Antibody data
- Antibody Data
- Antigen structure
- References [4]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 39-1000 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- PPAP2B Monoclonal Antibody (ZV002)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- ZV002
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
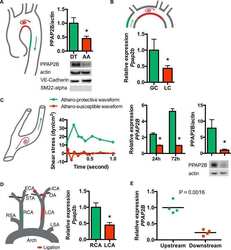
Submitted references Endothelial lipid phosphate phosphatase-3 deficiency that disrupts the endothelial barrier function is a modifier of cardiovascular development.
Mechanosensitive PPAP2B Regulates Endothelial Responses to Atherorelevant Hemodynamic Forces.
Lipid phosphate phosphatase-3 regulates tumor growth via β-catenin and CYCLIN-D1 signaling.
Lipid phosphate phosphatase 3 stabilization of beta-catenin induces endothelial cell migration and formation of branching point structures.
Chatterjee I, Baruah J, Lurie EE, Wary KK
Cardiovascular research 2016 Jul 1;111(1):105-18
Cardiovascular research 2016 Jul 1;111(1):105-18
Mechanosensitive PPAP2B Regulates Endothelial Responses to Atherorelevant Hemodynamic Forces.
Wu C, Huang RT, Kuo CH, Kumar S, Kim CW, Lin YC, Chen YJ, Birukova A, Birukov KG, Dulin NO, Civelek M, Lusis AJ, Loyer X, Tedgui A, Dai G, Jo H, Fang Y
Circulation research 2015 Jul 31;117(4):e41-e53
Circulation research 2015 Jul 31;117(4):e41-e53
Lipid phosphate phosphatase-3 regulates tumor growth via β-catenin and CYCLIN-D1 signaling.
Chatterjee I, Humtsoe JO, Kohler EE, Sorio C, Wary KK
Molecular cancer 2011 May 11;10:51
Molecular cancer 2011 May 11;10:51
Lipid phosphate phosphatase 3 stabilization of beta-catenin induces endothelial cell migration and formation of branching point structures.
Humtsoe JO, Liu M, Malik AB, Wary KK
Molecular and cellular biology 2010 Apr;30(7):1593-606
Molecular and cellular biology 2010 Apr;30(7):1593-606
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of PPAP2B was performed using 70% confluent log phase HeLa cells treated with 50 ng/mL EGF for 12 h. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with PPAP2B (ZV002) Mouse Monoclonal Antibody (Product # 39-1000) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing punctate cytoplasmic localization. Panel e shows the untreated cells. Panel f shows the control without primary antibody. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
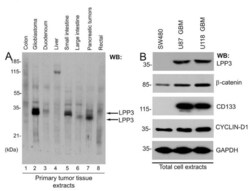
- Figure 1 Expression of LPP3 in primary tumors . (A) Western blot was probed with an anti-LPP3-C-cyto antibody. Each lane contained 10 mug total protein prepared from primary human tumor tissues. LPP3 protein is indicated by two black arrows. (B) Tumor line cell extracts were analyzed by WB with the indicated antibodies. All blots shown are representative of those obtained from at least three separate experiments with similar results.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
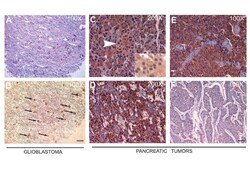
- Figure 2 Immunohistochemistry of primary tumors . (A) A representative serial section of human glioblastoma was stained with hematoxylin and eosin (H&E), white arrows indicate microvascular proliferation, and (B) black arrows indicate anti-LPP3-RGD positive glioblastoma cells. Scale bars, 100 mum. (C) Anti-LPP3 staining in normal pancreas show cytosolic expression in both endocrine (Islet of Langerhans-arrow) and exocrine components. (D, E) In pancreatic endocrine tumors, 65% of the tumors show cytosolic staining. Inset: Nuclear localization of LPP3 is also detected in 40% of cases. (F) An example of LPP3-negative tumor cells. All experiments are representative of those obtained in at least three separate experiments, with similar results.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
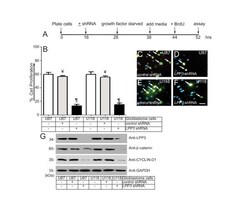
- Figure 3 LPP3 regulates glioblastoma cell proliferation . (A) The time line indicates when glioblastoma cell lines were plated, shRNA (retrovirus) mediated knockdown was conducted, cells were growth factor and serum starved, fresh medium was added, BrdU (1.0 mug/ml) was added, and the proliferation assay was performed. (B) Quantification of glioblastoma cell proliferation assay. The data represent the mean +- s.e.m. n = 5-7 from five to seven independent experiments, Y= P < 0.05 vs. control untreated; P P < 0.05 vs. control shRNA treated group. (C-F) Representative images of the BrdU assay. Arrows indicate BrdU positive cells. Scale bar, 150 mum. (G) The efficiency of knockdown of LPP3 was assessed by western blotting with the indicated antibodies. Figure 3G (top panel, last lane, anti-LPP3), a partial band is likely produced by over flow from the neighboring well. All experiments are representative of those obtained in at least three separate experiments with similar results.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
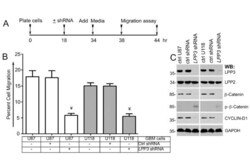
- Experimental details
- Figure 4 LPP3 regulates glioblastoma cell migration . (A) The time line indicates when glioblastoma cell lines were plated, shRNA (retrovirus) mediated knockdown was conducted, medium was added, and the migration assay was performed as described in materials and methods. (B) Quantification of glioblastoma cell migration through the Boyden chamber. The data represent the mean +- s.e.m. n = 5-7, Y= P < 0.05 vs. control shRNA treated group. (C) Efficiency of knockdown of LPP3 was assessed by western blotting with the indicated antibodies. All experiments are representative of those obtained in at least three separate experiments with similar results.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
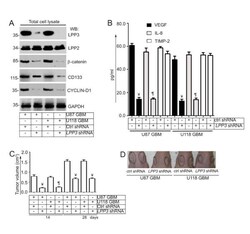
- Experimental details
- Figure 5 LPP3 regulates glioblastoma tumor growth . (A) Stable clones of U87 and U118 were selected and efficiency of knockdown was determined by western blotting with the indicated antibodies. LPP3 -knockdown decreased total beta-catenin, CYCLIN-D1, and CD133 proteins, whereas it had no effect on LPP2 or GAPDH. (B) Conditioned media were collected and subjected to ELISA for VEGF, IL-8, and TIMP-2. LPP3 -knockdown in U87 and U118 cells reduced the concentration of VEGF and IL-8, whereas TIMP-2 was unaffected. (C) In the xenograft assay, mice receiving cells that expressed control shRNA showed increased tumor growth (n = 12, Y= P < 0.05) after 14 and 28 days. LPP3 -knockdown reduced U87 and U118 tumor growth (n = 12, P P < 0.05). (D) Representative images from the xenograft assay. Dotted circles indicate the location of tumor implants.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 LPP3- knockdown results in increased vascular endothelial cell permeability. ( A ) Timeline of the knockdown, beta-catenin cDNA transfection, and TER assay. ( B ) LPP3-depletion results in decreased TER and increased LMVECs barrier disruption, whereas no change was observed in LMVECs receiving control shRNA . Following transfection of beta-catenin cDNA into LPP3-depleted LMVECs, the TER level was restored nearly to control group, while the transfection with p120-catenin cDNA into LPP3-depleted LMVECs had only a modest effect. Experiments were carried out in triplicates and repeated at least three times; * P < 0.05. ( C ) The efficiency of Lpp3 -knockdown was assessed by WB with the indicated antibodies. Experiments were repeated at least three times with similar results.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry