Antibody data
- Antibody Data
- Antigen structure
- References [38]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [4]
- Immunohistochemistry [1]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-16705 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Desmin Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 1 mL
- Concentration
- 0.25 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references μ-Crystallin in Mouse Skeletal Muscle Promotes a Shift from Glycolytic toward Oxidative Metabolism.
Microtubules orchestrate local translation to enable cardiac growth.
Young and Undamaged rMSA Improves the Healthspan and Lifespan of Mice.
Premature termination codon readthrough upregulates progranulin expression and improves lysosomal function in preclinical models of GRN deficiency.
Functional skeletal muscle model derived from SOD1-mutant ALS patient iPSCs recapitulates hallmarks of disease progression.
Heterogenetic parabiosis between healthy and dystrophic mice improve the histopathology in muscular dystrophy.
Splice modulating antisense oligonucleotides restore some acid-alpha-glucosidase activity in cells derived from patients with late-onset Pompe disease.
Accumulation of myeloid lineage cells is mapping out liver fibrosis post injury: a targetable lesion using Ketanserin.
TGF-β1 affects cell-cell adhesion in the heart in an NCAM1-dependent mechanism.
Mesothelin/mucin 16 signaling in activated portal fibroblasts regulates cholestatic liver fibrosis.
Sodium 4-phenylbutyrate reduces myofiber damage in a mouse model of Duchenne muscular dystrophy.
Tumour resistance in induced pluripotent stem cells derived from naked mole-rats.
Towards clinical application of pronuclear transfer to prevent mitochondrial DNA disease.
Hepatocellular carcinoma originates from hepatocytes and not from the progenitor/biliary compartment.
Reconstruction of hepatic stellate cell-incorporated liver capillary structures in small hepatocyte tri-culture using microporous membranes.
Diagnostic pitfalls of differentiating desmoplastic small round cell tumor (DSRCT) from Wilms tumor (WT): overlapping morphologic and immunohistochemical features.
Anti-interleukin-6 receptor antibody (MR16-1) promotes muscle regeneration via modulation of gene expressions in infiltrated macrophages.
Endothelial-specific Notch blockade inhibits vascular function and tumor growth through an eNOS-dependent mechanism.
Midkine-deficient mice delayed degeneration and regeneration after skeletal muscle injury.
Mesodermal mesenchymal cells give rise to myofibroblasts, but not epithelial cells, in mouse liver injury.
Netrin-4 promotes mural cell adhesion and recruitment to endothelial cells.
Solitary tumours associated with Jaagsiekte retrovirus in sheep are heterogeneous and contain cells expressing markers identifying progenitor cells in lung repair.
Analysis of transplanted bone marrow-derived cells in chronic pancreatitis.
Citral is renoprotective for focal segmental glomerulosclerosis by inhibiting oxidative stress and apoptosis and activating Nrf2 pathway in mice.
Downregulation of microRNA-9 in iPSC-derived neurons of FTD/ALS patients with TDP-43 mutations.
Fate tracing reveals hepatic stellate cells as dominant contributors to liver fibrosis independent of its aetiology.
Modeling key pathological features of frontotemporal dementia with C9ORF72 repeat expansion in iPSC-derived human neurons.
The impact of KRAS mutations on VEGF-A production and tumour vascular network.
Evidence for a common progenitor of epithelial and mesenchymal components of the liver.
Lactic acid bacteria convert human fibroblasts to multipotent cells.
HSCs play a distinct role in different phases of oval cell-mediated liver regeneration.
Induced pluripotent stem cell models of progranulin-deficient frontotemporal dementia uncover specific reversible neuronal defects.
Oral administration of recombinant adeno-associated virus-mediated bone morphogenetic protein-7 suppresses CCl(4)-induced hepatic fibrosis in mice.
Alterations in intermediate filaments expression in disc cells from the rat temporomandibular joint following exposure to continuous compressive force.
Lymphoid tissue damage in HIV-1 infection depletes naïve T cells and limits T cell reconstitution after antiretroviral therapy.
Rosmarinic acid and baicalin epigenetically derepress peroxisomal proliferator-activated receptor γ in hepatic stellate cells for their antifibrotic effect.
Sporadic haemangioblastoma of the kidney with rhabdoid features and focal CD10 expression: report of a case and literature review.
Gene expression during normal and FSHD myogenesis.
Kinney CJ, O'Neill A, Noland K, Huang W, Muriel J, Lukyanenko V, Kane MA, Ward CW, Collier AF, Roche JA, McLenithan JC, Reed PW, Bloch RJ
Current research in physiology 2021;4:47-59
Current research in physiology 2021;4:47-59
Microtubules orchestrate local translation to enable cardiac growth.
Scarborough EA, Uchida K, Vogel M, Erlitzki N, Iyer M, Phyo SA, Bogush A, Kehat I, Prosser BL
Nature communications 2021 Mar 11;12(1):1547
Nature communications 2021 Mar 11;12(1):1547
Young and Undamaged rMSA Improves the Healthspan and Lifespan of Mice.
Tang J, Ju A, Li B, Zhang S, Gong Y, Ma B, Jiang Y, Liu H, Fu Y, Luo Y
Biomolecules 2021 Aug 12;11(8)
Biomolecules 2021 Aug 12;11(8)
Premature termination codon readthrough upregulates progranulin expression and improves lysosomal function in preclinical models of GRN deficiency.
Frew J, Baradaran-Heravi A, Balgi AD, Wu X, Yan TD, Arns S, Shidmoossavee FS, Tan J, Jaquith JB, Jansen-West KR, Lynn FC, Gao FB, Petrucelli L, Feldman HH, Mackenzie IR, Roberge M, Nygaard HB
Molecular neurodegeneration 2020 Mar 16;15(1):21
Molecular neurodegeneration 2020 Mar 16;15(1):21
Functional skeletal muscle model derived from SOD1-mutant ALS patient iPSCs recapitulates hallmarks of disease progression.
Badu-Mensah A, Guo X, McAleer CW, Rumsey JW, Hickman JJ
Scientific reports 2020 Aug 31;10(1):14302
Scientific reports 2020 Aug 31;10(1):14302
Heterogenetic parabiosis between healthy and dystrophic mice improve the histopathology in muscular dystrophy.
Lu A, Guo P, Wang L, Tseng C, Huard M, Allen C, McCarrick-Walmsley R, Whitney KE, Huard J
Scientific reports 2020 Apr 27;10(1):7075
Scientific reports 2020 Apr 27;10(1):7075
Splice modulating antisense oligonucleotides restore some acid-alpha-glucosidase activity in cells derived from patients with late-onset Pompe disease.
Aung-Htut MT, Ham KA, Tchan M, Johnsen R, Schnell FJ, Fletcher S, Wilton SD
Scientific reports 2020 Apr 21;10(1):6702
Scientific reports 2020 Apr 21;10(1):6702
Accumulation of myeloid lineage cells is mapping out liver fibrosis post injury: a targetable lesion using Ketanserin.
Amini-Nik S, Sadri AR, Diao L, Belo C, Jeschke MG
Experimental & molecular medicine 2018 Jul 19;50(7):1-13
Experimental & molecular medicine 2018 Jul 19;50(7):1-13
TGF-β1 affects cell-cell adhesion in the heart in an NCAM1-dependent mechanism.
Ackermann MA, Petrosino JM, Manring HR, Wright P, Shettigar V, Kilic A, Janssen PML, Ziolo MT, Accornero F
Journal of molecular and cellular cardiology 2017 Nov;112:49-57
Journal of molecular and cellular cardiology 2017 Nov;112:49-57
Mesothelin/mucin 16 signaling in activated portal fibroblasts regulates cholestatic liver fibrosis.
Koyama Y, Wang P, Liang S, Iwaisako K, Liu X, Xu J, Zhang M, Sun M, Cong M, Karin D, Taura K, Benner C, Heinz S, Bera T, Brenner DA, Kisseleva T
The Journal of clinical investigation 2017 Apr 3;127(4):1254-1270
The Journal of clinical investigation 2017 Apr 3;127(4):1254-1270
Sodium 4-phenylbutyrate reduces myofiber damage in a mouse model of Duchenne muscular dystrophy.
Begam M, Abro VM, Mueller AL, Roche JA
Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabolisme 2016 Oct;41(10):1108-1111
Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabolisme 2016 Oct;41(10):1108-1111
Tumour resistance in induced pluripotent stem cells derived from naked mole-rats.
Miyawaki S, Kawamura Y, Oiwa Y, Shimizu A, Hachiya T, Bono H, Koya I, Okada Y, Kimura T, Tsuchiya Y, Suzuki S, Onishi N, Kuzumaki N, Matsuzaki Y, Narita M, Ikeda E, Okanoya K, Seino K, Saya H, Okano H, Miura K
Nature communications 2016 May 10;7:11471
Nature communications 2016 May 10;7:11471
Towards clinical application of pronuclear transfer to prevent mitochondrial DNA disease.
Hyslop LA, Blakeley P, Craven L, Richardson J, Fogarty NM, Fragouli E, Lamb M, Wamaitha SE, Prathalingam N, Zhang Q, O'Keefe H, Takeda Y, Arizzi L, Alfarawati S, Tuppen HA, Irving L, Kalleas D, Choudhary M, Wells D, Murdoch AP, Turnbull DM, Niakan KK, Herbert M
Nature 2016 Jun 16;534(7607):383-6
Nature 2016 Jun 16;534(7607):383-6
Hepatocellular carcinoma originates from hepatocytes and not from the progenitor/biliary compartment.
Mu X, Español-Suñer R, Mederacke I, Affò S, Manco R, Sempoux C, Lemaigre FP, Adili A, Yuan D, Weber A, Unger K, Heikenwälder M, Leclercq IA, Schwabe RF
The Journal of clinical investigation 2015 Oct 1;125(10):3891-903
The Journal of clinical investigation 2015 Oct 1;125(10):3891-903
Reconstruction of hepatic stellate cell-incorporated liver capillary structures in small hepatocyte tri-culture using microporous membranes.
Kasuya J, Sudo R, Masuda G, Mitaka T, Ikeda M, Tanishita K
Journal of tissue engineering and regenerative medicine 2015 Mar;9(3):247-56
Journal of tissue engineering and regenerative medicine 2015 Mar;9(3):247-56
Diagnostic pitfalls of differentiating desmoplastic small round cell tumor (DSRCT) from Wilms tumor (WT): overlapping morphologic and immunohistochemical features.
Arnold MA, Schoenfield L, Limketkai BN, Arnold CA
The American journal of surgical pathology 2014 Sep;38(9):1220-6
The American journal of surgical pathology 2014 Sep;38(9):1220-6
Anti-interleukin-6 receptor antibody (MR16-1) promotes muscle regeneration via modulation of gene expressions in infiltrated macrophages.
Fujita R, Kawano F, Ohira T, Nakai N, Shibaguchi T, Nishimoto N, Ohira Y
Biochimica et biophysica acta 2014 Oct;1840(10):3170-80
Biochimica et biophysica acta 2014 Oct;1840(10):3170-80
Endothelial-specific Notch blockade inhibits vascular function and tumor growth through an eNOS-dependent mechanism.
Patenaude A, Fuller M, Chang L, Wong F, Paliouras G, Shaw R, Kyle AH, Umlandt P, Baker JH, Diaz E, Tong J, Minchinton AI, Karsan A
Cancer research 2014 May 1;74(9):2402-11
Cancer research 2014 May 1;74(9):2402-11
Midkine-deficient mice delayed degeneration and regeneration after skeletal muscle injury.
Ikutomo M, Sakakima H, Matsuda F, Yoshida Y
Acta histochemica 2014 Mar;116(2):319-26
Acta histochemica 2014 Mar;116(2):319-26
Mesodermal mesenchymal cells give rise to myofibroblasts, but not epithelial cells, in mouse liver injury.
Lua I, James D, Wang J, Wang KS, Asahina K
Hepatology (Baltimore, Md.) 2014 Jul;60(1):311-22
Hepatology (Baltimore, Md.) 2014 Jul;60(1):311-22
Netrin-4 promotes mural cell adhesion and recruitment to endothelial cells.
Lejmi E, Bouras I, Camelo S, Roumieux M, Minet N, Leré-Déan C, Merkulova-Rainon T, Autret G, Vayssettes C, Clement O, Plouët J, Leconte L
Vascular cell 2014 Jan 28;6(1):1
Vascular cell 2014 Jan 28;6(1):1
Solitary tumours associated with Jaagsiekte retrovirus in sheep are heterogeneous and contain cells expressing markers identifying progenitor cells in lung repair.
De las Heras M, de Martino A, Borobia M, Ortín A, Álvarez R, Borderías L, Giménez-Más JA
Journal of comparative pathology 2014 Feb-Apr;150(2-3):138-47
Journal of comparative pathology 2014 Feb-Apr;150(2-3):138-47
Analysis of transplanted bone marrow-derived cells in chronic pancreatitis.
Westphalen CB, Marrache F, Wang TC
Methods in molecular biology (Clifton, N.J.) 2013;980:291-300
Methods in molecular biology (Clifton, N.J.) 2013;980:291-300
Citral is renoprotective for focal segmental glomerulosclerosis by inhibiting oxidative stress and apoptosis and activating Nrf2 pathway in mice.
Yang SM, Hua KF, Lin YC, Chen A, Chang JM, Kuoping Chao L, Ho CL, Ka SM
PloS one 2013;8(9):e74871
PloS one 2013;8(9):e74871
Downregulation of microRNA-9 in iPSC-derived neurons of FTD/ALS patients with TDP-43 mutations.
Zhang Z, Almeida S, Lu Y, Nishimura AL, Peng L, Sun D, Wu B, Karydas AM, Tartaglia MC, Fong JC, Miller BL, Farese RV Jr, Moore MJ, Shaw CE, Gao FB
PloS one 2013;8(10):e76055
PloS one 2013;8(10):e76055
Fate tracing reveals hepatic stellate cells as dominant contributors to liver fibrosis independent of its aetiology.
Mederacke I, Hsu CC, Troeger JS, Huebener P, Mu X, Dapito DH, Pradere JP, Schwabe RF
Nature communications 2013;4:2823
Nature communications 2013;4:2823
Modeling key pathological features of frontotemporal dementia with C9ORF72 repeat expansion in iPSC-derived human neurons.
Almeida S, Gascon E, Tran H, Chou HJ, Gendron TF, Degroot S, Tapper AR, Sellier C, Charlet-Berguerand N, Karydas A, Seeley WW, Boxer AL, Petrucelli L, Miller BL, Gao FB
Acta neuropathologica 2013 Sep;126(3):385-99
Acta neuropathologica 2013 Sep;126(3):385-99
The impact of KRAS mutations on VEGF-A production and tumour vascular network.
Figueras A, Arbos MA, Quiles MT, Viñals F, Germà JR, Capellà G
BMC cancer 2013 Mar 18;13:125
BMC cancer 2013 Mar 18;13:125
Evidence for a common progenitor of epithelial and mesenchymal components of the liver.
Conigliaro A, Amicone L, Costa V, De Santis Puzzonia M, Mancone C, Sacchetti B, Cicchini C, Garibaldi F, Brenner DA, Kisseleva T, Bianco P, Tripodi M
Cell death and differentiation 2013 Aug;20(8):1116-23
Cell death and differentiation 2013 Aug;20(8):1116-23
Lactic acid bacteria convert human fibroblasts to multipotent cells.
Ohta K, Kawano R, Ito N
PloS one 2012;7(12):e51866
PloS one 2012;7(12):e51866
HSCs play a distinct role in different phases of oval cell-mediated liver regeneration.
Chen L, Zhang W, Zhou QD, Yang HQ, Liang HF, Zhang BX, Long X, Chen XP
Cell biochemistry and function 2012 Oct;30(7):588-96
Cell biochemistry and function 2012 Oct;30(7):588-96
Induced pluripotent stem cell models of progranulin-deficient frontotemporal dementia uncover specific reversible neuronal defects.
Almeida S, Zhang Z, Coppola G, Mao W, Futai K, Karydas A, Geschwind MD, Tartaglia MC, Gao F, Gianni D, Sena-Esteves M, Geschwind DH, Miller BL, Farese RV Jr, Gao FB
Cell reports 2012 Oct 25;2(4):789-98
Cell reports 2012 Oct 25;2(4):789-98
Oral administration of recombinant adeno-associated virus-mediated bone morphogenetic protein-7 suppresses CCl(4)-induced hepatic fibrosis in mice.
Hao ZM, Cai M, Lv YF, Huang YH, Li HH
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Nov;20(11):2043-51
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Nov;20(11):2043-51
Alterations in intermediate filaments expression in disc cells from the rat temporomandibular joint following exposure to continuous compressive force.
Magara J, Nozawa-Inoue K, Suzuki A, Kawano Y, Ono K, Nomura S, Maeda T
Journal of anatomy 2012 Jun;220(6):612-21
Journal of anatomy 2012 Jun;220(6):612-21
Lymphoid tissue damage in HIV-1 infection depletes naïve T cells and limits T cell reconstitution after antiretroviral therapy.
Zeng M, Southern PJ, Reilly CS, Beilman GJ, Chipman JG, Schacker TW, Haase AT
PLoS pathogens 2012 Jan;8(1):e1002437
PLoS pathogens 2012 Jan;8(1):e1002437
Rosmarinic acid and baicalin epigenetically derepress peroxisomal proliferator-activated receptor γ in hepatic stellate cells for their antifibrotic effect.
Yang MD, Chiang YM, Higashiyama R, Asahina K, Mann DA, Mann J, Wang CC, Tsukamoto H
Hepatology (Baltimore, Md.) 2012 Apr;55(4):1271-81
Hepatology (Baltimore, Md.) 2012 Apr;55(4):1271-81
Sporadic haemangioblastoma of the kidney with rhabdoid features and focal CD10 expression: report of a case and literature review.
Yin WH, Li J, Chan JK
Diagnostic pathology 2012 Apr 12;7:39
Diagnostic pathology 2012 Apr 12;7:39
Gene expression during normal and FSHD myogenesis.
Tsumagari K, Chang SC, Lacey M, Baribault C, Chittur SV, Sowden J, Tawil R, Crawford GE, Ehrlich M
BMC medical genomics 2011 Sep 27;4:67
BMC medical genomics 2011 Sep 27;4:67
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot of Desmin using Desmin Polyclonal Antibody (Product # PA5-16705) on SW156 Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Desmin Polyclonal Antibody (Product # PA5-16705) and a 55 kDa bands corresponding to Desmin was observed in Mouse Skeletal Muscle, Rat Skeletal Muscle, Mouse Heart, Rat Heart, Cardiomyocytes and RD cell line but not in Mouse Kidney and MCF7. An isoform at 45 kDa was also observed in Cardiomyocyte and RD cell line. An uncharacterized band of ~60 kDa was also observed in Cardiomyocytes. Tissue extracts (30 µg lysate) of Mouse Skeletal Muscle (Lane 1), Rat Skeletal Muscle (Lane 2), Mouse Heart (Lane 3), Rat Heart (Lane 4), Mouse Kidney (Lane 6) and Whole cell extracts (30 µg lysate) of Cardiomyocytes (Lane 5), RD (Lane 7) and MCF7 (Lane 8) were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:4000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
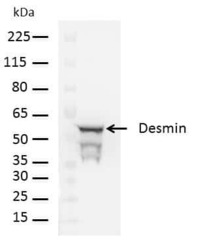
- Experimental details
- Western blot analysis of Desmin was performed by loading 20 µg of human hepatic stellate cell lysate and 10 µL of Molecular Weight Protein Ladder (Product # 26634) per well onto a Novex Bolt 4-12% Bis-Tris plus polyacrylamide gel (NW04125). Proteins were transferred to a PVDF membrane using iBlot 2 transfer device (Product # IB21001), and blocked with 5% non-fat dry milk in TBS+0.1%Tween-20 for one hour at room temperature. Desmin was detected at 53 kDa using Desmin polyclonal antibody (Product # PA5-16705) at a dilution of 1:5000 at 4oC overnight on a rocking platform, followed by a goat anti-rabbit HRP conjugated antibody (Product # A24537) at a dilution of 1:7000 for one hour. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Desmin (Green) in primary human hepatic stellate cells. The cells were fixed with 1% buffered formaldehyde for 15 minutes, permeabilized with 0.1% TritonX-100 in DPBS for 15 minutes, and blocked with blocking buffer (1% BSA+10% Goat serum+0.3M Glycine in DPBS) for 1 hour at room temperature. Cells were stained overnight with anti-Desmin polyclonal antibody (Product # PA5-16705) at a dilution of 1:100 at 4oC and then incubated with alexa fluor 488 labeled secondary antibody (Product # A-11034) at a dilution of 1:1000 for 1 hour at room temperature. Cells were counterstained with ProLong® Gold antifade mountant with DAPI (Product # P36941) and imaged.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Desmin (Green) in primary human hepatic stellate cells. The cells were fixed with 1% buffered formaldehyde for 15 minutes, permeabilized with 0.1% TritonX-100 in DPBS for 15 minutes, and blocked with blocking buffer (1% BSA+10% Goat serum+0.3M Glycine in DPBS) for 1 hour at room temperature. Cells were stained overnight with anti-Desmin polyclonal antibody (Product # PA5-16705) at a dilution of 1:100 at 4oC and then incubated with alexa fluor 488 labeled secondary antibody (Product # A-11034) at a dilution of 1:1000 for 1 hour at room temperature. Cells were counterstained with ProLong® Gold antifade mountant with DAPI (Product # P36941) and imaged.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis Desmin was performed using 70% confluent log phase RD cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with Desmin Polyclonal Antibody (Product PA5-16705) at 1:100 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then with Goat anti-Rabbit IgG (H+L), Superclonal™ Recombinant Secondary Antibody, Alexa Fluor 488 (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoskeletal localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Desmin (Green) in primary human hepatic stellate cells. The cells were fixed with 1% buffered formaldehyde for 15 minutes, permeabilized with 0.1% TritonX-100 in DPBS for 15 minutes, and blocked with blocking buffer (1% BSA+10% Goat serum+0.3M Glycine in DPBS) for 1 hour at room temperature. Cells were stained overnight with anti-Desmin polyclonal antibody (Product # PA5-16705) at a dilution of 1:100 at 4oC and then incubated with alexa fluor 488 labeled secondary antibody (Product # A-11034) at a dilution of 1:1000 for 1 hour at room temperature. Cells were counterstained with ProLong® Gold antifade mountant with DAPI (Product # P36941) and imaged.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Formalin-fixed, paraffin-embedded human leiomyoma stained with Desmin using peroxidase-conjugate and AEC. Note cytoplasmic staining of tumor cells
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
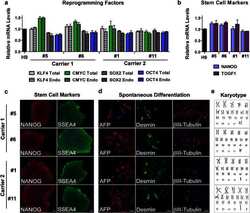
- Experimental details
- Fig. 1 Generation and characterization of iPSC lines from carriers of C9ORF72 expanded repeats. Total and endogenous (Endo) mRNA levels of the reprogramming factors KLF4, CMYC, SOX2, and OCT4 in iPSC lines from carriers 1 and 2 relative to the values in human embryonic stem cell line H9 were assessed by qRT-PCR. Values are mean +- SEM ( a ). The expression of the pluripotency markers NANOG and TDGF1 (or CRIPTO) was measured at the mRNA level. The values from H9 cells were set to 1. Values are mean +- SEM ( b ). Immunofluorescence analysis of pluripotency markers NANOG and SSEA4 in iPSC lines from carriers 1 and 2 is shown and cell nuclei were counterstained with Hoechst ( blue ). Scale bar 50 mum ( c ). After in vitro spontaneous differentiation of iPSC lines into cells of three embryonic germ layers, cells were immunostained with alpha-fetoprotein (AFP, endoderm), desmin (mesoderm), betaIII-tubulin (ectoderm), and Hoechst (nuclei). Scale bar 50 mum ( d ). All iPSC lines maintained a normal karyotype ( e )
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
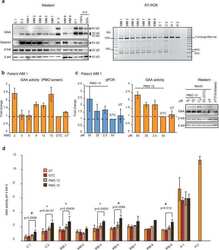
- Experimental details
- Figure 3 The efficiencies of selective PMOs in myogenic cells derived from the patient after forced myogenesis. ( a ) Western analysis of muscle-specific protein desmin and RT-PCR analysis of GAA transcript (exon 1 to 5) in patients'' and healthy controls'' fibroblasts forced into the myogenic lineage by infection with MyoD expressing virus. beta tub; beta-tubulin, beta act; beta-actin. ( b ) GAA activity of patient WM 1 forced myogenic cells after treatment with various PMOs. Data represent the mean +- SDs of triplicates. ( c ) qPCR analysis of GAA transcript with exon 2 (the mean +- SDs of triplicates), GAA activity (the mean +- SDs of triplicates) and GAA protein in patient WM 1 forced myogenic cells after treatment with various dosages of PMO 15, Gene Tools Control (GTC) and untreated (UT). ( d ) Comparison of GAA activity in nine patients'' and two healthy controls'' (H 1 and H 2) forced myogenic cell cultures after treatment with PMO 13, 15 and GTC at 50 uM and untreated (UT) samples. Data represent the mean +- SDs of at least three biological replicates (n >= 3) for patient C 1, C 2, WM 1, 2, 4, 5, 7, and 9. The difference of the means, compared to GTC is significant at *p < 0.05 and # p < 0.1 using ANOVA with Bonferroni correction. No biological replicates were performed for WM 8. The gels were cropped for presentation, and full-size gels are presented in Supplementary Fig. S5 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
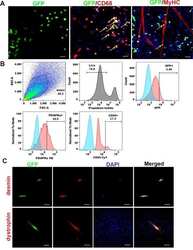
- Experimental details
- Figure 5 Identification of GFP + cells in the mdx muscle in vitro . ( A ) Representative image of GFP positive cells isolated from mdx mice paired with GFP mice (GFP, green). Yellow arrows indicate GFP + cells that are colocalized with CD68 (red), and not colocalized with MyHC (red). The nuclei were stained with DAPI (blue). ( B ) Flow cytometric analyses of the GFP + circulating cells isolated from the muscle of parabiont mdx mic e . ( C ) Representative images show the desmin staining (top, red) and the colocalization with GFP (green). The myotubes were stained with dystrophin (red, bottom) after 3 days differentiation. Representative images show the colocalization with GFP (green). The nuclei were stained with DAPI (blue). In panel A, scale bar = 50 um for GFP, scale bar = 25 um for CD68 and MyHC. In panel B, scale bar = 25 um for desmin, scale bar = 50 um for dystrophin).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
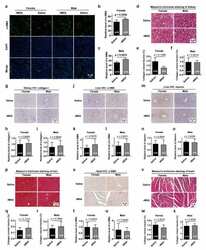
- Figure 5 Effects of rMSA injection on the fibrosis level of kidney, liver and heart ( a ) The representative images of alpha-SMA in mice kidney. Scale bar, 50 mum. ( b , c ) The quantitative results of alpha-SMA level in female ( b ) and male ( c ) mice. ( d ) The Masson's trichrome staining of mice kidney. Scale bar, 50 mum. ( e , f ) The collagen volume fraction of the kidney in female ( e ) and male ( f ) mice. ( g ) The immunohistochemical staining for collagen I in mice kidney. Scale bar, 50 mum. ( h , i ) The relative level of collagen I in female ( h ) and male ( i ) mice. ( j ) The immunohistochemical staining for alpha-SMA in mice liver. Scale bar, 50 mum. ( k , l ) The relative level of alpha-SMA in female ( k ) and male ( l ) mice. ( m ) The immunohistochemical staining for desmin in mice liver. Scale bar, 50 mum. ( n , o ) The relative level of desmin in female ( n ) and male ( o ) mice. ( p ) The Masson's trichrome staining of mice liver. Scale bar, 50 mum. ( q , r ) The collagen volume fraction of the liver in female ( q ) and male ( r ) mice. ( s ) The immunohistochemical staining for alpha-SMA in mice heart. Scale bar, 50 mum. ( t , u ) The relative level of alpha-SMA in female ( t ) and male ( u ) mice. ( v ) The Masson's trichrome staining of mice cardiac muscle. Scale bar, 50 mum. ( w , x ) The collagen volume fraction of the cardiac muscle in female ( w ) and male ( x ) mice. All graphs represent mean with SEM, with p values calculated by the two-tail t te
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
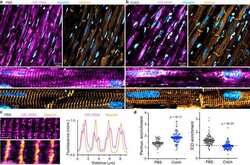
- Fig. 5 Microtubules control the localization of ribosomes in vivo. a Representative image of PBS-injected rat ventricular myocardium, with zoomed-in image below and white arrow highlighting ICD region. b Representative image of Colch-injected rat ventricular myocardium with similar zoom and highlight as in a . c (left) Zoomed-in image of PBS-injected rat ventricular myocardium. (right) Line scan analysis of the composite image shown at bottom left. d Nuclear:cytosolic and ICD:cytosolic ratios of 18S rRNA mean fluorescence intensities of experiment depicted in a and b . PBS ( N = 2), ( n = 71, N/C), ( n = 137, ICD/C), Colch ( N = 2), ( n = 92, N/C), ( n = 108, ICD/C). Statistical significance determined via two sample, two-tailed t -test. For all graphs shown in figure, the mean line is shown, with whiskers denoting standard error (SE) from the mean. Source data are provided as a Source data file.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
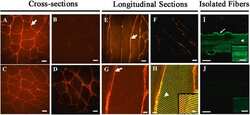
- Fig. 2 Immunolabeling of mu-crystallin in control and Crym tg skeletal muscles. A-D, Cross (A-D), longitudinal sections (E-H), of Crym tg (A, C, E, G, H) and control (B, D, F) TA (A, B, E-H) and soleus muscles (C, D) were stained with anti-mu-crystallin antibody and Alexa Fluor-568-conjugated secondary antibody. In G-H, longitudinal sections of Crym tg TA muscle (G, mu-crystallin only) were colabeled with anti-desmin and Alexa Fluor-488-conjugated secondary antibody (H, yellow color shows colabeled structures). I, J, Flexor digitorum brevis (FDB) myofibers in culture from Crym tg (I) and control mice (J) were labeled with anti-mu-crystallin antibody and Alexa-Fluor 488-conjugated secondary antibody. The results show that mu-crystallin was detected at higher levels in tg muscles than in controls. In TA muscle and FDB myofibers it was enriched at the levels of the sarcolemma (arrows, A,E,G, I) and Z-disks, colabeled with desmin (arrowhead, H) or shown without desmin colabel (arrowhead, I). Inset panels (H-J) are brightened and magnified. Crym tg (I) and control (J) FDB images were brightened and magnified equivalently. A-F, scale bars = 20 mum; G, H, scale bars = 5 mum; I, J, scale bars = 10 mum. Fig. 2
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
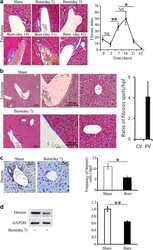
- Fig. 1 Thermal injury primes liver fibrosis in a spatiotemporal pattern . a There is a temporal trend in fibrosis around PVs post-thermal injury. Mice were examined after 2, 7, 14, 21, and 42 days post-thermal injury; n = 5-7, scale bar 50 mum, * P < 0.05, ** P < 0.001. NS not significant. b Trichrome staining shows that collagen is deposited predominately around PVs rather than CVs. c-d Thermal injury promotes a reduction in desmin+ cells n = 3-5
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
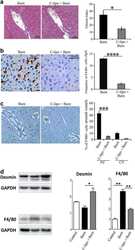
- Experimental details
- Fig. 3 Myeloid cells have an essential role in the induction of fibrosis post-thermal injury . a Mice treated with liposomal clodronate compared with control treated group. Trichrome staining shows less fibrosis around portal venules post-thermal injury when myeloid cells are depleted; n = 9, scale bar 50 mum. b Immunohistochemistry shows a highly effective ablation of F4/80+ cells in liposomal clodronate-treated group in comparison to the control group; n = 9, scale bar 50 mum. c There is a global ablation of F4/80+ cells in the liver of mice treated with liposomal clodronate, scale bar 100 mum. d In contrast to earlier findings, without F4/80+ cells, there is an increase in desmin+ cells post-thermal injury; n = 3. * P < 0.05, ** P < 0.01, *** P < 0.001, **** P < 0.0001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
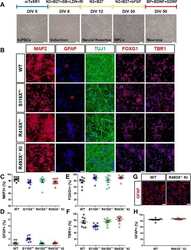
- Experimental details
- Fig. 2 Differentiation and characterization of human cortical neurons and astrocytes derived from FTD- GRN patient and control hiPSCs. a Brightfield images of the cells at different stages of cortical neuronal differentiation. SB = SB 431542 , LDN = LDN 193189, RI = Y-27632, BP = BrainPhys(tm), N2 = N-2 supplement, B27 = B-27 supplement. b Representative immunofluorescence images of DIV 50 WT, S116X +/- , R418X +/- , and R493X -/- KI hiPSC-derived cortical neurons stained for MAP2, GFAP, TUJ1, FOXG1 (forebrain), and TBR1 (cortical layer VI). Cell nuclei were counterstained with DAPI (blue). Scale bar, 50 mum. c - f Cells positive for MAP2, GFAP, FOXG1, and TBR1 (neuronal, astrocytes, forebrain, and cortical layer VI markers, respectively) as a percentage of DAPI + cells. On average, ~ 900 cells were analyzed per replicate, in ( c - f ) n = 3 independent cultures, 3 images per biological replicate; values are shown as mean +- SEM. g Representative immunofluorescence images of DIV 60+ WT and R493X -/- KI hiPSC-derived astrocytes stained for GFAP. Cell nuclei were counterstained with DAPI (blue). Scale bar, 50 mum. h Cells positive for GFAP as a percentage of DAPI + cells. On average, ~ 400 cells were analyzed per replicate, n = 3 independent cultures, 3 images per biological replicate; values are shown as mean +- SEM
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot