Antibody data
- Antibody Data
- Antigen structure
- References [26]
- Comments [0]
- Validations
- Other assay [12]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-1979-82 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD197 (CCR7) Monoclonal Antibody (3D12), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 3D12 monoclonal antibody reacts with human CCR7, also known as EBI-1 and CD197. CCR7 is a member of the G-protein-coupled chemokine receptor family with seven membrane-spanning domains and functions as a receptor for 6Ckine/SLC (secondary lymphoid-tissue chemokine), CCL19 and CCL21. CCR7 has been shown to be internalized via clathrin-coated pits and the majority recycled back to the plasma membrane. CCR7 is expressed on T cells and can be used to distinguish populations of naive from central and effector memory T cells. CCR7 has been shown to play a role in migration of memory T cells to inflamed tissue. Expression of CCR7 is also found on DC's. During DC maturation CCR7 expression increases and is thought to be involved in a variety of functions: chemotaxis to the lymph node, cellular architecture, rate of endocytosis, survival and maturation. Expression of CCR7 on the cell surface can be down regulated upon ligand binding. Applications Reported: This 3D12 antibody has been reported for use in flow cytometric analysis and immunohistology staining of paraffin embedded tissue sections. Applications Tested: This 3D12 antibody has been tested by flow cytometric analysis of human peripheral blood cells. This can be used at less than or equal to 0.5 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. It is recommended that the staining incubation time be increased to at least 45 minutes at 2-8°C for optimal staining. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- 3D12
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C
Submitted references Alteration of the Immune Microenvironment in HBsAg and HBeAg Dual-Positive Pregnant Women Presenting a High HBV Viral Load.
Neuroimmune Consequences of eIF4E Phosphorylation on Chemotherapy-Induced Peripheral Neuropathy.
Application of the chemokine-chemokine receptor axis increases the tumor-targeted migration ability of cytokine-induced killer cells in patients with colorectal cancer.
The Diagnostic Value of Chemokine/Chemokine Receptor Pairs in Hepatocellular Carcinoma and Colorectal Liver Metastasis.
Intracellular delivery of mRNA to human primary T cells with microfluidic vortex shedding.
Boiling Histotripsy-induced Partial Mechanical Ablation Modulates Tumour Microenvironment by Promoting Immunogenic Cell Death of Cancers.
The Calcineurin Inhibitor Tacrolimus Specifically Suppresses Human T Follicular Helper Cells.
Biochanin a Enhances the Defense Against Salmonella enterica Infection Through AMPK/ULK1/mTOR-Mediated Autophagy and Extracellular Traps and Reversing SPI-1-Dependent Macrophage (MΦ) M2 Polarization.
Characteristic patterns of HLA presentation and T cell differentiation in adult-onset Still's disease.
Oral Supplementation with Baker's Yeast Beta Glucan Is Associated with Altered Monocytes, T Cells and Cytokines following a Bout of Strenuous Exercise.
Monocyte differentiation and macrophage priming are regulated differentially by pentraxins and their ligands.
Efficient generation of antigen-specific CTLs by the BAFF-activated human B Lymphocytes as APCs: a novel approach for immunotherapy.
FOXP3+ Regulatory T Cells in Hepatic Fibrosis and Splenomegaly Caused by Schistosoma japonicum: The Spleen May Be a Major Source of Tregs in Subjects with Splenomegaly.
Brief Report: IFIH1 Mutation Causes Systemic Lupus Erythematosus With Selective IgA Deficiency.
Innate immune responses and rapid control of inflammation in African green monkeys treated or not with interferon-alpha during primary SIVagm infection.
Prevailing role of contact guidance in intrastromal T-cell trapping in human pancreatic cancer.
Secondary lymphoid organ homing phenotype of human myeloid dendritic cells disrupted by an intracellular oral pathogen.
Peptide-induced immune regulation by a promiscuous and immunodominant CD4T-cell epitope of Timothy grass pollen: a role of Cbl-b and Itch in regulation.
Kinetics of myeloid dendritic cell trafficking and activation: impact on progressive, nonprogressive and controlled SIV infections.
Helios+ and Helios- cells coexist within the natural FOXP3+ T regulatory cell subset in humans.
The effect of pregnancy on the uterine NK cell KIR repertoire.
IL-15 transpresentation promotes both human T-cell reconstitution and T-cell-dependent antibody responses in vivo.
CD137 ligand signaling induces human monocyte to dendritic cell differentiation.
Role of TLX1 in T-cell acute lymphoblastic leukaemia pathogenesis.
Proliferation and differentiation potential of human CD8+ memory T-cell subsets in response to antigen or homeostatic cytokines.
Two subsets of memory T lymphocytes with distinct homing potentials and effector functions.
Gao F, Wang H, Li X, Guo F, Yuan Y, Wang X, Zhang Y, Bai G
Journal of inflammation research 2021;14:5619-5632
Journal of inflammation research 2021;14:5619-5632
Neuroimmune Consequences of eIF4E Phosphorylation on Chemotherapy-Induced Peripheral Neuropathy.
Agalave NM, Mody PH, Szabo-Pardi TA, Jeong HS, Burton MD
Frontiers in immunology 2021;12:642420
Frontiers in immunology 2021;12:642420
Application of the chemokine-chemokine receptor axis increases the tumor-targeted migration ability of cytokine-induced killer cells in patients with colorectal cancer.
Zou Y, Liang J, Li D, Fang J, Wang L, Wang J, Zhang J, Guo Q, Yan X, Tang H
Oncology letters 2020 Jul;20(1):123-134
Oncology letters 2020 Jul;20(1):123-134
The Diagnostic Value of Chemokine/Chemokine Receptor Pairs in Hepatocellular Carcinoma and Colorectal Liver Metastasis.
Jiao X, Shu G, Liu H, Zhang Q, Ma Z, Ren C, Guo H, Shi J, Liu J, Zhang C, Wang Y, Gao Y
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2019 May;67(5):299-308
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2019 May;67(5):299-308
Intracellular delivery of mRNA to human primary T cells with microfluidic vortex shedding.
Jarrell JA, Twite AA, Lau KHWJ, Kashani MN, Lievano AA, Acevedo J, Priest C, Nieva J, Gottlieb D, Pawell RS
Scientific reports 2019 Mar 1;9(1):3214
Scientific reports 2019 Mar 1;9(1):3214
Boiling Histotripsy-induced Partial Mechanical Ablation Modulates Tumour Microenvironment by Promoting Immunogenic Cell Death of Cancers.
Pahk KJ, Shin CH, Bae IY, Yang Y, Kim SH, Pahk K, Kim H, Oh SJ
Scientific reports 2019 Jun 21;9(1):9050
Scientific reports 2019 Jun 21;9(1):9050
The Calcineurin Inhibitor Tacrolimus Specifically Suppresses Human T Follicular Helper Cells.
Wallin EF, Hill DL, Linterman MA, Wood KJ
Frontiers in immunology 2018;9:1184
Frontiers in immunology 2018;9:1184
Biochanin a Enhances the Defense Against Salmonella enterica Infection Through AMPK/ULK1/mTOR-Mediated Autophagy and Extracellular Traps and Reversing SPI-1-Dependent Macrophage (MΦ) M2 Polarization.
Zhao X, Tang X, Guo N, An Y, Chen X, Shi C, Wang C, Li Y, Li S, Xu H, Liu M, Wang Y, Yu L
Frontiers in cellular and infection microbiology 2018;8:318
Frontiers in cellular and infection microbiology 2018;8:318
Characteristic patterns of HLA presentation and T cell differentiation in adult-onset Still's disease.
Jung JY, Choi B, Sayeed HM, Suh CH, Kim YW, Kim HA, Sohn S
International journal of immunopathology and pharmacology 2018 Jan-Dec;32:2058738418791284
International journal of immunopathology and pharmacology 2018 Jan-Dec;32:2058738418791284
Oral Supplementation with Baker's Yeast Beta Glucan Is Associated with Altered Monocytes, T Cells and Cytokines following a Bout of Strenuous Exercise.
McFarlin BK, Venable AS, Carpenter KC, Henning AL, Ogenstad S
Frontiers in physiology 2017;8:786
Frontiers in physiology 2017;8:786
Monocyte differentiation and macrophage priming are regulated differentially by pentraxins and their ligands.
Pilling D, Galvis-Carvajal E, Karhadkar TR, Cox N, Gomer RH
BMC immunology 2017 Jun 15;18(1):30
BMC immunology 2017 Jun 15;18(1):30
Efficient generation of antigen-specific CTLs by the BAFF-activated human B Lymphocytes as APCs: a novel approach for immunotherapy.
Yiwen Z, Shilin G, Yingshi C, Lishi S, Baohong L, Chao L, Linghua L, Ting P, Hui Z
Oncotarget 2016 Nov 22;7(47):77732-77748
Oncotarget 2016 Nov 22;7(47):77732-77748
FOXP3+ Regulatory T Cells in Hepatic Fibrosis and Splenomegaly Caused by Schistosoma japonicum: The Spleen May Be a Major Source of Tregs in Subjects with Splenomegaly.
Romano A, Hou X, Sertorio M, Dessein H, Cabantous S, Oliveira P, Li J, Oyegue S, Arnaud V, Luo X, Daujat-Chavanieu M, Mariani O, Sastre X, Dombey AM, He H, Li Y, Dessein A
PLoS neglected tropical diseases 2016 Jan;10(1):e0004306
PLoS neglected tropical diseases 2016 Jan;10(1):e0004306
Brief Report: IFIH1 Mutation Causes Systemic Lupus Erythematosus With Selective IgA Deficiency.
Van Eyck L, De Somer L, Pombal D, Bornschein S, Frans G, Humblet-Baron S, Moens L, de Zegher F, Bossuyt X, Wouters C, Liston A
Arthritis & rheumatology (Hoboken, N.J.) 2015 Jun;67(6):1592-7
Arthritis & rheumatology (Hoboken, N.J.) 2015 Jun;67(6):1592-7
Innate immune responses and rapid control of inflammation in African green monkeys treated or not with interferon-alpha during primary SIVagm infection.
Jacquelin B, Petitjean G, Kunkel D, Liovat AS, Jochems SP, Rogers KA, Ploquin MJ, Madec Y, Barré-Sinoussi F, Dereuddre-Bosquet N, Lebon P, Le Grand R, Villinger F, Müller-Trutwin M
PLoS pathogens 2014 Jul;10(7):e1004241
PLoS pathogens 2014 Jul;10(7):e1004241
Prevailing role of contact guidance in intrastromal T-cell trapping in human pancreatic cancer.
Hartmann N, Giese NA, Giese T, Poschke I, Offringa R, Werner J, Ryschich E
Clinical cancer research : an official journal of the American Association for Cancer Research 2014 Jul 1;20(13):3422-33
Clinical cancer research : an official journal of the American Association for Cancer Research 2014 Jul 1;20(13):3422-33
Secondary lymphoid organ homing phenotype of human myeloid dendritic cells disrupted by an intracellular oral pathogen.
Miles B, Zakhary I, El-Awady A, Scisci E, Carrion J, O'Neill JC, Rawlings A, Stern JK, Susin C, Cutler CW
Infection and immunity 2014 Jan;82(1):101-11
Infection and immunity 2014 Jan;82(1):101-11
Peptide-induced immune regulation by a promiscuous and immunodominant CD4T-cell epitope of Timothy grass pollen: a role of Cbl-b and Itch in regulation.
Till SJ, Raynsford EJ, Reynolds CJ, Quigley KJ, Grzybowska-Kowalczyk A, Saggar LR, Goldstone A, Maillere B, Kwok WW, Altmann DM, Durham SR, Boyton RJ
Thorax 2014 Apr;69(4):335-45
Thorax 2014 Apr;69(4):335-45
Kinetics of myeloid dendritic cell trafficking and activation: impact on progressive, nonprogressive and controlled SIV infections.
Wijewardana V, Kristoff J, Xu C, Ma D, Haret-Richter G, Stock JL, Policicchio BB, Mobley AD, Nusbaum R, Aamer H, Trichel A, Ribeiro RM, Apetrei C, Pandrea I
PLoS pathogens 2013;9(10):e1003600
PLoS pathogens 2013;9(10):e1003600
Helios+ and Helios- cells coexist within the natural FOXP3+ T regulatory cell subset in humans.
Himmel ME, MacDonald KG, Garcia RV, Steiner TS, Levings MK
Journal of immunology (Baltimore, Md. : 1950) 2013 Mar 1;190(5):2001-8
Journal of immunology (Baltimore, Md. : 1950) 2013 Mar 1;190(5):2001-8
The effect of pregnancy on the uterine NK cell KIR repertoire.
Male V, Sharkey A, Masters L, Kennedy PR, Farrell LE, Moffett A
European journal of immunology 2011 Oct;41(10):3017-27
European journal of immunology 2011 Oct;41(10):3017-27
IL-15 transpresentation promotes both human T-cell reconstitution and T-cell-dependent antibody responses in vivo.
Huntington ND, Alves NL, Legrand N, Lim A, Strick-Marchand H, Mention JJ, Plet A, Weijer K, Jacques Y, Becker PD, Guzman C, Soussan P, Kremsdorf D, Spits H, Di Santo JP
Proceedings of the National Academy of Sciences of the United States of America 2011 Apr 12;108(15):6217-22
Proceedings of the National Academy of Sciences of the United States of America 2011 Apr 12;108(15):6217-22
CD137 ligand signaling induces human monocyte to dendritic cell differentiation.
Kwajah M M S, Schwarz H
European journal of immunology 2010 Jul;40(7):1938-49
European journal of immunology 2010 Jul;40(7):1938-49
Role of TLX1 in T-cell acute lymphoblastic leukaemia pathogenesis.
Riz I, Hawley TS, Johnston H, Hawley RG
British journal of haematology 2009 Apr;145(1):140-3
British journal of haematology 2009 Apr;145(1):140-3
Proliferation and differentiation potential of human CD8+ memory T-cell subsets in response to antigen or homeostatic cytokines.
Geginat J, Lanzavecchia A, Sallusto F
Blood 2003 Jun 1;101(11):4260-6
Blood 2003 Jun 1;101(11):4260-6
Two subsets of memory T lymphocytes with distinct homing potentials and effector functions.
Sallusto F, Lenig D, Förster R, Lipp M, Lanzavecchia A
Nature 1999 Oct 14;401(6754):708-12
Nature 1999 Oct 14;401(6754):708-12
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 T cell activation marker expression is unaffected in cells processed via uVS . Flow cytometry was used to quantify the surface level expression of key T cell activation markers (CD69, CD154, CD44, CCR7, CD45RA and CD25) in T cells processed with uVS (processed cells, red dashed) compared to non-processed cells (handling control, blue solid) 24 hours post transfection. Histogram overlays represent expression levels as a percent of maximum value of each marker for a representative sample for each condition. Experiment was performed in triplicate.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Expression levels of chemokine receptors on CIK cells generated from patients with CRC and healthy donors. (A) Analysis of the expression levels of CCR4, CCR5, CCR7, CXCR3 and CXCR4 on CIK cells via flow cytometry revealed that CXCR3 and CXCR4 had higher expression levels on CIK cells compared with isotype, expression profiles of CCR4, CCR5 and CCR7 did not show significant changes compared with isotype. (B) Dynamic changes of chemokine receptors CCR4, CCR5, CCR7, CXCR3 and CXCR4 of CIK cells on D7, 14, 21 and 28 were detected in patients with CRC and HD CIK cells via flow cytometry. The result revealed that expression levels of CXCR3 and CXCR4 were significantly higher on CIK cells cultured from CRC compared with HD at all the time points analyzed, while the expression levels of CCR4, CCR5 and CCR7 were significantly higher on CIK cells cultured from CRC compared with HD at different time points analyzed (CCR4 on D7 and D28; CCR5 on D28; CCR7 on D7, D21 and D28). All comparisons were performed using unpaired Student''s t-test with Welch''s correction. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Paclitaxel dysregulates CD4 + and CD8 + T-cell subpopulations in an eIF4E-dependent manner. (A) Gating strategy used for flow cytometry of lymphocytes from popliteal and inguinal lymph nodes. T-cells were separated from the whole lymphoid cells population using CD3. CD3 + cells were then gated for CD4 + T-cell and CD8 + T-cells. For each of these subsets, cells were further gated for CCR7 or CD25 and CD44. (B) Quantification of CD4 + T-cells from the CD3 population from male mice. (C) CCR7 + T-cells from isolated CD4 + population in males, ** p 0.0063. (D) CD4 + cells gated for CD25 and/or CD44 in males. (E) Quantification of CD4 + T-cells from the CD3 + population from female mice, * p 0.0101. (F) CCR7 + T-cells from isolated CD4 + population in female mice, * p 0.0250. (G) CD4 + cells gated for CD25 and/or CD44 in female mice, ** p 0.0011. (H) Quantification of CD8 + T-cells from the CD3 + population from male mice. (I) CCR7 + T-cells from isolated CD8 + population in males, * p 0.0433. (J) CD8 + cells gated for CD25 and/or CD44 in males, * p 0.0396. (K) Quantification of CD8 + T-cells from the CD3 population from female mice, * p 0.041. (L) CCR7 + T-cells from isolated CD8 + population in female mice, ** p 0.0097. (M) CD8 + cells gated for CD25 and/or CD44 in female mice. All data are presented as mean +- standard error of the mean, n = 3 mice each for male/female WT vehicle-treated and WT paclitaxel groups, n = 4 mice each for male/female eIF4E S209A vehicle-trea
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Effect of BYBG on CD4+ T cells. CD4+ T cell concentrations were determined using peripheral blood mononuclear cells (PBMC) isolated from human whole blood samples. PBMCs were analyzed using flow cytometry to determine total ( A , CD3+/CD4+), T EM ( B , CCR7-/CD45RA-), and T CM ( C , CCR7+/CD45RA-) concentrations. Placebo (black bars) and BYBG (green bars) are represented separately and were statistically compared. Values represent the mean +- SEM. * indicates BYBG significantly different than placebo ( p < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
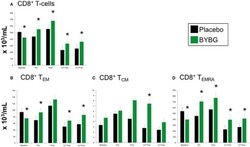
- Figure 6 Effect of BYBG on CD8+ T cells. CD8+ T cell concentrations were determined using peripheral blood mononuclear cells (PBMC) isolated from human whole blood samples. PBMCs were analyzed using flow cytometry to determine total ( A , CD3+/CD8+), T EM ( B , CCR7-/CD45RA-), and T CM ( C , CCR7+/CD45RA-), and T EMRA ( D , CCR7-/CD45RA+) concentrations. Placebo (black bars) and BYBG (green bars) are represented separately and were statistically compared. Values represent the mean +- SEM. * indicates BYBG significantly different than placebo ( p < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 HBV-related samples display higher proportion of effector/memory CD8+ T cells by flow cytometry assay. ( A ) Examples of CD3, CD4 and CCR7 staining to determine effector/memory CD4+ T cells. ( B ) Examples of CD3, CD8 and CCR7 staining to determine effector/memory CD8+ T cells. ( C ) The average proportion of effector/memory CD4+ T cells and CD8+ T cells in HBV and control groups (*p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. Percentage of surface-stained cells presenting CD4+, CD8+, CCR7+, CD4+CCR7+, CD8+CCR7+, CD4+CD62L-, CD8+CD62L-, CD4+naive T cell, CD4+ effector memory T cell, CD4+ central memory T cells, CD8+ naive T cells, CD8+ effector memory T cells, and CD8+ central memory T cells in patients with AOSD, a patient with rheumatoid arthritis (RA), and a healthy control (HC). Results were obtained from 14 patients with AOSD, 20 RA patients, and 20 HCs. The horizontal line indicates the mean value for each group. The P value was determined by the Mann-Whitney U test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Percentage of surface-stained cells presenting HLA-DQ on (a) monocytes, (b) granulocytes, (c) CD8+CCR7+T cells, and (d) CD4+ central memory T cells according to disease activity in patients with AOSD. The results were obtained from six samples from AOSD patients when disease are active, untreated, and after their disease activity resolved. The horizontal line indicates the mean value for each group. The P value was determined with the Wilcoxon signed-rank test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 BCA regulate MPhi polarization in vitro and in vivo . (A,B) Raw246.7 cells were treated with 4 mug/ml BCA for 2 h. Then, the cells were infected with Salmonella at MOI = 10:1 for 2 h. (A) The expression of surface markers (CCR7, CD86, CD163, and CD206) was determined by flow cytometry. The results are presented as MFI. (B) TNF-alpha and IL-10 in the supernatants were detected by ELISA. ** P < 0.01, *** P < 0.001. The data are representative of three experiments with similar results. (C,D) Mice were infected by intragastric administration of an overnight culture of Salmonella (10 5 bacteria in 0.1 ml PBS) through a gavage tube and then treated with 6.25 mg/kg BCA intragastrically by gavage daily (5 mice for each group). On the 5th day p.i., the mice were sacrificed and the peritoneal fluid was collected. (C) The expression of surface markers (CCR7, CD86, CD163, and CD206) in mouse peritoneal MPhis were determined by flow cytometry The results are presented as MFI. (D) TNF-alpha and IL-10 in ascitic fluid were detected by ELISA. * P < 0.05, ** P < 0.01, *** P < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 2 Knockdown of TLX1 by shRNA results in changes in the expression of CD1b, CD55 and CCR7 cell surface molecules on TLX1 + T-ALL cell lines. (A) ALL-SIL and K3P cells expressing the 95 TLX1 shRNA (red histograms) or the pLKO.1-puro lentiviral vector backbone (blue histograms) were analysed by flow cytometry after staining with anti-CD1b-Alexa Fluor 647 (Cat. no. 51-0018), anti-CD55-PE (Cat. no. 12-0559) or anti-CCR7-fluorescein isothiocyanate (FITC) (Cat no. 11-1979) monoclonal antibodies (all from eBioscience, Inc., San Diego, CA). Green histograms represent unstained cell populations. Viable cells were gated by a combination of forward and orthogonal light scatter, and data was acquired on a FACSAria instrument (BD Biosciences) and analysed using WinList software (Verity Software House, Topsham, ME). Note the bimodal CD55 expression pattern in pLKO.1-puro-transduced ALL-SIL cells. Whereas CD1b and CD55 exhibited expression patterns following TLX1 knockdown consistent with a partial release of the early DP differentiation block, CCR7, the receptor for the medullary chemokines CCL19 and CCL21, is not normally expressed in early cortical thymocytes and represents a potential TLX1 target gene involved in T-ALL pathogenesis (see Table SII). (B) ALL-SIL cells expressing the 95 TLX1 shRNA were viably sorted into CD1b hi CD55 lo and CD1b lo CD55 hi subpopulations. Whole cell extracts were prepared from aliquots of the sorted cells and TLX1 protein levels were analysed by Western blo
 Explore
Explore Validate
Validate Learn
Learn Immunohistochemistry
Immunohistochemistry Other assay
Other assay