. (A) Domain structure of Golgin-97, showing the GRIP domain and the coiled-coil domain. (B) Anti-myc immunoprecipitation (IP) followed by anti-HA immunoblotting (IB) from COS cells co-transfected with myc-Golgi 97 (Full length, Grip domain only, or coiled-coiled domain only) and HA-Kir2.1 wild type, WT, or the TGN-export mutant (Delta314-315) or (D) the HA-tagged Kir2.1 COOH-terminal cytoplasmic domain (CTCD). (C,E) Quantitative summaries comparing relative amounts of Kir2.1 (WT or Delta314-315) immunoprecipitated with full-length Golgin-97, or GRIP or Coiled-Coil domain, * P < 0.05. (F) GST pulldowns: GST alone or GST fusion proteins of the Kir2.1 CTCD were used to test for direct interaction with His-tagged GRIP domains of Golgin-97 or Golgin-245 purified from bacteria. Protein bound is shown relative to 5% input.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
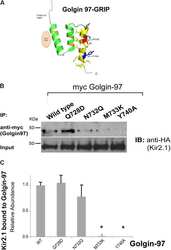
- Experimental details
- FIGURE 4 Mapping the residues in GRIP domain involved in Kir2.1 binding. (A) Model of the Golgin-97 GRIP domain. First helix directly interacts with Arl1, while the third helix contains residues (M733, Y740) that are necessary for binding to Kir2.1. (B) The HA-tagged Kir2.1 channel was co-transfected with myc-tagged Golgin-97 (WT or GRIP domain mutants), and cell lysates were subjected to immunoprecipitation with anti-myc and then immunoblotted with anti-HA to detect Kir2.1 bound. (C) Summary Data, Relative to Average WT Kir2.1 immunoprecipitated with wild type (WT) Golgin-97 GRIP. * P < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
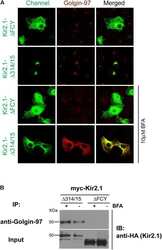
- Experimental details
- FIGURE 5 Golgin-97 Interacts with Kir2.1 in the Golgi. (A) Confocal images of Kir2.1 (TGN-export mutant (Delta314-315) compared to ER-export mutant DeltaFCY) in COS-cells (green) and Golgi-97 (red) before (top) and after 10 min of brefeldin A treatment. (B) Anti-Golgin-97 immunoprecipitation (IP) followed by anti-HA immunoblotting (IB) from COS cells transfected with the HA-Kir2.1 mutants and treated with vehicle (-) or Brefeldin (+).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
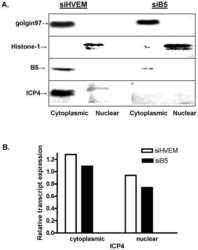
- Experimental details
- Figure 6 B5 silencing does not block polyadenylation or nuclear export of RNA. (A) Transfected cells were infected with HSV-2(G) (moi 3 pfu/cell) and 3 h pi, cells were harvested and nuclear and cytoplasmic fractions isolated and Western blots were prepared and probed for golgin97, histone-1, B5 and ICP4. (B) Polyadenylated mRNA was isolated from the total RNA and from the cytoplasmic and nuclear fractions and then analyzed for ICP4 by qRT-PCR. Results are presented as the ratio of ICP4/RPLPO in each fraction (cytoplasmic or nuclear) to ICP4/RPLPO in the unfractionated total polyA RNA and are representative of 3 independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
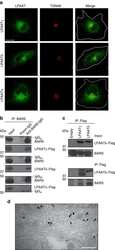
- Experimental details
- Figure 1 BARS interacts with LPAATdelta. ( a ) Representative confocal microscopy images of COS7 cells transfected with Flag-tagged LPAATgamma, LPAATdelta and LPAATeta, and fixed and processed for immunofluorescence with a monoclonal anti-Flag antibody (green) and with a polyclonal anti-TGN46 antibody (red; as indicated). Dotted lines indicate cell borders. ( b ) BARS immunoprecipitation (IP:BARS) of lysate from HeLa cells co-expressing BARS and LPAATgamma-Flag, LPAATdelta-Flag or LPAATeta-Flag. Representative western blotting (antibodies as indicated) of total lysate (input) and immunoprecipitated proteins with preimmune-IgG (Preim-IgG) or anti-BARS-IgG (as indicated). IgG H , IgG heavy chain. ( c ) Immunoprecipitation with an anti-Flag antibody (IP:Flag) of lysate from HeLa cells co-transfected with BARS and LPAATgamma-Flag, LPAATdelta-Flag or the empty vector. Representative western blotting of total lysate (input) and Flag-immunoprecipitated proteins with an anti-Flag monoclonal antibody or the anti-BARS polyclonal antibody (as indicated). ( d ) Representative electron microscopy image of HeLa cells transfected with Flag-tagged LPAATdelta for 24 h, and fixed and processed for cryo-immuno-electron microscopy with a monoclonal anti-Golgin-97 antibody (15-nm gold particles) and with a polyclonal anti-LPAATdelta antibody (10-nm gold particles). Molecular weight standards (kDa) in b and c , are indicated on the left of each panel. Data are representative of three independent e
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
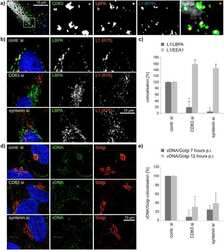
- Experimental details
- Figure 8 Trafficking of HPV16 to multivesicular endosomes requires CD63/syntenin-1 complex. ( a ) Representative pictures of CD63-GFP (green), LBPA (red), and HPV16 L1 (blue) triple staining with LBPA mAb and L1 pAb K75 antibodies. ( b,c ) Colocalisation analysis of HPV16 L1 and LBPA. HeLa cells were transfected with control siRNA, CD63 siRNA or syntenin-1 siRNA and infected with HPV16 PsV and stained with LBPA mAb and L1 pAb K75 antibodies. ( b ) Shows representative pictures of L1 (red) and LBPA (green) colocalisation. Nuclei are shown in blue. ( c ) Colocalisation of LBPA and L1 as well as early endosome marker EEA1 and L1 was analysed as in Fig. 3e ( P = 0,062 for CD63 siRNA and 0,069 for syntenin-1 siRNA). ( d,e ) Colocalisation analysis of viral DNA and the Golgi. HeLa cells were transfected with control, CD63 or syntenin-1 siRNA and infected with EdU-HPV16 PsV for 7 or 12 hours. ( d ) Shows representative pictures of viral DNA (vDNA, green) and Golgi (red) 12 hours post infection. ( e ) Colocalisation of viral DNA and Golgi was analysed as above and normalized to infected control siRNA treated cells. * P < 0.05 compared to control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
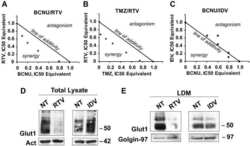
- Experimental details
- Figure 4 (A) Isobolograms analysis of inhibition of growth of U87MG cells (A-C) and effects of RTV and IDV treatment on GLUT1/SLC2A1 transporter levels in U87MG cells (D-E). (A-C) The ""line of additivity"" connects the values of the IC50 of the two drugs used in combination, thus separating the quadrant into two parts; points plotted below the line of additivity indicate synergy (activity of the combination is larger than expected for the simple combination of the two drugs), whereas points above the line indicate antagonism (activity of the combination is lower than the expected for the simple combination of the two drugs). BCNU-RTV: (A) The two drugs at all tested concentrations show synergic activity. TMZ-RTV: (B) The two drugs at all tested concentrations show synergic activity. BCNU-IDV: (C) The two drugs show addictive or antagonistic behavior at all concentrations except for the highest, when we obtained an indication of minimal synergic activity. (D) Western blot of total lysates of U87MG cells: RTV reduced the level of GLUT1/SLC2A1 immunoreactivity, whereas IDV increased GLUT1/SLC2A1 level compared with untreated cells (NT). The same membrane was stripped and reacted with an antibody against actin (Act) whose level was similar in all samples. (E) As in D, but the cells were fractionated, and only the fraction containing the LDMs, where the GLUTs-containing vesicles reside, was analyzed by Western blot. As in the total extract, the GLUT1/SLC2A1 signal was decreased o
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
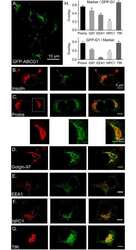
- Experimental details
- Fig 2 Examination of GFP-G1 by confocal fluorescence microscopy shows that it accumulates and colocalizes with markers of the trans-Golgi network and endosomal recycling compartment. (A) Low magnification survey illustrating that GFP-G1 generally is accumulated perinuclearly, mostly in a finely granular pattern with occasional surrounding brighter puncta. (B-G) Higher magnification images comparing the distribution of GFP-G1 to organelle markers. (B) Minimal co-localization with insulin at the cell periphery where mature granules accumulate but noticeable overlap perinuclearly where storage granules are being formed. (C) Extensive overlap with proinsulin accumulated perinuclearly and resembling the perinuclear overlap seen in panel B. Expanded insets (below) show the GFP-G1 fluorescence has the same pattern as proinsulin but is not especially co-concentrated with proinsulin in brighter puncta of the latter. (D) GFP-G1 exhibits a highly similar pattern to the trans-Golgi/TGN tethering protein Golgin-97 but is more diffusely distributed. (E) Limited overlap of GFP-G1 with EEA1 marking early endosomes. (F) Limited overlap of GFP-G1 with NPC1 marking late endosomes and lysosomes. (G) Substantial overlap of GFP-G1 with transferrin receptor (TfR), which is concentrated in the endosomal recycling compartment. GFP-G1 is also visible at the plasma membrane and diffusely within the cytosol (presumably ER). (H) Coefficients determined by Manders' overlap analysis for each of the m
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
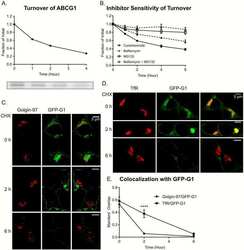
- Experimental details
- Fig 4 ABCG1 and GFP-G1 exhibit rapid turnover. (A) Pulse-chase biosynthetic labeling with 35 S-amino acids and immunoadsorption of ABCG1. Phosphorimage (below) and quantification (above) show that the transporter in INS1 cells is short-lived with a halftime of turnover of ~2h, as in other cell types. Data are taken from one experiment (full phosphorimage shown in Figure A in S3 Fig ). (B) Results from western blotting showing loss of ABCG1 following inhibition of protein synthesis (by cycloheximide) occurs by both ERAD/proteasomal degradation (inhibited by MG132) and lysosomal degradation (inhibited by the acidification blocker bafilomycin A). Data are from 7 experiments; error bars indicate SEM. Results for individual experiments are in S1 File . (C) Representative fluorescence images from samples treated for 0, 2, and 6h with cycloheximide (CHX). Both diffuse cytoplasmic GFP-G1 (presumed to be ER associated) and GFP-G1 colocalized with the TGN marker Golgin-97 are lost by 2h while the remainder of accumulated perinuclear GFP-G1 and much of the cell surface transporter is lost by 6h. (D) Complementary images to panel C showing perinuclear GFP-G1 remains colocalized with TfR at 2h post cycloheximide addition but is lost by 6h. Images for panels C and D are taken from one of two independent experiments that were performed to evaluate the loss of GFP-G1 colocalized with both Golgin-97 and TfR. (E) Manders' overlap analysis for colocalization of GFP-G1 with Golgin-97 an
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
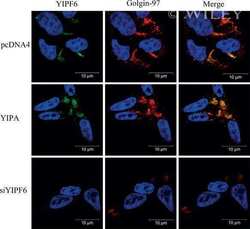
- Experimental details
- YIPF6 localizes to the Golgi apparatus. Immunofluorescence of modified 22Rv1 cells stained with antibodies specific for YIPF6 (green) and Golgin-97 (red). pcDNA4 is 22Rv1 cells transfected with vector control. YIPA cells overexpress the full length variant of YIPF6, and the siYIPF6 represents 22Rv1 cells that have been treated with siRNA specific for YIPF6.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
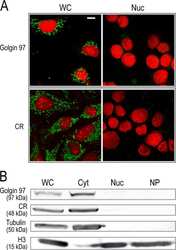
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
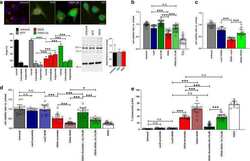
- Experimental details
- Fig. 3 Compound 2a counteracts G93A-mediated Golgi fragmentation. a merged confocal stacks for GFP and Golgin97 in untransfected-; pcDNA3.1(+)SOD1 G93A (1 µg/well)-pCAAG-GFP (0.2 µg/well)-; pcDNA3.1(+)SOD1 G93A (1 µg/well)-pCAAG-GFP (0.2 µg/well) + 2a (10 µM)- and pCAAG-GFP (1.2 µg/well)-transfected Neuro2a (4 x 10 4 cells/well). Morphological analysis of Golgi in normal, intermediate or fragmented was done in nontransfected cells ( n = 83 independent cells), G93A-GFP ( n = 88 independent cells), G93A-GFP+ 2a ( n = 84 independent cells), and GFP ( n = 109 independent cells). The histogram in panel a shows Golgi subclasses (% of the total number of Golgi constructs, means +- SD) examined over four independent experiments, p < 0.0001 for all comparisons. Protein lysates from nontransfected and transfected Neuro2a cells (3 x 10 5 cells/well, pcDNA3.1(+)SOD1 G93A (2 µg/well)-pCAAG-GFP (0.5 µg/well) or with pCAAG-GFP (2 µg/well) were immunoblotted with antibodies against Golgin97 and beta-Actin. The histogram shows quantifications (mean fold changes +- SD versus untreated cells, n = 3 wells/group). b Neuro2a cell viabilities (mean ratio +- SD compared to untransfected cells, n = 19 independent wells) upon the following treatments: pCMV-LacZ (1.25 µg/well, n = 18 independent wells), pcDNA3.1(+)SOD1 G93A (1.25 µg/well, n = 18 independent wells), pcDNA3.1(+)SOD1 G93A (1.25 µg/well) + 2a (10 µM, n = 24 indep
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
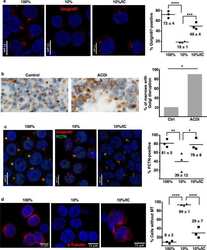
- Experimental details
- Fig. 1 Organelle perturbations associated with erythroid iron restriction. a Documentation of Golgi disruption by immunofluorescence on human CD34+ progenitors cultured 3 days in iron-replete (100% TSAT) or deficient (10% TSAT) erythroid medium +- isocitrate rescue (IC). Red: Golgin97; blue: DAPI. Graph: % cells with detectable Golgi. Numbers indicate mean +- SEM; n = 3 biologically independent experiments; ***, **** P = 0.002, 0.0001, one-way ANOVA with Tukey post hoc test. b Golgi assessment in human marrow samples by immunoperoxidase on clot sections from non-anemic controls and patients with anemia of chronic disease and inflammation (ACDI). 200x magnification. Brown: Giantin; blue: hematoxylin. Graph: % of marrow samples with Golgi disruption in >50% erythroblasts. n = 5 for control and 10 for ACDI patient samples; * P = 0.017, Fisher''s exact two-tailed test. c Effects on pericentriolar material (Pericentrin: PCTN) and adjacent Golgi documented by immunofluorescence on cells as in a . Red: Golgin97; green: Pericentrin; blue: DAPI Graph: % cells with PCTN+ centrosomes. Numbers indicate mean +- SEM; n = 3 independent experiments; *, ** P = 0.049, 0.037, one-way ANOVA with Tukey post hoc test. d Microtubule alterations demonstrated by immunofluorescence on cells as in a . Red: beta-tubulin; blue: DAPI. Graph: % cells with undetectable microtubule structures (MT). Numbers indicate mean +- SEM; n = 3 independent experiments; **** P = 0.0001, one-way ANOVA with Tukey post hoc
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
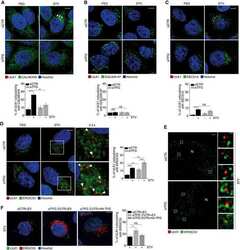
- Experimental details
- Figure 2 TFG is essential for proper ULK1 complex distribution during autophagy A-D HeLa cells were transfected with specific RNAi oligonucleotides (siTFG) or unrelated oligonucleotides as a negative control (siCTR) and grown in fed or starved conditions for 1 h. Cells were fixed and co-labelled with ULK1 (red) antibody and (A) anti-CALNEXIN (green) antibody to highlight ER structures ( n = 3 independent experiments, n >= 16 fields analysed), (B) GOLGIN-97 (green) antibody to label GOLGI complex ( n = 3 independent experiments, n = 12 fields analysed), (C) SEC31A (green) to point out COPII vesicles ( n = 3 independent experiments, n >= 16 fields analysed) and (D) ERGIC53 (green) to mark ERGIC compartment ( n = 3 independent experiments, n >= 14 fields analysed). Analysis of ULK1 localization with each compartment is reported in each graph. Co-localization analyses were performed by ImageJ plugin Jacop. Values of Mander's coefficient for ULK1 are expressed in percentage as mean +- SEM. Statistical analyses were performed by two-way ANOVA followed by Tukey's multiple comparison test. ** P < 0.01; *** P < 0.001; **** P < 0.0001; and ns, not significant. Scale bars 5 mum. White arrowheads point at ULK1 puncta associated with the analysed markers. E HeLa cells were transfected as in A), labelled with the indicated antibodies and imaged by SIM microscope. Scale bar in large panels, 5 mum; in small panels, 1 mum. N, nucleus. F HeLa cells were co-transfected with control siRNA and an
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
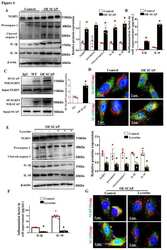
- Experimental details
- Figure 4. Sterol-resistant SCAP overexpression induces the activation of NLRP3 in?ammasomes in VSMCs. (A) Representative immunoblots for NLRP3, Procaspase-1, cleaved caspase-1, IL-1beta, and IL-18 in SCAP-overexpressing VSMCs. (B) Expression of IL-1beta and IL-18 in VSMC supernatants as measured by ELISA. (C) Immunoblotting of immunoprecipitation with anti-SCAP or anti-NLRP3 in SCAP-overexpressing VSMCs. (D) Representative images of immuno?uorescence staining of NLRP3 (green) or SCAP (green) with Golgi(red) in control and SCAP-overexpressing VSMCs. (E) Representative immunoblot for NLRP3, Pro-caspase-1, cleaved caspase-1, IL-1beta, and IL-18 in SCAP-overexpressing VSMCs treated with lycorine for 4 h. (F) Expression of IL-1beta and IL-18 in VSMC supernatants as measured by ELISA. (G) Representative images of the immuno?uorescence staining of NLRP3 (green) or SCAP (green) with Golgi (red) in SCAP VSMCs after treatment with lycorine for 4 h. The data are presented as the means+-SD of 3 independent experiments. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
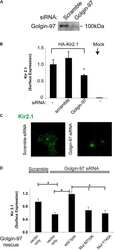
- Experimental details
- FIGURE 6 Golgin-97 is required for efficient delivery of Kir2.1 to the cell surface membrane from the Golgi. (A) Immunoblot of Golgin-97 from COS cells transfected with scramble or Golgi-97 siRNA. (B) Surface Expression of external HA-epitope tagged Kir2.1 in COS cells co-transfected with scramble or Golgi-97 siRNA or nothing. Mock are cells transfected with vector alone. (C) Representative confocal microscopy of Kir2.1 in COS cells transfected with scrample, or Golgin-97 siRNA. (D) Rescue Studies, Surface Expression of external HA-epitope tagged Kir2.1 in COS cells co-transfected with scramble or Golgi-97 siRNA and Golgin-97 rescue cDNA (WT vs mutant), * P < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
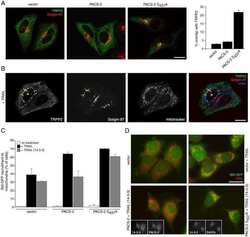
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
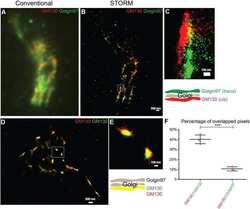
- Experimental details
- Figure 1 Two-color STORM can be utilized to reveal spatial relationships of Golgi-localized proteins at high resolution. (A,B) Localization patterns of a cis -Golgi marker (GM130) and a trans -Golgi marker (Golgin-97) in the same COS7 cell visualized through conventional fluorescent microscopy (A) or through two-color STORM (B) . Scale bar, 500 nm. (C) A magnified view of the indicated area in (B) . Scale bar, 100 nm. (D) Localization patterns of a cis -Golgi marker (GM130) labeled with two different fluorescent dyes in the COS7 cell revealed through two-color STORM. Scale bar, 500 nm. (E) A magnified view of the indicated area in (D) . Scale bar, 100 nm. (F) Quantification of the percentage of overlapped pixels between GM130 and Golgin-97 and between Alexa Fluor 750-labeled GM130 and Alexa Fluor 647-labeled GM130 based on four super-resolution images acquired from one experiment in each experimental group (mean +- SD). *** p < 0.001 by two-tailed Student's t -test. The super-resolution images showed the localization patterns of the indicated protein in the whole juxtanuclear area. Each dot in the graphs represents data of the whole juxtanuclear Golgi area of each individual cell.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
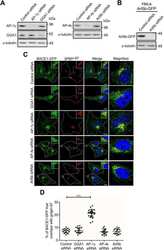
- Experimental details
- FIGURE 4: Depletion of adaptor protein AP-1 results in BACE1 accumulation in the TGN. (A) HeLa cells stably expressing BACE1-GFP were transfected with either control siRNA, AP-1gamma siRNA, GGA1 siRNA, AP-4epsilon siRNA, or Arl5b siRNA for 72 h. (B) HeLa cells stably expressing Arl5b(Q70L)-GFP were transfected with either control siRNA or Arl5b siRNA for 72 h. Cells in A and B were lysed in RIPA buffer, and 20 mug of cell extracts was subjected to SDS-PAGE on 4-12% gradient polyacrylamide gel. Proteins were transferred to a polyvinylidene fluoride (PVDF) membrane and probed with (A) mouse anti-AP-1gamma, rabbit anti-GGA1, mouse anti-AP-4epsilon, (B) mouse anti-GFP, and (A and B) mouse anti-a-tubulin antibodies using a chemiluminescence detection system. (C) Monolayers of HeLa cells were fixed, permeabilized, and blocked, followed by staining with mouse anti-golgin-97 antibodies (red) and DAPI (blue). Higher magnification of the merged images of the cells marked * are also shown. Bar represents 10 mum. (D) The percentage of BACE1-GFP at the TGN was calculated as a percentage of total BACE1-GFP pixels that overlapped with golgin-97. Data are represented as the mean +- SD of three independent experiments ( n = 15) and analyzed by one-way ANOVA using Tukey's test. *** p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
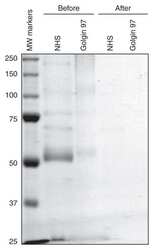
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
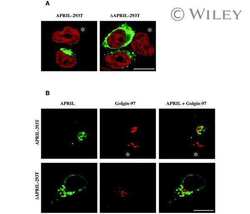
- Experimental details
- APRIL associates with the Golgi apparatus. ( A ) APRIL is intracellularly localized. APRIL staining is shown in green, DNA staining with Topro-3 in red. ( B ) APRIL colocalizes with anti-human golgin-97 (CF4) monoclonal antibody, a marker for Golgi apparatus. APRIL staining is shown in green, staining for golgin-97 (CF4) in red. Yellow visualizes the overlap of the two fluorescent antibodies. Images are shown of transiently transfected 293T cells. Similar images were obtained on stably transfected cells. Asterisks indicate non-transfected cells serving as negative controls. Bar, 50 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
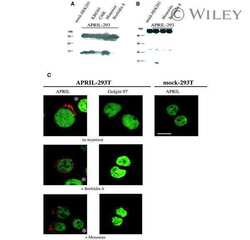
- Experimental details
- Processing of APRIL takes place in the Golgi apparatus. ( A ) BFA abrogates cleavage of APRIL. HEK 293T cells were transiently transfected with APRIL, and treated for 5 h with BFA (3.5 muM), monensin (3.5 muM), chloromethylketone derivative (CMK; furin convertase inhibitor, 50 muM) or KB8301 (metalloprotease inhibitor, 10 muM) and subsequently analysed by western blot employing affinity-purified polyclonal anti-APRIL antibody. Proteins were separated in 15% SDS-PAGE. ( B ) Soluble APRIL cannot be immunoprecipitated from culture supernatants of BFA- or monensin-treated APRIL transfectants. Immunoprecipitates of culture supernatants from APRIL-transfected cells treated with BFA or monensin for 5 h were analysed by western blot. Proteins were separated on a 15% SDS-polyacrylamide gel. ( C ) Treatment with BFA, but not monensin, causes delocalization of APRIL. Cells were incubated with BFA (3.5 muM) or monensin (3.5 muM) for 5 h prior to fixing. Staining for golgin-97 or APRIL is shown in red, DNA staining with SybrGreen in green. Asterisks indicate non-transfected cells serving as negative controls. Bar, 10 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
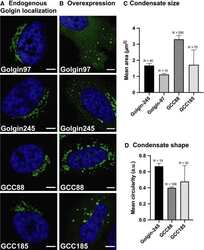
- Experimental details
- Fig. 2 Overexpressed trans-golgins form condensates. (A) HeLa cells were fixed and immunostained for endogenous golgins or (B) HeLa cells were transfected with golgin97-FLAG/Myc, golgin245-FLAG/Myc, GCC88-FLAG/Myc, or GCC185-FLAG/Myc, respectively, fixed 6 h post-transfection and immunostained for FLAG (exogenous golgin). Secondary antibody conjugated to Alexa Fluor 488 was used in (A) and (B). Each overexpressed golgin formed characteristic condensates. (C) Mean condensate area and (D) mean condensate circularity, where N denotes the number of condensates analyzed and error bars denote the standard error of the mean (SEM). The mean area (and SEM) was calculated based on condensates with area >0.8 um 2 , thereby excluding contributions from random fluctuations in protein concentration or fluorescence signal. The mean circularity (and SEM) was calculated by including condensates with area >1.5 um 2 because circularity became ill-defined for smaller particles. Golgin97 was excluded for the circularity analysis because too few condensates satisfied these criteria for accurate statistics. Scale bars: 5 um.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot