Antibody data
- Antibody Data
- Antigen structure
- References [125]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [2]
- Other assay [81]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 71-7800 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Claudin 1 Polyclonal Antibody (MH25)
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- This antibody reacts with the ~ 22 kDa Claudin-1 protein. Reactivity was confirmed by western blotting and immunofluorescence. Positive controls include MDCK cells (dog), Caco-2cells (human), mouse liver, rat liver, and rat kidney.
- Antibody clone number
- MH25
- Concentration
- 0.25 mg/mL
Submitted references TMAO reductase, a biomarker for gut permeability defect induced inflammation, in mouse model of chronic kidney disease and dextran sulfate solution-induced mucositis.
The protective effect and potential mechanisms of eugenol against Salmonella in vivo and in vitro.
Disruption of the crypt niche promotes outgrowth of mutated colorectal tumor stem cells.
SARS-CoV-2-Induced Pathology-Relevance to COVID-19 Pathophysiology.
Phiclust: a clusterability measure for single-cell transcriptomics reveals phenotypic subpopulations.
Isolation of DiNP-Degrading Microbes from the Mouse Colon and the Influence DiNP Exposure Has on the Microbiota, Intestinal Integrity, and Immune Status of the Colon.
DevKidCC allows for robust classification and direct comparisons of kidney organoid datasets.
Dietary branched-chain amino acids modulate the dynamics of calcium absorption and reabsorption in protein-restricted pigs.
Tackling recalcitrant Pseudomonas aeruginosa infections in critical illness via anti-virulence monotherapy.
Matrix Metalloproteinase MMP-12 Promotes Macrophage Transmigration Across Intestinal Epithelial Tight Junctions and Increases Severity of Experimental Colitis.
Organic osmolytes increase expression of specific tight junction proteins in skin and alter barrier function in keratinocytes.
Fenofibrate promotes PPARα-targeted recovery of the intestinal epithelial barrier at the host-microbe interface in dogs with diabetes mellitus.
Cystine reduces tight junction permeability and intestinal inflammation induced by oxidative stress in Caco-2 cells.
Probiotics mixture reinforces barrier function to ameliorate necrotizing enterocolitis by regulating PXR-JNK pathway.
Tight Junction Protein Claudin-7 Is Essential for Intestinal Epithelial Stem Cell Self-Renewal and Differentiation.
Novel Human NKCC1 Mutations Cause Defects in Goblet Cell Mucus Secretion and Chronic Inflammation.
Astragalus mongholicus Bunge and Panax Notoginseng Formula (A&P) Combined With Bifidobacterium Contribute a Renoprotective Effect in Chronic Kidney Disease Through Inhibiting Macrophage Inflammatory Response in Kidney and Intestine.
The Histone Demethylase JMJD1C Regulates CAMKK2-AMPK Signaling to Participate in Cardiac Hypertrophy.
Impaired Airway Epithelial Barrier Integrity in Response to Stenotrophomonas maltophilia Proteases, Novel Insights Using Cystic Fibrosis Bronchial Epithelial Cell Secretomics.
oprC Impairs Host Defense by Increasing the Quorum-Sensing-Mediated Virulence of Pseudomonas aeruginosa.
Intestinal vitamin D receptor signaling ameliorates dextran sulfate sodium-induced colitis by suppressing necroptosis of intestinal epithelial cells.
Tissue-scale tensional homeostasis in skin regulates structure and physiological function.
Phosphorylation and dephosphorylation of Ser852 and Ser889 control the clustering, localization and function of PAR3.
Toxicological effects of bioactive peptide fractions obtained from Bothrops jararaca snake venom on the structure and function of mouse seminiferous epithelium.
Intestinal vitamin D receptor knockout protects from oxazolone-induced colitis.
Regulatory Effect of Lactobacillus brevis Bmb6 on Gut Barrier Functions in Experimental Colitis.
RAS-mediated suppression of PAR3 and its effects on SCC initiation and tissue architecture occur independently of hyperplasia.
Matriptase Cleaves EpCAM and TROP2 in Keratinocytes, Destabilizing Both Proteins and Associated Claudins.
Staphylococcus aureus Internalized by Skin Keratinocytes Evade Antibiotic Killing.
Anthocyanins protect the gastrointestinal tract from high fat diet-induced alterations in redox signaling, barrier integrity and dysbiosis.
Probiotics Prevents Sensitization to Oral Antigen and Subsequent Increases in Intestinal Tight Junction Permeability in Juvenile-Young Adult Rats.
The Differentiation-Associated Keratinocyte Protein Cornifelin Contributes to Cell-Cell Adhesion of Epidermal and Mucosal Keratinocytes.
Newly synthesized claudins but not occludin are added to the basal side of the tight junction.
Protease-Activated Receptors 2-Antagonist Suppresses Asthma by Inhibiting Reactive Oxygen Species-Thymic Stromal Lymphopoietin Inflammation and Epithelial Tight Junction Degradation.
Rescue of tight junctional localization of a claudin-16 mutant D97S by antimalarial medicine primaquine in Madin-Darby canine kidney cells.
Geniposide and Chlorogenic Acid Combination Ameliorates Non-alcoholic Steatohepatitis Involving the Protection on the Gut Barrier Function in Mouse Induced by High-Fat Diet.
Vitamin D Receptor Deletion Leads to the Destruction of Tight and Adherens Junctions in Lungs.
HPV16-E6 Oncoprotein Activates TGF-β and Wnt/β-Catenin Pathways in the Epithelium-Mesenchymal Transition of Cataracts in a Transgenic Mouse Model.
Esrp1-Regulated Splicing of Arhgef11 Isoforms Is Required for Epithelial Tight Junction Integrity.
Perineurial-like Cells and EMA Expression in the Suprachoroidal Region of the Human Eye.
Analysis of the bystander effect in cone photoreceptors via a guided neural network platform.
VPS33B and VIPAR are essential for epidermal lamellar body biogenesis and function.
Adherens junctions influence tight junction formation via changes in membrane lipid composition.
Enteropathogenic E. coli effectors EspF and Map independently disrupt tight junctions through distinct mechanisms involving transcriptional and post-transcriptional regulation.
Establishing normal metabolism and differentiation in hepatocellular carcinoma cells by culturing in adult human serum.
(-)-Epicatechin protects the intestinal barrier from high fat diet-induced permeabilization: Implications for steatosis and insulin resistance.
Inflammatory cytokines down-regulate the barrier-protective prostasin-matriptase proteolytic cascade early in experimental colitis.
TRPV4 Regulates Tight Junctions and Affects Differentiation in a Cell Culture Model of the Corneal Epithelium.
Obesity-induces Organ and Tissue Specific Tight Junction Restructuring and Barrier Deregulation by Claudin Switching.
Epithelial response to a high-protein diet in rat colon.
Down-regulation of Claudin-2 Expression and Proliferation by Epigenetic Inhibitors in Human Lung Adenocarcinoma A549 Cells.
Matriptase-mediated cleavage of EpCAM destabilizes claudins and dysregulates intestinal epithelial homeostasis.
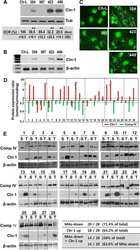
Downregulation of lipolysis-stimulated lipoprotein receptor promotes cell invasion via claudin-1-mediated matrix metalloproteinases in human endometrial cancer.
Lactate-mediated mitoribosomal defects impair mitochondrial oxidative phosphorylation and promote hepatoma cell invasiveness.
Serglycin in tumor microenvironment promotes non-small cell lung cancer aggressiveness in a CD44-dependent manner.
T-Lymphocytes Traffic into the Brain across the Blood-CSF Barrier: Evidence Using a Reconstituted Choroid Plexus Epithelium.
Urinary Podocyte Loss Is Increased in Patients with Fabry Disease and Correlates with Clinical Severity of Fabry Nephropathy.
Liver kinase B1 regulates hepatocellular tight junction distribution and function in vivo.
HOXA5 determines cell fate transition and impedes tumor initiation and progression in breast cancer through regulation of E-cadherin and CD24.
Dual effects of a high-protein diet on DSS-treated mice during colitis resolution phase.
Hypotonic Stress-induced Down-regulation of Claudin-1 and -2 Mediated by Dephosphorylation and Clathrin-dependent Endocytosis in Renal Tubular Epithelial Cells.
Regulation of post-Golgi LH3 trafficking is essential for collagen homeostasis.
Maintaining physical activity during refeeding improves body composition, intestinal hyperpermeability and behavior in anorectic mice.
Altered Prostasin (CAP1/Prss8) Expression Favors Inflammation and Tissue Remodeling in DSS-induced Colitis.
Salmonella enteritidis Effector AvrA Stabilizes Intestinal Tight Junctions via the JNK Pathway.
Planar cell polarity signaling in the uterus directs appropriate positioning of the crypt for embryo implantation.
Combined Treatment with Epigenetic, Differentiating, and Chemotherapeutic Agents Cooperatively Targets Tumor-Initiating Cells in Triple-Negative Breast Cancer.
Proliferation of cultured mouse choroid plexus epithelial cells.
Defining a conformational consensus motif in cotransin-sensitive signal sequences: a proteomic and site-directed mutagenesis study.
The impact of CLAUDIN-1 on follicular thyroid carcinoma aggressiveness.
The characterization of the human nasal epithelial cell line RPMI 2650 under different culture conditions and their optimization for an appropriate in vitro nasal model.
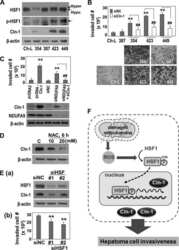
Mitochondrial Respiratory Dysfunction Induces Claudin-1 Expression via Reactive Oxygen Species-mediated Heat Shock Factor 1 Activation, Leading to Hepatoma Cell Invasiveness.
Toll-like receptor 2 regulates the barrier function of human bronchial epithelial monolayers through atypical protein kinase C zeta, and an increase in expression of claudin-1.
Evidence for a role of claudin 2 as a proximal tubular stress responsive paracellular water channel.
Alteration of intestinal barrier function during activity-based anorexia in mice.
The reversible increase in tight junction permeability induced by capsaicin is mediated via cofilin-actin cytoskeletal dynamics and decreased level of occludin.
Nf1 loss and Ras hyperactivation in oligodendrocytes induce NOS-driven defects in myelin and vasculature.
Papillomavirus E6 oncoprotein up-regulates occludin and ZO-2 expression in ovariectomized mice epidermis.
A bradykinin-potentiating peptide (BPP-10c) from Bothrops jararaca induces changes in seminiferous tubules.
Contribution of tight junction proteins to ion, macromolecule, and water barrier in keratinocytes.
Expression of claudins -2 and -4 and cingulin is coordinated with the start of stratification and differentiation in corneal epithelial cells: retinoic acid reversibly disrupts epithelial barrier.
GLP-2 enhances barrier formation and attenuates TNFα-induced changes in a Caco-2 cell model of the intestinal barrier.
Oxidative stress induced by potassium bromate exposure results in altered tight junction protein expression in renal proximal tubule cells.
Cingulin is dispensable for epithelial barrier function and tight junction structure, and plays a role in the control of claudin-2 expression and response to duodenal mucosa injury.
Differential effects of flavonoids on barrier integrity in human intestinal Caco-2 cells.
Functional characterization and localization of a gill-specific claudin isoform in Atlantic salmon.
Tight junction proteins expression and modulation in immune cells and multiple sclerosis.
EGFR regulation of epidermal barrier function.
Expression of claudin-1 and -11 in immature and mature pheasant (Phasianus colchicus) testes.
CD44 regulates tight-junction assembly and barrier function.
The potency of the fs260 connexin43 mutant to impair keratinocyte differentiation is distinct from other disease-linked connexin43 mutants.
Increased intraocular insulin-like growth factor-I triggers blood-retinal barrier breakdown.
Claudin expression modulations reflect an injury response in the murine epidermis.
Changes in the distribution pattern of Claudin tight junction proteins during the progression of mouse skin tumorigenesis.
Astrovirus increases epithelial barrier permeability independently of viral replication.
Claudin immunolocalization in neonatal mouse epithelial tissues.
Dynamic changes in the cervical epithelial tight junction complex and differentiation occur during cervical ripening and parturition.
Proteomic and bioinformatic analysis of epithelial tight junction reveals an unexpected cluster of synaptic molecules.
Leukocyte diapedesis in vivo induces transient loss of tight junction protein at the blood-retina barrier.
Simultaneous cell death and desquamation of the embryonic diffusion barrier during epidermal development.
Expression and function of tight junctions in the crypt epithelium of human palatine tonsils.
Connexin 26-mediated gap junctional intercellular communication suppresses paracellular permeability of human intestinal epithelial cell monolayers.
Interferon-gamma down-regulates claudin-1 and impairs the epithelial barrier function in primary cultured human thyrocytes.
Interferon-gamma down-regulates claudin-1 and impairs the epithelial barrier function in primary cultured human thyrocytes.
Simian virus 40 small tumor antigen induces deregulation of the actin cytoskeleton and tight junctions in kidney epithelial cells.
Localization of claudin-3 in tight junctions of the blood-brain barrier is selectively lost during experimental autoimmune encephalomyelitis and human glioblastoma multiforme.
Protection against hypoxia-induced increase in blood-brain barrier permeability: role of tight junction proteins and NFkappaB.
Protection against hypoxia-induced increase in blood-brain barrier permeability: role of tight junction proteins and NFkappaB.
SRC-induced disintegration of adherens junctions of madin-darby canine kidney cells is dependent on endocytosis of cadherin and antagonized by Tiam-1.
SRC-induced disintegration of adherens junctions of madin-darby canine kidney cells is dependent on endocytosis of cadherin and antagonized by Tiam-1.
Proinflammatory cytokines disrupt epithelial barrier function by apoptosis-independent mechanisms.
Proinflammatory cytokines disrupt epithelial barrier function by apoptosis-independent mechanisms.
2,3-butanedione monoxime (BDM), a potent inhibitor of actin-myosin interaction, induces ion and fluid transport in MDCK monolayers.
2,3-butanedione monoxime (BDM), a potent inhibitor of actin-myosin interaction, induces ion and fluid transport in MDCK monolayers.
Protein phosphatase 2A associates with and regulates atypical PKC and the epithelial tight junction complex.
E-Cadherin and tight junctions between epithelial cells of different animal species.
Tight junctions are sensitive to peptides eliminated in the urine.
Neutrophil transepithelial migration: evidence for sequential, contact-dependent signaling events and enhanced paracellular permeability independent of transjunctional migration.
Neutrophil transepithelial migration: evidence for sequential, contact-dependent signaling events and enhanced paracellular permeability independent of transjunctional migration.
JEAP, a novel component of tight junctions in exocrine cells.
The renal segmental distribution of claudins changes with development.
Regulated expression of claudin-4 decreases paracellular conductance through a selective decrease in sodium permeability.
The coiled-coil domain of occludin can act to organize structural and functional elements of the epithelial tight junction.
Restoration of tight junction structure and barrier function by down-regulation of the mitogen-activated protein kinase pathway in ras-transformed Madin-Darby canine kidney cells.
Restoration of tight junction structure and barrier function by down-regulation of the mitogen-activated protein kinase pathway in ras-transformed Madin-Darby canine kidney cells.
Boonhai S, Bootdee K, Saisorn W, Takkavatakarn K, Sitticharoenchai P, Tungsanga S, Tiranathanagul K, Leelahavanichkul A
Asian Pacific journal of allergy and immunology 2023 Jun;41(2):168-178
Asian Pacific journal of allergy and immunology 2023 Jun;41(2):168-178
The protective effect and potential mechanisms of eugenol against Salmonella in vivo and in vitro.
Zhao X, Zheng S, Wei S, Tian Q, Tao Y, Bo R, Liu M, Li J
Poultry science 2022 May;101(5):101801
Poultry science 2022 May;101(5):101801
Disruption of the crypt niche promotes outgrowth of mutated colorectal tumor stem cells.
Klingler S, Hsu KS, Hua G, Martin ML, Adileh M, Baslan T, Zhang Z, Paty PB, Fuks Z, Brown AM, Kolesnick R
JCI insight 2022 Mar 8;7(5)
JCI insight 2022 Mar 8;7(5)
SARS-CoV-2-Induced Pathology-Relevance to COVID-19 Pathophysiology.
Zinserling VA, Semenova NY, Bikmurzina AE, Kruglova NM, Rybalchenko OV, Markov AG
Pathophysiology : the official journal of the International Society for Pathophysiology 2022 Jun 10;29(2):281-297
Pathophysiology : the official journal of the International Society for Pathophysiology 2022 Jun 10;29(2):281-297
Phiclust: a clusterability measure for single-cell transcriptomics reveals phenotypic subpopulations.
Mircea M, Hochane M, Fan X, Chuva de Sousa Lopes SM, Garlaschelli D, Semrau S
Genome biology 2022 Jan 10;23(1):18
Genome biology 2022 Jan 10;23(1):18
Isolation of DiNP-Degrading Microbes from the Mouse Colon and the Influence DiNP Exposure Has on the Microbiota, Intestinal Integrity, and Immune Status of the Colon.
Chiu KK, Bashir ST, Abdel-Hamid AM, Clark LV, Laws MJ, Cann I, Nowak RA, Flaws JA
Toxics 2022 Feb 6;10(2)
Toxics 2022 Feb 6;10(2)
DevKidCC allows for robust classification and direct comparisons of kidney organoid datasets.
Wilson SB, Howden SE, Vanslambrouck JM, Dorison A, Alquicira-Hernandez J, Powell JE, Little MH
Genome medicine 2022 Feb 22;14(1):19
Genome medicine 2022 Feb 22;14(1):19
Dietary branched-chain amino acids modulate the dynamics of calcium absorption and reabsorption in protein-restricted pigs.
Habibi M, Shili CN, Sutton J, Goodarzi P, Pezeshki A
Journal of animal science and biotechnology 2022 Feb 10;13(1):15
Journal of animal science and biotechnology 2022 Feb 10;13(1):15
Tackling recalcitrant Pseudomonas aeruginosa infections in critical illness via anti-virulence monotherapy.
Singh VK, Almpani M, Maura D, Kitao T, Ferrari L, Fontana S, Bergamini G, Calcaterra E, Pignaffo C, Negri M, de Oliveira Pereira T, Skinner F, Gkikas M, Andreotti D, Felici A, Déziel E, Lépine F, Rahme LG
Nature communications 2022 Aug 30;13(1):5103
Nature communications 2022 Aug 30;13(1):5103
Matrix Metalloproteinase MMP-12 Promotes Macrophage Transmigration Across Intestinal Epithelial Tight Junctions and Increases Severity of Experimental Colitis.
Nighot M, Ganapathy AS, Saha K, Suchanec E, Castillo EF, Gregory A, Shapiro S, Ma T, Nighot P
Journal of Crohn's & colitis 2021 Oct 7;15(10):1751-1765
Journal of Crohn's & colitis 2021 Oct 7;15(10):1751-1765
Organic osmolytes increase expression of specific tight junction proteins in skin and alter barrier function in keratinocytes.
El-Chami C, Foster AR, Johnson C, Clausen RP, Cornwell P, Haslam IS, Steward MC, Watson REB, Young HS, O'Neill CA
The British journal of dermatology 2021 Mar;184(3):482-494
The British journal of dermatology 2021 Mar;184(3):482-494
Fenofibrate promotes PPARα-targeted recovery of the intestinal epithelial barrier at the host-microbe interface in dogs with diabetes mellitus.
Crakes KR, Pires J, Quach N, Ellis-Reis RE, Greathouse R, Chittum KA, Steiner JM, Pesavento P, Marks SL, Dandekar S, Gilor C
Scientific reports 2021 Jun 29;11(1):13454
Scientific reports 2021 Jun 29;11(1):13454
Cystine reduces tight junction permeability and intestinal inflammation induced by oxidative stress in Caco-2 cells.
Hasegawa T, Mizugaki A, Inoue Y, Kato H, Murakami H
Amino acids 2021 Jul;53(7):1021-1032
Amino acids 2021 Jul;53(7):1021-1032
Probiotics mixture reinforces barrier function to ameliorate necrotizing enterocolitis by regulating PXR-JNK pathway.
Zhao X, Zhou J, Liang W, Sheng Q, Lu L, Chen T, Chen J, Tan K, Lv Z
Cell & bioscience 2021 Jan 19;11(1):20
Cell & bioscience 2021 Jan 19;11(1):20
Tight Junction Protein Claudin-7 Is Essential for Intestinal Epithelial Stem Cell Self-Renewal and Differentiation.
Xing T, Benderman LJ, Sabu S, Parker J, Yang J, Lu Q, Ding L, Chen YH
Cellular and molecular gastroenterology and hepatology 2020;9(4):641-659
Cellular and molecular gastroenterology and hepatology 2020;9(4):641-659
Novel Human NKCC1 Mutations Cause Defects in Goblet Cell Mucus Secretion and Chronic Inflammation.
Koumangoye R, Omer S, Kabeer MH, Delpire E
Cellular and molecular gastroenterology and hepatology 2020;9(2):239-255
Cellular and molecular gastroenterology and hepatology 2020;9(2):239-255
Astragalus mongholicus Bunge and Panax Notoginseng Formula (A&P) Combined With Bifidobacterium Contribute a Renoprotective Effect in Chronic Kidney Disease Through Inhibiting Macrophage Inflammatory Response in Kidney and Intestine.
Rui-Zhi T, Hui D, Jian-Chun L, Xia Z, Xiao-Jia W, Dan W, Jun-Ming F, Li W
Frontiers in physiology 2020;11:583668
Frontiers in physiology 2020;11:583668
The Histone Demethylase JMJD1C Regulates CAMKK2-AMPK Signaling to Participate in Cardiac Hypertrophy.
Yu S, Li Y, Zhao H, Wang Q, Chen P
Frontiers in physiology 2020;11:539
Frontiers in physiology 2020;11:539
Impaired Airway Epithelial Barrier Integrity in Response to Stenotrophomonas maltophilia Proteases, Novel Insights Using Cystic Fibrosis Bronchial Epithelial Cell Secretomics.
Molloy K, Cagney G, Dillon ET, Wynne K, Greene CM, McElvaney NG
Frontiers in immunology 2020;11:198
Frontiers in immunology 2020;11:198
oprC Impairs Host Defense by Increasing the Quorum-Sensing-Mediated Virulence of Pseudomonas aeruginosa.
Gao P, Guo K, Pu Q, Wang Z, Lin P, Qin S, Khan N, Hur J, Liang H, Wu M
Frontiers in immunology 2020;11:1696
Frontiers in immunology 2020;11:1696
Intestinal vitamin D receptor signaling ameliorates dextran sulfate sodium-induced colitis by suppressing necroptosis of intestinal epithelial cells.
Shi Y, Cui X, Sun Y, Zhao Q, Liu T
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 Oct;34(10):13494-13506
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 Oct;34(10):13494-13506
Tissue-scale tensional homeostasis in skin regulates structure and physiological function.
Kimura S, Tsuchiya A, Ogawa M, Ono M, Suda N, Sekimoto K, Takeo M, Tsuji T
Communications biology 2020 Oct 30;3(1):637
Communications biology 2020 Oct 30;3(1):637
Phosphorylation and dephosphorylation of Ser852 and Ser889 control the clustering, localization and function of PAR3.
Yamashita K, Mizuno K, Furukawa K, Hirose H, Sakurai N, Masuda-Hirata M, Amano Y, Hirose T, Suzuki A, Ohno S
Journal of cell science 2020 Nov 30;133(22)
Journal of cell science 2020 Nov 30;133(22)
Toxicological effects of bioactive peptide fractions obtained from Bothrops jararaca snake venom on the structure and function of mouse seminiferous epithelium.
Alberto-Silva C, Franzin CS, Gilio JM, Bonfim RS, Querobino SM
The journal of venomous animals and toxins including tropical diseases 2020 Jun 22;26:e20200007
The journal of venomous animals and toxins including tropical diseases 2020 Jun 22;26:e20200007
Intestinal vitamin D receptor knockout protects from oxazolone-induced colitis.
Shi Y, Liu Z, Cui X, Zhao Q, Liu T
Cell death & disease 2020 Jun 15;11(6):461
Cell death & disease 2020 Jun 15;11(6):461
Regulatory Effect of Lactobacillus brevis Bmb6 on Gut Barrier Functions in Experimental Colitis.
Shin MY, Yong CC, Oh S
Foods (Basel, Switzerland) 2020 Jul 2;9(7)
Foods (Basel, Switzerland) 2020 Jul 2;9(7)
RAS-mediated suppression of PAR3 and its effects on SCC initiation and tissue architecture occur independently of hyperplasia.
Ling J, Sckaff M, Tiwari M, Chen Y, Li J, Jones J, Sen GL
Journal of cell science 2020 Dec 7;133(23)
Journal of cell science 2020 Dec 7;133(23)
Matriptase Cleaves EpCAM and TROP2 in Keratinocytes, Destabilizing Both Proteins and Associated Claudins.
Wu CJ, Lu M, Feng X, Nakato G, Udey MC
Cells 2020 Apr 21;9(4)
Cells 2020 Apr 21;9(4)
Staphylococcus aureus Internalized by Skin Keratinocytes Evade Antibiotic Killing.
Al Kindi A, Alkahtani AM, Nalubega M, El-Chami C, O'Neill C, Arkwright PD, Pennock JL
Frontiers in microbiology 2019;10:2242
Frontiers in microbiology 2019;10:2242
Anthocyanins protect the gastrointestinal tract from high fat diet-induced alterations in redox signaling, barrier integrity and dysbiosis.
Cremonini E, Daveri E, Mastaloudis A, Adamo AM, Mills D, Kalanetra K, Hester SN, Wood SM, Fraga CG, Oteiza PI
Redox biology 2019 Sep;26:101269
Redox biology 2019 Sep;26:101269
Probiotics Prevents Sensitization to Oral Antigen and Subsequent Increases in Intestinal Tight Junction Permeability in Juvenile-Young Adult Rats.
Tulyeu J, Kumagai H, Jimbo E, Watanabe S, Yokoyama K, Cui L, Osaka H, Mieno M, Yamagata T
Microorganisms 2019 Oct 16;7(10)
Microorganisms 2019 Oct 16;7(10)
The Differentiation-Associated Keratinocyte Protein Cornifelin Contributes to Cell-Cell Adhesion of Epidermal and Mucosal Keratinocytes.
Wagner T, Beer L, Gschwandtner M, Eckhart L, Kalinina P, Laggner M, Ellinger A, Gruber R, Kuchler U, Golabi B, Tschachler E, Mildner M
The Journal of investigative dermatology 2019 Nov;139(11):2292-2301.e9
The Journal of investigative dermatology 2019 Nov;139(11):2292-2301.e9
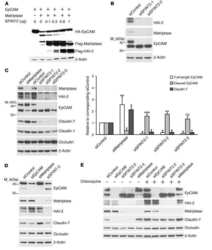
Newly synthesized claudins but not occludin are added to the basal side of the tight junction.
Van Itallie CM, Lidman KF, Tietgens AJ, Anderson JM
Molecular biology of the cell 2019 Jun 1;30(12):1406-1424
Molecular biology of the cell 2019 Jun 1;30(12):1406-1424
Protease-Activated Receptors 2-Antagonist Suppresses Asthma by Inhibiting Reactive Oxygen Species-Thymic Stromal Lymphopoietin Inflammation and Epithelial Tight Junction Degradation.
Kim HJ, Lee SH, Jeong S, Hong SJ
Allergy, asthma & immunology research 2019 Jul;11(4):560-571
Allergy, asthma & immunology research 2019 Jul;11(4):560-571
Rescue of tight junctional localization of a claudin-16 mutant D97S by antimalarial medicine primaquine in Madin-Darby canine kidney cells.
Marunaka K, Fujii N, Kimura T, Furuta T, Hasegawa H, Matsunaga T, Endo S, Ikari A
Scientific reports 2019 Jul 4;9(1):9647
Scientific reports 2019 Jul 4;9(1):9647
Geniposide and Chlorogenic Acid Combination Ameliorates Non-alcoholic Steatohepatitis Involving the Protection on the Gut Barrier Function in Mouse Induced by High-Fat Diet.
Peng JH, Leng J, Tian HJ, Yang T, Fang Y, Feng Q, Zhao Y, Hu YY
Frontiers in pharmacology 2018;9:1399
Frontiers in pharmacology 2018;9:1399
Vitamin D Receptor Deletion Leads to the Destruction of Tight and Adherens Junctions in Lungs.
Chen H, Lu R, Zhang YG, Sun J
Tissue barriers 2018;6(4):1-13
Tissue barriers 2018;6(4):1-13
HPV16-E6 Oncoprotein Activates TGF-β and Wnt/β-Catenin Pathways in the Epithelium-Mesenchymal Transition of Cataracts in a Transgenic Mouse Model.
Rodríguez-Uribe G, Serafín-Higuera N, Damian-Morales G, Cortés-Malagón EM, García-Hernández V, Verdejo-Torres O, Campos-Blázquez JP, Trejo-Muñoz CR, Contreras RG, Ocadiz-Delgado R, Palacios-Reyes C, Lambert PF, Griep AE, Mancilla-Percino T, Escobar-Herrera J, Álvarez-Ríos E, Ugarte-Briones C, Moreno J, Gariglio P, Bonilla-Delgado J
BioMed research international 2018;2018:2847873
BioMed research international 2018;2018:2847873
Esrp1-Regulated Splicing of Arhgef11 Isoforms Is Required for Epithelial Tight Junction Integrity.
Lee S, Cieply B, Yang Y, Peart N, Glaser C, Chan P, Carstens RP
Cell reports 2018 Nov 27;25(9):2417-2430.e5
Cell reports 2018 Nov 27;25(9):2417-2430.e5
Perineurial-like Cells and EMA Expression in the Suprachoroidal Region of the Human Eye.
Gilbert AR, Chévez-Barrios P, Cykowski MD
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2018 May;66(5):367-375
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2018 May;66(5):367-375
Analysis of the bystander effect in cone photoreceptors via a guided neural network platform.
Ma Y, Han X, de Castro RB, Zhang P, Zhang K, Hu Z, Qin L
Science advances 2018 May;4(5):eaas9274
Science advances 2018 May;4(5):eaas9274
VPS33B and VIPAR are essential for epidermal lamellar body biogenesis and function.
Rogerson C, Gissen P
Biochimica et biophysica acta. Molecular basis of disease 2018 May;1864(5 Pt A):1609-1621
Biochimica et biophysica acta. Molecular basis of disease 2018 May;1864(5 Pt A):1609-1621
Adherens junctions influence tight junction formation via changes in membrane lipid composition.
Shigetomi K, Ono Y, Inai T, Ikenouchi J
The Journal of cell biology 2018 Jul 2;217(7):2373-2381
The Journal of cell biology 2018 Jul 2;217(7):2373-2381
Enteropathogenic E. coli effectors EspF and Map independently disrupt tight junctions through distinct mechanisms involving transcriptional and post-transcriptional regulation.
Singh AP, Sharma S, Pagarware K, Siraji RA, Ansari I, Mandal A, Walling P, Aijaz S
Scientific reports 2018 Feb 27;8(1):3719
Scientific reports 2018 Feb 27;8(1):3719
Establishing normal metabolism and differentiation in hepatocellular carcinoma cells by culturing in adult human serum.
Steenbergen R, Oti M, Ter Horst R, Tat W, Neufeldt C, Belovodskiy A, Chua TT, Cho WJ, Joyce M, Dutilh BE, Tyrrell DL
Scientific reports 2018 Aug 3;8(1):11685
Scientific reports 2018 Aug 3;8(1):11685
(-)-Epicatechin protects the intestinal barrier from high fat diet-induced permeabilization: Implications for steatosis and insulin resistance.
Cremonini E, Wang Z, Bettaieb A, Adamo AM, Daveri E, Mills DA, Kalanetra KM, Haj FG, Karakas S, Oteiza PI
Redox biology 2018 Apr;14:588-599
Redox biology 2018 Apr;14:588-599
Inflammatory cytokines down-regulate the barrier-protective prostasin-matriptase proteolytic cascade early in experimental colitis.
Buzza MS, Johnson TA, Conway GD, Martin EW, Mukhopadhyay S, Shea-Donohue T, Antalis TM
The Journal of biological chemistry 2017 Jun 30;292(26):10801-10812
The Journal of biological chemistry 2017 Jun 30;292(26):10801-10812
TRPV4 Regulates Tight Junctions and Affects Differentiation in a Cell Culture Model of the Corneal Epithelium.
Martínez-Rendón J, Sánchez-Guzmán E, Rueda A, González J, Gulias-Cañizo R, Aquino-Jarquín G, Castro-Muñozledo F, García-Villegas R
Journal of cellular physiology 2017 Jul;232(7):1794-1807
Journal of cellular physiology 2017 Jul;232(7):1794-1807
Obesity-induces Organ and Tissue Specific Tight Junction Restructuring and Barrier Deregulation by Claudin Switching.
Ahmad R, Rah B, Bastola D, Dhawan P, Singh AB
Scientific reports 2017 Jul 11;7(1):5125
Scientific reports 2017 Jul 11;7(1):5125
Epithelial response to a high-protein diet in rat colon.
Beaumont M, Andriamihaja M, Armand L, Grauso M, Jaffrézic F, Laloë D, Moroldo M, Davila AM, Tomé D, Blachier F, Lan A
BMC genomics 2017 Jan 31;18(1):116
BMC genomics 2017 Jan 31;18(1):116
Down-regulation of Claudin-2 Expression and Proliferation by Epigenetic Inhibitors in Human Lung Adenocarcinoma A549 Cells.
Hichino A, Okamoto M, Taga S, Akizuki R, Endo S, Matsunaga T, Ikari A
The Journal of biological chemistry 2017 Feb 10;292(6):2411-2421
The Journal of biological chemistry 2017 Feb 10;292(6):2411-2421
Matriptase-mediated cleavage of EpCAM destabilizes claudins and dysregulates intestinal epithelial homeostasis.
Wu CJ, Feng X, Lu M, Morimura S, Udey MC
The Journal of clinical investigation 2017 Feb 1;127(2):623-634
The Journal of clinical investigation 2017 Feb 1;127(2):623-634
Downregulation of lipolysis-stimulated lipoprotein receptor promotes cell invasion via claudin-1-mediated matrix metalloproteinases in human endometrial cancer.
Shimada H, Satohisa S, Kohno T, Konno T, Takano KI, Takahashi S, Hatakeyama T, Arimoto C, Saito T, Kojima T
Oncology letters 2017 Dec;14(6):6776-6782
Oncology letters 2017 Dec;14(6):6776-6782
Lactate-mediated mitoribosomal defects impair mitochondrial oxidative phosphorylation and promote hepatoma cell invasiveness.
Lee YK, Lim JJ, Jeoun UW, Min S, Lee EB, Kwon SM, Lee C, Yoon G
The Journal of biological chemistry 2017 Dec 8;292(49):20208-20217
The Journal of biological chemistry 2017 Dec 8;292(49):20208-20217
Serglycin in tumor microenvironment promotes non-small cell lung cancer aggressiveness in a CD44-dependent manner.
Guo JY, Hsu HS, Tyan SW, Li FY, Shew JY, Lee WH, Chen JY
Oncogene 2017 Apr 27;36(17):2457-2471
Oncogene 2017 Apr 27;36(17):2457-2471
T-Lymphocytes Traffic into the Brain across the Blood-CSF Barrier: Evidence Using a Reconstituted Choroid Plexus Epithelium.
Strazielle N, Creidy R, Malcus C, Boucraut J, Ghersi-Egea JF
PloS one 2016;11(3):e0150945
PloS one 2016;11(3):e0150945
Urinary Podocyte Loss Is Increased in Patients with Fabry Disease and Correlates with Clinical Severity of Fabry Nephropathy.
Fall B, Scott CR, Mauer M, Shankland S, Pippin J, Jefferson JA, Wallace E, Warnock D, Najafian B
PloS one 2016;11(12):e0168346
PloS one 2016;11(12):e0168346
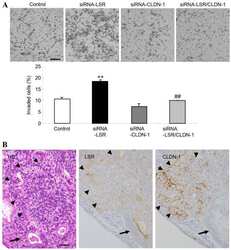
Liver kinase B1 regulates hepatocellular tight junction distribution and function in vivo.
Porat-Shliom N, Tietgens AJ, Van Itallie CM, Vitale-Cross L, Jarnik M, Harding OJ, Anderson JM, Gutkind JS, Weigert R, Arias IM
Hepatology (Baltimore, Md.) 2016 Oct;64(4):1317-29
Hepatology (Baltimore, Md.) 2016 Oct;64(4):1317-29
HOXA5 determines cell fate transition and impedes tumor initiation and progression in breast cancer through regulation of E-cadherin and CD24.
Teo WW, Merino VF, Cho S, Korangath P, Liang X, Wu RC, Neumann NM, Ewald AJ, Sukumar S
Oncogene 2016 Oct 20;35(42):5539-5551
Oncogene 2016 Oct 20;35(42):5539-5551
Dual effects of a high-protein diet on DSS-treated mice during colitis resolution phase.
Lan A, Blais A, Coelho D, Capron J, Maarouf M, Benamouzig R, Lancha AH Jr, Walker F, Tomé D, Blachier F
American journal of physiology. Gastrointestinal and liver physiology 2016 Oct 1;311(4):G624-G633
American journal of physiology. Gastrointestinal and liver physiology 2016 Oct 1;311(4):G624-G633
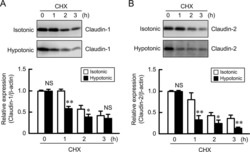
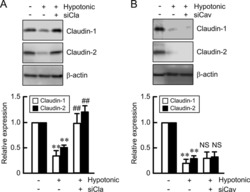
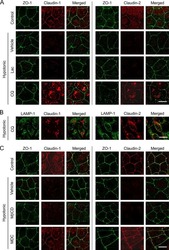
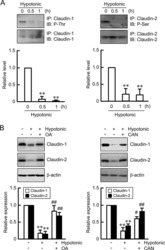
Hypotonic Stress-induced Down-regulation of Claudin-1 and -2 Mediated by Dephosphorylation and Clathrin-dependent Endocytosis in Renal Tubular Epithelial Cells.
Fujii N, Matsuo Y, Matsunaga T, Endo S, Sakai H, Yamaguchi M, Yamazaki Y, Sugatani J, Ikari A
The Journal of biological chemistry 2016 Nov 18;291(47):24787-24799
The Journal of biological chemistry 2016 Nov 18;291(47):24787-24799
Regulation of post-Golgi LH3 trafficking is essential for collagen homeostasis.
Banushi B, Forneris F, Straatman-Iwanowska A, Strange A, Lyne AM, Rogerson C, Burden JJ, Heywood WE, Hanley J, Doykov I, Straatman KR, Smith H, Bem D, Kriston-Vizi J, Ariceta G, Risteli M, Wang C, Ardill RE, Zaniew M, Latka-Grot J, Waddington SN, Howe SJ, Ferraro F, Gjinovci A, Lawrence S, Marsh M, Girolami M, Bozec L, Mills K, Gissen P
Nature communications 2016 Jul 20;7:12111
Nature communications 2016 Jul 20;7:12111
Maintaining physical activity during refeeding improves body composition, intestinal hyperpermeability and behavior in anorectic mice.
Achamrah N, Nobis S, Breton J, Jésus P, Belmonte L, Maurer B, Legrand R, Bôle-Feysot C, do Rego JL, Goichon A, Rego JC, Déchelotte P, Fetissov SO, Claeyssens S, Coëffier M
Scientific reports 2016 Feb 24;6:21887
Scientific reports 2016 Feb 24;6:21887
Altered Prostasin (CAP1/Prss8) Expression Favors Inflammation and Tissue Remodeling in DSS-induced Colitis.
Keppner A, Malsure S, Nobile A, Auberson M, Bonny O, Hummler E
Inflammatory bowel diseases 2016 Dec;22(12):2824-2839
Inflammatory bowel diseases 2016 Dec;22(12):2824-2839
Salmonella enteritidis Effector AvrA Stabilizes Intestinal Tight Junctions via the JNK Pathway.
Lin Z, Zhang YG, Xia Y, Xu X, Jiao X, Sun J
The Journal of biological chemistry 2016 Dec 23;291(52):26837-26849
The Journal of biological chemistry 2016 Dec 23;291(52):26837-26849
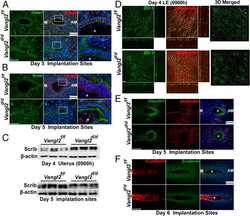
Planar cell polarity signaling in the uterus directs appropriate positioning of the crypt for embryo implantation.
Yuan J, Cha J, Deng W, Bartos A, Sun X, Ho HH, Borg JP, Yamaguchi TP, Yang Y, Dey SK
Proceedings of the National Academy of Sciences of the United States of America 2016 Dec 13;113(50):E8079-E8088
Proceedings of the National Academy of Sciences of the United States of America 2016 Dec 13;113(50):E8079-E8088
Combined Treatment with Epigenetic, Differentiating, and Chemotherapeutic Agents Cooperatively Targets Tumor-Initiating Cells in Triple-Negative Breast Cancer.
Merino VF, Nguyen N, Jin K, Sadik H, Cho S, Korangath P, Han L, Foster YMN, Zhou XC, Zhang Z, Connolly RM, Stearns V, Ali SZ, Adams C, Chen Q, Pan D, Huso DL, Ordentlich P, Brodie A, Sukumar S
Cancer research 2016 Apr 1;76(7):2013-2024
Cancer research 2016 Apr 1;76(7):2013-2024
Proliferation of cultured mouse choroid plexus epithelial cells.
Barkho BZ, Monuki ES
PloS one 2015;10(3):e0121738
PloS one 2015;10(3):e0121738
Defining a conformational consensus motif in cotransin-sensitive signal sequences: a proteomic and site-directed mutagenesis study.
Klein W, Westendorf C, Schmidt A, Conill-Cortés M, Rutz C, Blohs M, Beyermann M, Protze J, Krause G, Krause E, Schülein R
PloS one 2015;10(3):e0120886
PloS one 2015;10(3):e0120886
The impact of CLAUDIN-1 on follicular thyroid carcinoma aggressiveness.
Zwanziger D, Badziong J, Ting S, Moeller LC, Schmid KW, Siebolts U, Wickenhauser C, Dralle H, Fuehrer D
Endocrine-related cancer 2015 Oct;22(5):819-30
Endocrine-related cancer 2015 Oct;22(5):819-30
The characterization of the human nasal epithelial cell line RPMI 2650 under different culture conditions and their optimization for an appropriate in vitro nasal model.
Kreft ME, Jerman UD, Lasič E, Lanišnik Rižner T, Hevir-Kene N, Peternel L, Kristan K
Pharmaceutical research 2015 Feb;32(2):665-79
Pharmaceutical research 2015 Feb;32(2):665-79
Mitochondrial Respiratory Dysfunction Induces Claudin-1 Expression via Reactive Oxygen Species-mediated Heat Shock Factor 1 Activation, Leading to Hepatoma Cell Invasiveness.
Lee JH, Lee YK, Lim JJ, Byun HO, Park I, Kim GH, Xu WG, Wang HJ, Yoon G
The Journal of biological chemistry 2015 Aug 28;290(35):21421-31
The Journal of biological chemistry 2015 Aug 28;290(35):21421-31
Toll-like receptor 2 regulates the barrier function of human bronchial epithelial monolayers through atypical protein kinase C zeta, and an increase in expression of claudin-1.
Ragupathy S, Esmaeili F, Paschoud S, Sublet E, Citi S, Borchard G
Tissue barriers 2014;2:e29166
Tissue barriers 2014;2:e29166
Evidence for a role of claudin 2 as a proximal tubular stress responsive paracellular water channel.
Wilmes A, Aschauer L, Limonciel A, Pfaller W, Jennings P
Toxicology and applied pharmacology 2014 Sep 1;279(2):163-72
Toxicology and applied pharmacology 2014 Sep 1;279(2):163-72
Alteration of intestinal barrier function during activity-based anorexia in mice.
Jésus P, Ouelaa W, François M, Riachy L, Guérin C, Aziz M, Do Rego JC, Déchelotte P, Fetissov SO, Coëffier M
Clinical nutrition (Edinburgh, Scotland) 2014 Dec;33(6):1046-53
Clinical nutrition (Edinburgh, Scotland) 2014 Dec;33(6):1046-53
The reversible increase in tight junction permeability induced by capsaicin is mediated via cofilin-actin cytoskeletal dynamics and decreased level of occludin.
Shiobara T, Usui T, Han J, Isoda H, Nagumo Y
PloS one 2013;8(11):e79954
PloS one 2013;8(11):e79954
Nf1 loss and Ras hyperactivation in oligodendrocytes induce NOS-driven defects in myelin and vasculature.
Mayes DA, Rizvi TA, Titus-Mitchell H, Oberst R, Ciraolo GM, Vorhees CV, Robinson AP, Miller SD, Cancelas JA, Stemmer-Rachamimov AO, Ratner N
Cell reports 2013 Sep 26;4(6):1197-212
Cell reports 2013 Sep 26;4(6):1197-212
Papillomavirus E6 oncoprotein up-regulates occludin and ZO-2 expression in ovariectomized mice epidermis.
Hernández-Monge J, Garay E, Raya-Sandino A, Vargas-Sierra O, Díaz-Chávez J, Popoca-Cuaya M, Lambert PF, González-Mariscal L, Gariglio P
Experimental cell research 2013 Oct 15;319(17):2588-603
Experimental cell research 2013 Oct 15;319(17):2588-603
A bradykinin-potentiating peptide (BPP-10c) from Bothrops jararaca induces changes in seminiferous tubules.
Gilio JM, Portaro FC, Borella MI, Lameu C, Camargo AC, Alberto-Silva C
The journal of venomous animals and toxins including tropical diseases 2013 Nov 6;19(1):28
The journal of venomous animals and toxins including tropical diseases 2013 Nov 6;19(1):28
Contribution of tight junction proteins to ion, macromolecule, and water barrier in keratinocytes.
Kirschner N, Rosenthal R, Furuse M, Moll I, Fromm M, Brandner JM
The Journal of investigative dermatology 2013 May;133(5):1161-9
The Journal of investigative dermatology 2013 May;133(5):1161-9
Expression of claudins -2 and -4 and cingulin is coordinated with the start of stratification and differentiation in corneal epithelial cells: retinoic acid reversibly disrupts epithelial barrier.
Ortiz-Melo MT, Sánchez-Guzmán E, González-Robles A, Valdés J, Gómez-Flores E, Castro-Muñozledo F
Biology open 2013 Feb 15;2(2):132-43
Biology open 2013 Feb 15;2(2):132-43
GLP-2 enhances barrier formation and attenuates TNFα-induced changes in a Caco-2 cell model of the intestinal barrier.
Moran GW, O'Neill C, McLaughlin JT
Regulatory peptides 2012 Oct 10;178(1-3):95-101
Regulatory peptides 2012 Oct 10;178(1-3):95-101
Oxidative stress induced by potassium bromate exposure results in altered tight junction protein expression in renal proximal tubule cells.
Limonciel A, Wilmes A, Aschauer L, Radford R, Bloch KM, McMorrow T, Pfaller W, van Delft JH, Slattery C, Ryan MP, Lock EA, Jennings P
Archives of toxicology 2012 Nov;86(11):1741-51
Archives of toxicology 2012 Nov;86(11):1741-51
Cingulin is dispensable for epithelial barrier function and tight junction structure, and plays a role in the control of claudin-2 expression and response to duodenal mucosa injury.
Guillemot L, Schneider Y, Brun P, Castagliuolo I, Pizzuti D, Martines D, Jond L, Bongiovanni M, Citi S
Journal of cell science 2012 Nov 1;125(Pt 21):5005-14
Journal of cell science 2012 Nov 1;125(Pt 21):5005-14
Differential effects of flavonoids on barrier integrity in human intestinal Caco-2 cells.
Noda S, Tanabe S, Suzuki T
Journal of agricultural and food chemistry 2012 May 9;60(18):4628-33
Journal of agricultural and food chemistry 2012 May 9;60(18):4628-33
Functional characterization and localization of a gill-specific claudin isoform in Atlantic salmon.
Engelund MB, Yu AS, Li J, Madsen SS, Færgeman NJ, Tipsmark CK
American journal of physiology. Regulatory, integrative and comparative physiology 2012 Jan 15;302(2):R300-11
American journal of physiology. Regulatory, integrative and comparative physiology 2012 Jan 15;302(2):R300-11
Tight junction proteins expression and modulation in immune cells and multiple sclerosis.
Mandel I, Paperna T, Glass-Marmor L, Volkowich A, Badarny S, Schwartz I, Vardi P, Koren I, Miller A
Journal of cellular and molecular medicine 2012 Apr;16(4):765-75
Journal of cellular and molecular medicine 2012 Apr;16(4):765-75
EGFR regulation of epidermal barrier function.
Tran QT, Kennedy LH, Leon Carrion S, Bodreddigari S, Goodwin SB, Sutter CH, Sutter TR
Physiological genomics 2012 Apr 15;44(8):455-69
Physiological genomics 2012 Apr 15;44(8):455-69
Expression of claudin-1 and -11 in immature and mature pheasant (Phasianus colchicus) testes.
Park CJ, Lee JE, Oh YS, Shim S, Nah WH, Choi KJ, Gye MC
Theriogenology 2011 Feb;75(3):445-58
Theriogenology 2011 Feb;75(3):445-58
CD44 regulates tight-junction assembly and barrier function.
Kirschner N, Haftek M, Niessen CM, Behne MJ, Furuse M, Moll I, Brandner JM
The Journal of investigative dermatology 2011 Apr;131(4):932-43
The Journal of investigative dermatology 2011 Apr;131(4):932-43
The potency of the fs260 connexin43 mutant to impair keratinocyte differentiation is distinct from other disease-linked connexin43 mutants.
Churko JM, Langlois S, Pan X, Shao Q, Laird DW
The Biochemical journal 2010 Aug 1;429(3):473-83
The Biochemical journal 2010 Aug 1;429(3):473-83
Increased intraocular insulin-like growth factor-I triggers blood-retinal barrier breakdown.
Haurigot V, Villacampa P, Ribera A, Llombart C, Bosch A, Nacher V, Ramos D, Ayuso E, Segovia JC, Bueren JA, Ruberte J, Bosch F
The Journal of biological chemistry 2009 Aug 21;284(34):22961-9
The Journal of biological chemistry 2009 Aug 21;284(34):22961-9
Claudin expression modulations reflect an injury response in the murine epidermis.
Arabzadeh A, Troy TC, Turksen K
The Journal of investigative dermatology 2008 Jan;128(1):237-40
The Journal of investigative dermatology 2008 Jan;128(1):237-40
Changes in the distribution pattern of Claudin tight junction proteins during the progression of mouse skin tumorigenesis.
Arabzadeh A, Troy TC, Turksen K
BMC cancer 2007 Oct 18;7:196
BMC cancer 2007 Oct 18;7:196
Astrovirus increases epithelial barrier permeability independently of viral replication.
Moser LA, Carter M, Schultz-Cherry S
Journal of virology 2007 Nov;81(21):11937-45
Journal of virology 2007 Nov;81(21):11937-45
Claudin immunolocalization in neonatal mouse epithelial tissues.
Troy TC, Arabzadeh A, Yerlikaya S, Turksen K
Cell and tissue research 2007 Nov;330(2):381-8
Cell and tissue research 2007 Nov;330(2):381-8
Dynamic changes in the cervical epithelial tight junction complex and differentiation occur during cervical ripening and parturition.
Timmons BC, Mitchell SM, Gilpin C, Mahendroo MS
Endocrinology 2007 Mar;148(3):1278-87
Endocrinology 2007 Mar;148(3):1278-87
Proteomic and bioinformatic analysis of epithelial tight junction reveals an unexpected cluster of synaptic molecules.
Tang VW
Biology direct 2006 Dec 8;1:37
Biology direct 2006 Dec 8;1:37
Leukocyte diapedesis in vivo induces transient loss of tight junction protein at the blood-retina barrier.
Xu H, Dawson R, Crane IJ, Liversidge J
Investigative ophthalmology & visual science 2005 Jul;46(7):2487-94
Investigative ophthalmology & visual science 2005 Jul;46(7):2487-94
Simultaneous cell death and desquamation of the embryonic diffusion barrier during epidermal development.
Saathoff M, Blum B, Quast T, Kirfel G, Herzog V
Experimental cell research 2004 Oct 1;299(2):415-26
Experimental cell research 2004 Oct 1;299(2):415-26
Expression and function of tight junctions in the crypt epithelium of human palatine tonsils.
Go M, Kojima T, Takano K, Murata M, Ichimiya S, Tsubota H, Himi T, Sawada N
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Dec;52(12):1627-38
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Dec;52(12):1627-38
Connexin 26-mediated gap junctional intercellular communication suppresses paracellular permeability of human intestinal epithelial cell monolayers.
Morita H, Katsuno T, Hoshimoto A, Hirano N, Saito Y, Suzuki Y
Experimental cell research 2004 Aug 1;298(1):1-8
Experimental cell research 2004 Aug 1;298(1):1-8
Interferon-gamma down-regulates claudin-1 and impairs the epithelial barrier function in primary cultured human thyrocytes.
Tedelind S, Ericson LE, Karlsson JO, Nilsson M
European journal of endocrinology 2003 Sep;149(3):215-21
European journal of endocrinology 2003 Sep;149(3):215-21
Interferon-gamma down-regulates claudin-1 and impairs the epithelial barrier function in primary cultured human thyrocytes.
Tedelind S, Ericson LE, Karlsson JO, Nilsson M
European journal of endocrinology 2003 Sep;149(3):215-21
European journal of endocrinology 2003 Sep;149(3):215-21
Simian virus 40 small tumor antigen induces deregulation of the actin cytoskeleton and tight junctions in kidney epithelial cells.
Nunbhakdi-Craig V, Craig L, Machleidt T, Sontag E
Journal of virology 2003 Mar;77(5):2807-18
Journal of virology 2003 Mar;77(5):2807-18
Localization of claudin-3 in tight junctions of the blood-brain barrier is selectively lost during experimental autoimmune encephalomyelitis and human glioblastoma multiforme.
Wolburg H, Wolburg-Buchholz K, Kraus J, Rascher-Eggstein G, Liebner S, Hamm S, Duffner F, Grote EH, Risau W, Engelhardt B
Acta neuropathologica 2003 Jun;105(6):586-92
Acta neuropathologica 2003 Jun;105(6):586-92
Protection against hypoxia-induced increase in blood-brain barrier permeability: role of tight junction proteins and NFkappaB.
Brown RC, Mark KS, Egleton RD, Huber JD, Burroughs AR, Davis TP
Journal of cell science 2003 Feb 15;116(Pt 4):693-700
Journal of cell science 2003 Feb 15;116(Pt 4):693-700
Protection against hypoxia-induced increase in blood-brain barrier permeability: role of tight junction proteins and NFkappaB.
Brown RC, Mark KS, Egleton RD, Huber JD, Burroughs AR, Davis TP
Journal of cell science 2003 Feb 15;116(Pt 4):693-700
Journal of cell science 2003 Feb 15;116(Pt 4):693-700
SRC-induced disintegration of adherens junctions of madin-darby canine kidney cells is dependent on endocytosis of cadherin and antagonized by Tiam-1.
Palovuori R, Sormunen R, Eskelinen S
Laboratory investigation; a journal of technical methods and pathology 2003 Dec;83(12):1901-15
Laboratory investigation; a journal of technical methods and pathology 2003 Dec;83(12):1901-15
SRC-induced disintegration of adherens junctions of madin-darby canine kidney cells is dependent on endocytosis of cadherin and antagonized by Tiam-1.
Palovuori R, Sormunen R, Eskelinen S
Laboratory investigation; a journal of technical methods and pathology 2003 Dec;83(12):1901-15
Laboratory investigation; a journal of technical methods and pathology 2003 Dec;83(12):1901-15
Proinflammatory cytokines disrupt epithelial barrier function by apoptosis-independent mechanisms.
Bruewer M, Luegering A, Kucharzik T, Parkos CA, Madara JL, Hopkins AM, Nusrat A
Journal of immunology (Baltimore, Md. : 1950) 2003 Dec 1;171(11):6164-72
Journal of immunology (Baltimore, Md. : 1950) 2003 Dec 1;171(11):6164-72
Proinflammatory cytokines disrupt epithelial barrier function by apoptosis-independent mechanisms.
Bruewer M, Luegering A, Kucharzik T, Parkos CA, Madara JL, Hopkins AM, Nusrat A
Journal of immunology (Baltimore, Md. : 1950) 2003 Dec 1;171(11):6164-72
Journal of immunology (Baltimore, Md. : 1950) 2003 Dec 1;171(11):6164-72
2,3-butanedione monoxime (BDM), a potent inhibitor of actin-myosin interaction, induces ion and fluid transport in MDCK monolayers.
Castillo AM, Reyes JL, Sánchez E, Mondragón R, Meza I
Journal of muscle research and cell motility 2002;23(3):223-34
Journal of muscle research and cell motility 2002;23(3):223-34
2,3-butanedione monoxime (BDM), a potent inhibitor of actin-myosin interaction, induces ion and fluid transport in MDCK monolayers.
Castillo AM, Reyes JL, Sánchez E, Mondragón R, Meza I
Journal of muscle research and cell motility 2002;23(3):223-34
Journal of muscle research and cell motility 2002;23(3):223-34
Protein phosphatase 2A associates with and regulates atypical PKC and the epithelial tight junction complex.
Nunbhakdi-Craig V, Machleidt T, Ogris E, Bellotto D, White CL 3rd, Sontag E
The Journal of cell biology 2002 Sep 2;158(5):967-78
The Journal of cell biology 2002 Sep 2;158(5):967-78
E-Cadherin and tight junctions between epithelial cells of different animal species.
Contreras RG, Shoshani L, Flores-Maldonado C, Lázaro A, Monroy AO, Roldán ML, Fiorentino R, Cereijido M
Pflugers Archiv : European journal of physiology 2002 Jul;444(4):467-75
Pflugers Archiv : European journal of physiology 2002 Jul;444(4):467-75
Tight junctions are sensitive to peptides eliminated in the urine.
Gallardo JM, Hernández JM, Contreras RG, Flores-Maldonado C, González-Mariscal L, Cereijido M
The Journal of membrane biology 2002 Jul 1;188(1):33-42
The Journal of membrane biology 2002 Jul 1;188(1):33-42
Neutrophil transepithelial migration: evidence for sequential, contact-dependent signaling events and enhanced paracellular permeability independent of transjunctional migration.
Edens HA, Levi BP, Jaye DL, Walsh S, Reaves TA, Turner JR, Nusrat A, Parkos CA
Journal of immunology (Baltimore, Md. : 1950) 2002 Jul 1;169(1):476-86
Journal of immunology (Baltimore, Md. : 1950) 2002 Jul 1;169(1):476-86
Neutrophil transepithelial migration: evidence for sequential, contact-dependent signaling events and enhanced paracellular permeability independent of transjunctional migration.
Edens HA, Levi BP, Jaye DL, Walsh S, Reaves TA, Turner JR, Nusrat A, Parkos CA
Journal of immunology (Baltimore, Md. : 1950) 2002 Jul 1;169(1):476-86
Journal of immunology (Baltimore, Md. : 1950) 2002 Jul 1;169(1):476-86
JEAP, a novel component of tight junctions in exocrine cells.
Nishimura M, Kakizaki M, Ono Y, Morimoto K, Takeuchi M, Inoue Y, Imai T, Takai Y
The Journal of biological chemistry 2002 Feb 15;277(7):5583-7
The Journal of biological chemistry 2002 Feb 15;277(7):5583-7
The renal segmental distribution of claudins changes with development.
Reyes JL, Lamas M, Martin D, del Carmen Namorado M, Islas S, Luna J, Tauc M, González-Mariscal L
Kidney international 2002 Aug;62(2):476-87
Kidney international 2002 Aug;62(2):476-87
Regulated expression of claudin-4 decreases paracellular conductance through a selective decrease in sodium permeability.
Van Itallie C, Rahner C, Anderson JM
The Journal of clinical investigation 2001 May;107(10):1319-27
The Journal of clinical investigation 2001 May;107(10):1319-27
The coiled-coil domain of occludin can act to organize structural and functional elements of the epithelial tight junction.
Nusrat A, Chen JA, Foley CS, Liang TW, Tom J, Cromwell M, Quan C, Mrsny RJ
The Journal of biological chemistry 2000 Sep 22;275(38):29816-22
The Journal of biological chemistry 2000 Sep 22;275(38):29816-22
Restoration of tight junction structure and barrier function by down-regulation of the mitogen-activated protein kinase pathway in ras-transformed Madin-Darby canine kidney cells.
Chen Yh, Lu Q, Schneeberger EE, Goodenough DA
Molecular biology of the cell 2000 Mar;11(3):849-62
Molecular biology of the cell 2000 Mar;11(3):849-62
Restoration of tight junction structure and barrier function by down-regulation of the mitogen-activated protein kinase pathway in ras-transformed Madin-Darby canine kidney cells.
Chen Yh, Lu Q, Schneeberger EE, Goodenough DA
Molecular biology of the cell 2000 Mar;11(3):849-62
Molecular biology of the cell 2000 Mar;11(3):849-62
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
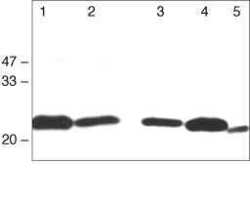
- Experimental details
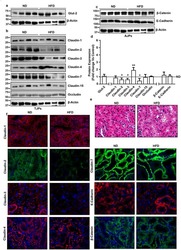
- Western blot analysis using Rabbit anti-Claudin-1 Polyclonal Antibody in: Lane 1: Rat liver. Lane 2: Rat kidney. Lane 3: MDCK cells. Lane 4: Caco-2 cells. Lane 5: Mouse hepatocytes (Product # 71-7800).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
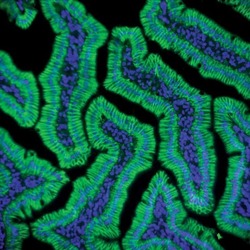
- Experimental details
- Immunofluorescent staining of rat small intestine using Rb anti-Claudin-1 (Product # 71-7800).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
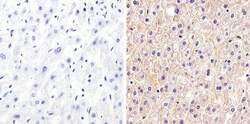
- Experimental details
- Immunohistochemistry analysis of Claudin-1 showing staining in the membrane and weak cytoplasm staining of paraffin-embedded human liver tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Claudin-1 Rabbit Polyclonal Antibody (Product # 71-7800) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
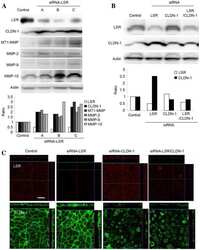
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
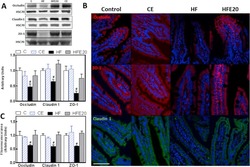
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
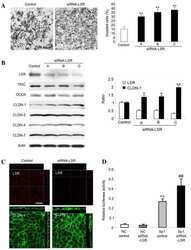
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
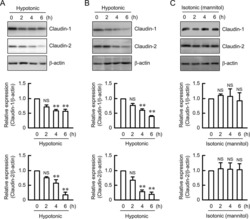
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
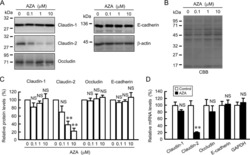
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
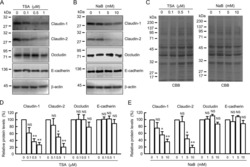
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
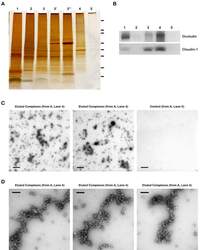
- Figure 4 Validation of specificity and enrichment of tight junction complexes . (A) Silver stained gel showing steps of purification. Lane 1, whole cell lysate; lane 2, 30,000 x g supernatant; lane 3, 100,000 x g membrane; Lane 3', output 100,000 x g membrane after immuno-isolation, Lane 3"", output 100,000 x g membrane after control immuno-isolation; lane 4, immuno-isolated tight junction complexes; lane 5, control immuno-isolation. MW markers are 200, 116, 96, 66, 45, 38, 25, 14. (b) Western blots of tight junction markers occludin and claudin-1. Lanes 1-5, same as A. (C) Negative staining of complexes showing assemblies of a heterogeneous population of proteins in various sizes. Control immunopurification shows relatively clean background. Scale bars, 100 nm. (D) Higher magnification of tight junction complexes shows globular proteins linked together forming beaded-necklace arrays reminiscent of tight junction seen in vivo (see Results).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
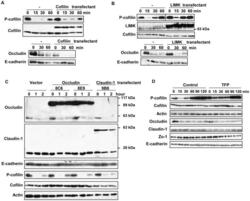
- Figure 5 Effects of suppression of cofilin activation or overexpresssion of TJ proteins on the capsaicin-induced cofilin dephosphorylation and decrease in occludin expression. (A-C) Flag-HA tagged cofilin and LIMK, and EGFP-occludin and claudin-1 stable transfectants were established. Monolayers prepared from the transfectants and non-transfected MDCK cells were exposed to 300 uM capsaicin for the time indicated. After lysis, protein expression was analyzed by immunoblotting. Each experiment was performed with at least two different clones and repeated at least twice. (D) MDCK monolayers were pretreated with vehicle or 50 uM TFP for 30 min, and then exposed to 100 uM capsaicin for the time indicated. The levels of occludin and phospho-cofilin were analyzed by western blotting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Cofilin activation, decrease in occludin expression and actin alteration in the recovery phase of capsaicin treatment. (A) Western blot detection of phospho-cofilin and occludin in total extracts exposed to 300 uM capsaicin for the times indicated. (B) MDCK monolayers were exposed to 300 uM capsaicin for the time indicated, and then fixed and stained with rhodamine-phalloidin to detect F-actin. Images from each z-section were deconvoluted and overlayed. Bar: 10 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
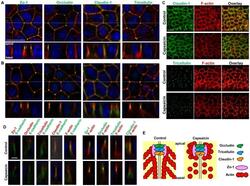
- Experimental details
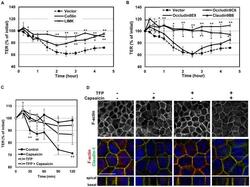
- Figure 3 Effects of capsaicin on the distribution of TJ proteins and F-actin. (A, B) Monolayers were exposed to vehicle (A) or 300 uM capsaicin (B) for 45 min and were labeled with each TJ antibody (green), rhodamine-phalloidin (red) and Hoechst (blue). Images were collected as a Z-series, and then deconvoluted and overlayed to display a single composite projection. Top : XY sections of merged images. Bottom : XZ sections of merged images. Scale bar: 10 um. The corresponding TJ antibodies are listed above the images. (C) 3D projection images in a 45deg-angle of claudin-1 and tricellulin staining, plus F-actin. Scale bar: 10 um. (D) MDCK monolayers exposed to vehicle (top) or capsaicin (bottom) as above were stained with E-cadherin. The colors corresponding to each antibody are listed above the images. Scale bar: 2.5 um. (E) Schematic explanation of the protein distributions.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
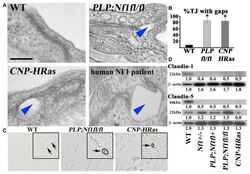
- Experimental details
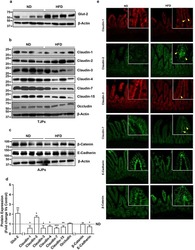
- Figure 3 Nf1 Loss or HRas Activation in the Optic Nerve Causes Changes in Claudins and BBB Permeability (A) Electron micrographs of optic-nerve cross-sections of capillaries 1 mm from the chiasm (50,000x; scale bar, 200nm) show electron-dense TJs between endothelial cells. Blue arrowheads: areas of TJ disruption. (B) Quantification of the total TJs with gaps, with 200-300 TJs counted per genotype. (C) Evans blue stain of longitudinal sections of PLPCre; Nf1fl / fl and CNP-HRas optic nerve. Insets: cross-sections; arrows: blood vessel. (D) Western blots from total optic-nerve lysates showing claudin-1 and claudin-5. All experiments included three to five animals per genotype. *p < 0.001. See also Figure S5 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
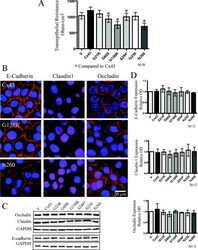
- Experimental details
- Figure 6 Cx43 mutant-expressing REKs exhibited reduced transepithelial resistance when compared with Cx43 overexpressing REKs in the absence of changes in the expression or localization of junctional proteins ( A ) Transepithelial resistance measurements of confluent monolayer cultures were taken daily for 5 days and the resistance from days 2-4 were averaged and plotted. ( B ) E-cadherin, claudin1 and occludin were localized by immunofluorescent labelling in REKs overexpressing GFP-tagged Cx43, G138R or fs260. Western blot analysis did not reveal any changes in the expression of occludin, E-cadherin or claudin-1 ( C and D ). * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
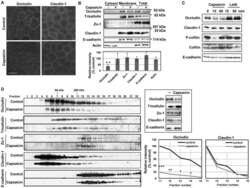
- Experimental details
- Figure 4 Capsaicin decreases occludin protein content and the protein-protein interactions involving occludin in TJ. (A,B) MDCK monolayers were exposed to ethanol control or 300 uM capsaicin for 45 min. (A) The cellular localization of the TJ proteins occludin and claudin-1 was examined by immunofluorescence. Scale bar: 10 um. (B) Western blot detection of occludin, tricellulin, Zo-1, claudin-1, E-cadherin and actin in the cytosol, membrane fractions and total cell extracts. The densitometic analysis of total proteins from three independent experiments performed with NIH ImageJ software. **represents p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Changes in Cldn1 expression in skin tumorigenesis . In the normal (not shown) and vehicle-treated ( a , after 12 weeks) epidermis, Cldn1 is localized in the basal and suprabasal layers; however in response to the two-stage chemical carcinogenesis protocol, the number of Cldn1-positive epithelial cells was progressively reduced starting from the basal layer and moving upwards at 2 ( b ), 4 ( c ), 8 ( d ) and 12 ( e ) weeks. Although a distinctly membranous Cldn1 association was maintained in the upper layers of the treated epidermis, as the number of Cldn1-negative epidermal cells in the lower epidermal layers increased, only sporadic Cldn1 localization was evident ( c-e ). The epidermal basal layer is indicated by a dotted line, and the suprabasal compartment is marked with a bracket ( a-c ); note that the basal layer is out of view in panels d and e ; the entire view is therefore the suprabasal compartment and is marked with a double-ended arrow.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
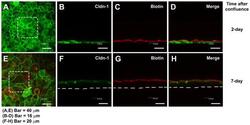
- Experimental details
- Fig. 2. Paracellular permeability of the cultured corneal epithelia was examined by use of the low molecular weight tracer, EZ-Link sulfo-NHS-LC-biotin. The tracer did not penetrate the epithelial sheet neither 1 day after confluence (7 days in cell culture) ( A-D ), nor in 7 day confluent epithelia ( E-H ), as indicated by the (C,G) lack of staining of biotinylated proteins at cell-cell boundaries in the lower layers of the epithelium. (D,H) Maximal projections of the merged channels, transverse optical sections of cultures stained for cldn-1 to immunolocalize TJ (green channel; B,D,F,H). Proteins biotinylated with the tracer are stained in red (C,D,G,H). (A,E) show the aspect of cldn-1 in xyz maximal projections of the stained cell cultures. Dashed squares show the fields examined in the transverse sections. Dashed lines indicate the basal side of the cultured epithelia. In A,E, Bar = 40 um; in B-D, Bar = 16 um; in F-H, Bar = 20 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
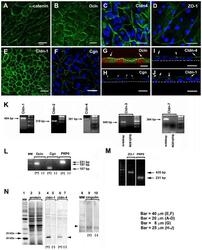
- Experimental details
- Fig. 3. TJ components detected in cultured RCE1(5T5) corneal epithelial cells. This was performed either by: ( A-J ) immunostaining, ( K-M ) end point RT-PCR, or ( N ) Western blot. (A) We used alpha-catenin as an indicator of the presence of adhesion complexes in the cultured epithelia. (B,G) Ocln, (D) ZO-1, (E) cldn-1, (C,I) cldn-4, and (F,H) cgn were easily located in cell borders of one day post-confluent cultures. (G-I) are transverse sections showing that TJs (arrows) were located at the cell boundaries of suprabasal cells. In (G), dashed line indicates the boundaries between a suprabasal cell and the basal cell layer; in (H-J) the dashed line indicate the basal side of the epithelium. Nuclei were stained either with propidium iodide or TO-PRO(r)-3. In 7-day confluent epithelia we found (K) the expression of cldn-1, -2, -4, but not of cldn-3 and -7; (L) cgn, ocln, and (M) ZO-1. Primers for cldn-3 and -7 were verified in PCR reactions using mouse kidney cDNA (K, m-kidney) that led to amplification products of the expected size and sequence. PR-P0 was used as an internal marker that does not change during the differentiation process. The antibodies were only appropriate to immunodetect cldn-1 and -4 and cgn (N); lanes 2 and 3 correspond to the loading control for the experiment. PCR reaction in the presence (+) or the absence of cDNA (-) (negative control). Results correspond to six duplicated experiments. A-D: Bar = 20 mum; E,F: Bar = 40 mum; G: Bar = 8 mum, and H-J: Bar
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
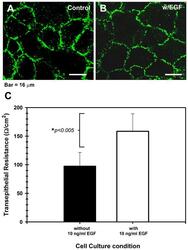
- Experimental details
- Fig. 5. Epidermal growth factor modulates the permeability of the corneal epithelial TJ. ( A ) Shows that the cldn-1 immunolocalization pattern is strong with punctuated discontinuities in cell cultures maintained in basal medium without EGF, while ( B ) cells cultured in the presence of 10 ng/ml EGF had a well-defined cell-cell contacts and a typical cldn-1 immunolocalization, with some discontinuities. The highest expression of cldn-1 correlated with ( C ) high TER values in the EGF-supplemented cultures which showed the tightest epithelial barrier. Effect of EGF was significant ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
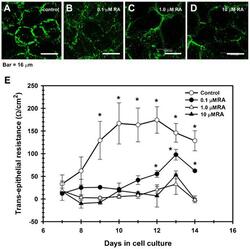
- Experimental details
- Fig. 6. TJ assembly is disrupted by retinoic acid (RA). In contrast with control cultures ( A ), corneal epithelial cells grown in the presence of RA, had a weak, discontinuous, punctuated pattern of cldn-1 at cell boundaries, which decreased in a concentration-dependent manner; and with cldn immunolocalization in cell cytoplasm in the RA-treated cells ( B-D ). ( E ) This change correlated with a partial (0.1 uM RA) or a complete blockage of the increase of TER observed in control cultures which were not treated with RA. ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 6 Overexpresssion of cofilin, LIMK and occludin, but not claudin-1, significantly diminishes the capsaicin-induced decrease in TER. (A,B) Transfectant monolayers were prepared in transwell inserts, exposed to 300 uM capsaicin, and subjected to TER measurements. Values represent mean +- S.D. Asterisks indicated significant difference from Vector control at the same time point; *, p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
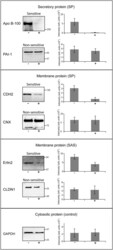
- Fig 2 Confirmation of cotransin sensitivity and cotransin resistance of selected proteins using SDS-PAGE/ immunoblotting. After transfection and treatment with cotransin (17 h, 30 muM) (+) or with DMSO (-) secretory and integral membrane proteins were isolated from HepG2 cells. The proteins were identified by SDS-PAGE/immunoblotting using specific primary antibodies and horseradish peroxidise-conjugated anti-mouse or anti-rabbit IgG as secondary antibodies. The cytosolic GAPDH protein does not contain a signal sequence and served as a control for a non-sensitive protein. As examples for secretory proteins (all SPs) cotransin-sensitive Apo B-100 and cotransin-resistant PAI-1 were used. For membrane protein possessing SPs, cotransin-sensitive CDH2 and cotransin-resistant CNX were analyzed. As examples for membrane proteins containing SASs, cotransin-sensitive Erlin2 and cotransin-resistant CLDN1 are shown. The immunoblots are representative of three independent experiments. The bar graphs shown at the right side of each immunoblot represent mean intensities of the respective protein bands of these three independent experiments +-SD (densitometric analysis using ImageJ).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
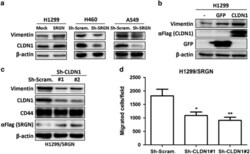
- Figure 5 SRGN elicits NSCLC aggressiveness mediated through Claudin-1 expression. ( a ) Cells were cultured in serum-free medium for 48 h, and western blotting was performed using anti-Vimentin (IF01, Calbiochem, Billerica, MA, USA) and anti-CLDN1 (71-7800, Invitrogen, Carlsbad, CA, USA). ( b ) H1299 cells were transiently transfected without or with vectors encoding GFP or CLDN1. Western blot analysis of designated proteins is shown. ( c ) H1299/SRGN cells stably harboring scramble-shRNA or CLDN1-shRNAs were subjected to western blot analysis of designated proteins. (d) Migration assay of H1299/SRGN cells stably harboring scramble-shRNA or CLDN1-shRNA is shown. Data are presented as the mean+-s.d. of three independent experiments. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
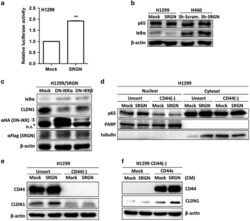
- Figure 6 SRGN/CD44 axis induces CLDN1 expression via NF-kappaB activation. ( a ) NF-kappaB reporter assay was performed in H1299/Mock and H1299/SRGN cells. ( b ) Cells were cultured in serum-free medium for 48 h, and subjected to western blot analysis using anti-IkappaBalpha (#4812, Cell Signaling Technology) and anti-p65 (sc-372, Santa Cruz Biotech). ( c ) H1299/SRGN cells were transiently transfected with vectors encoding dominant-negative IkappaB kinase alpha (DN-IKKalpha) or DN-IKKbeta and cultured in serum-free medium for 48 h, followed by western blot analysis of IkappaB and CLDN1. HA-fused DN-IKKs were detected by anti-hemagglutinin (sc-805, Santa Cruz Biotech). ( d ) Unsorted H1299 and CD44(-) cells stably harboring the Mock-control or SRGN-expressing vectors were cultured in serum-free medium for 48 h. Nuclear and cytosolic fractions were prepared for western blot analysis using anti-p65, anti-PARP (sc-7150, Santa Cruz Biotech) and anti-tubulin (GTX112141, GeneTex). ( e ) Cells described in d were cultured in serum-free medium for 48 h, and subjected to western blotting of CD44 and CLDN1. ( f ) H1299 CD44(-) cells stably harboring Mock-control or CD44s-expressing vectors were incubated in medium supplemented with CM collected from H1299/Mock or H1299/SRGN cells for 24 h, and subjected to western blot analysis of CLDN1. Data are presented as the mean+-s.d. of three independent experiments. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
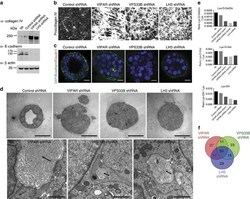
- Experimental details
- Figure 6 Knockdown mIMCD3 cells share molecular and cellular phenotypes. ( a ) Total cell lysates from mIMCD3 cell lines were analysed by western blot with antibodies against collagen IV, E-cadherin and beta-actin. Uncropped western blots are shown in Supplementary Fig. 13a . ( b ) mIMCD3 cell lines were grown on transwell supports for 2 weeks and immunostained with anti-collagen IV antibody. Figures show representative images of three independent experiments of n =3 views of multiple cells each. Scale bars, 10 mum. ( a , b ) Repeated three times. Representative images are shown. ( c ) mIMCD3 cell lines cultured for 3 days in 3D collagen I gels, immunostained with anti-E-Cadherin and anti-Claudin-1 antibodies. Scale bars, 10 mum. ( d ) EM of 3D cultured mIMCD3 cells (top panels), scale bars, 20 mum. Higher magnification of the areas of abnormal extracellular matrix deposits in kd cells (bottom panels). Representative images of n =9 (Control shRNA), n =7 (VIPAR shRNA), n =9 (VPS33B shRNA) and n =8 (LH3 shRNA) spheres are shown. Scale bars, 1 mum. ( e ) LC-MS-MS analysis for the relative quantification of the degree of Lys-O-GalGlc, Lys-O-Gal and Lys-OH in collagen IV from mIMCD3 cell lines. Two different control lines (wt and Control shRNA) and two different VIPAR shRNA clones were used. LH3 shRNA cells were used as a positive control. ( f ) Differentially expressed genes in kd mIMCD3 cell lines were detected using a moderated t -test implemented in the limma package in R 34 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 7 a Heatmap representing the expression values of mucin genes in colonocytes ( Muc ). Each row corresponds to one Muc gene. Each column corresponds to a single rat fed a normal-protein diet (NPD), or a high-protein diet (HPD). The color indicates the relative expression value (as indicated by the key) obtained from microarray experiment and normalized to the mean value in the NPD group. Mean expression values of the HPD and the NPD groups were compared with a t test. *: p < 0.05. b Relative expression values of defensin genes significantly differentially expressed (q < 0.01) in the colonocytes of rats fed a HPD when compared to a NPD. The expression values were obtained by microarray experiment and normalized to the mean value in the NPD group. Defb3, 4, 10, 15, 19, 22 and 30 (beta-defensin 3, 4, 10, 15, 19, 22 and 30). c Claudin 1 protein expression was assessed by western blot in colonocytes of rats fed an HPD or an NPD. Band intensity was quantified and normalized to the intensity of the band corresponding to actin. e - f Barrier function was evaluated with Ussing-chambers in distal colon of rats fed an NPD or an HDP. e Transmural resistance was measured for 15 min after mucosa mounting in the chamber. f FITC-dextran (FD4) transport from mucosal to serosal side was recorded during two hours and FD4 apparent permeability (FD4 P app ) was calculated. c - e For each parameter, mean values were compared with a t test. b - e Data presented on histograms are means +/- S.E.M
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
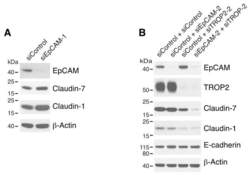
- Experimental details
- Figure 2 Both EpCAM and TROP2 regulate claudin expression in keratinocytes. HaCaT cells were transfected with control or EpCAM siRNAs ( A ), control, EpCAM, TROP2, or EpCAM and TROP2 siRNAs ( B ) using electroporation. 72 h after transfection, cell lysates were prepared, resolved using SDS-PAGE and immunoblotted with anti-EpCAM, anti-TROP2, anti-claudin-7 or anti-claudin-1 to assess corresponding protein levels. beta-actin was used as a loading control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
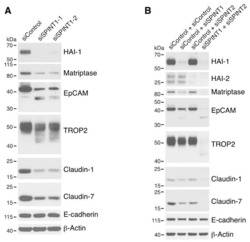
- Experimental details
- Figure 5 HAI-1 and HAI-2 regulate EpCAM and TROP2 cleavage and claudin levels. HaCaT cells were transfected using electroporation with control siRNA or one of two different SPINT1 siRNAs (siSPINT1-1 and siSPINT1-2) ( A ), control, SPINT1, SPINT2, or SPINT1, and SPINT2 siRNAs ( B ). After 72 h, cell lysates were prepared, subjected to electrophoresis and analyzed via Western blotting for HA1-1, HAI-2, matriptase, EpCAM, TROP2, claudin-1, claudin-7 and E-cadherin. beta-actin was used as a loading control. Representative data from 1 of 3-4 experiments is shown.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
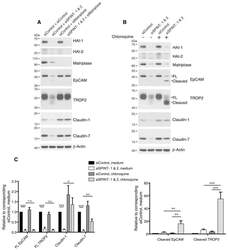
- Experimental details
- Figure 6 HAIs act via matriptase to regulate degradation of EpCAM, TROP2, and claudins in lysosomes. HaCaT cells were transfected using electroporation with control or SPINT1 plus SPINT2, matriptase, or SPINT1, SPINT2 plus matriptase siRNAs ( A ), or control or SPINT1 and SPINT2 siRNAs ( B ). Before being harvested at 72 h after transfection ( A , B ), cells were treated with or without 100 muM chloroquine for 20 h ( B ). RIPA cell lysates were resolved using SDS-PAGE and immunoblotted with anti-HAI-1, anti-HAI-2, anti-matriptase, anti-EpCAM anti-TROP2, anti-claudin-1, or anti-claudin-7. beta-actin was used as a loading control. Representative data from 1 of 5 experiments is shown. ( C ) Band intensities corresponding to full-length (FL) EpCAM, FL TROP2, claudin-1 and claudin-7 (left panel) and cleaved EpCAM and cleaved TROP2 (right panel) were quantified and normalized to beta-actin. Data are plotted as ratios (means +- SEM) relative to corresponding untreated siControls ( n = 5). A two-way ANOVA was used to calculate p values for multiple group comparisons to assess mean differences between groups (* p < 0.05, ** p < 0.01, *** p < 0.001 or 0.0001, n.s. p > 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
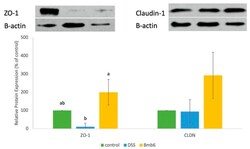
- Experimental details
- Figure 7 Western blot analysis of the expression ZO-1 and claudin-1 in DSS-induced colitis mice. Data represent the mean +- standard deviation ( n = 6 mice per group). Tukey's multiple comparison test was performed, and different lowercase letters indicate statistically significant differences ( p < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
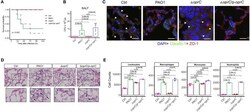
- Experimental details
- Figure 3 Delta oprC infection decreased mouse mortality and lung damage following P. aeruginosa infection. (A) C57BL6 mice were intranasally challenged with wild type PAO1, Delta oprC , and complemented strain at 2 x 10 7 CFU in 25 muL PBS; moribund mice were killed to obtain survival data ( n = 6). (B) Bacterial burdens in the BALF were determined 24 h after bacterial infection ( n = 6). (C) Representative images of immunofluorescence staining of the lungs infected with bacteria for Claudin-1 co-stained with ZO-1 and DAPI ( n = 3). Arrowheads show the membrane localization of Claudin-1. Scale bars, 20 mum. (D) Representative histological views of the lungs of mice 24 h after intranasal infection with bacteria by H&E staining (insets show the enlarged views) ( n = 3). Arrows show examples of neutrophil infiltration areas. Scale bars, 50 mum. (E) Quantification of leukocytes, macrophages, monocytes, and neutrophils numbers per 30,000 cells collected from lung infected with bacteria for 24 h. Error bars represent the mean +- s.d. Kolmogorov-Smirnov test was performed for survival curves analysis. One-way ANOVA with a post-hoc Tukey test was performed for comparison of means of groups in other cases.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
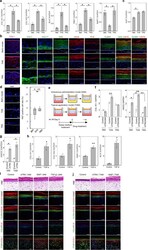
- Experimental details
- Fig. 4 Analysis reveals that the THS model promoted function through tension homoeostasis. a , c Real-time PCR analysis of dermal ECM proteins and epithelial-mesenchymal interaction-related genes ( COL1A1 , COL1A2 , FBN1 , ELN , MMP1 and KGF ) of HSEs. *P < 0.001, as assessed by Dunnett's test following two-way analysis of variance (ANOVA; P < 0.001); error bars represent the standard deviation. ( b ) Immunohistochemical analyses of epidermal and dermal morphogenesis markers in skin equivalents. These samples were stained with COL1, COL7, COL17, CK5, CK10, CLDN1, FLG and Ki67 antibodies. The dotted lines indicated boundary between epidermis and dermis. Scale bars, 50 um. d Analysis of the Ki67-positive basal keratinocytes in skin equivalents. Immunohistochemical analyses (left) and Ki67-positive cell ratio (right) were shown. Scale bars, 20 um. *P < 0.01 and **P < 0.001, as assessed by Dunnett's test following two-way analysis of variance (ANOVA; P < 0.001). e Schematic representation of the methods used for evaluating molecular activity. Functional molecules were applied to skin equivalents by dissolving in medium (as shown by IAM) or lotion (as shown by TAM) and treating for 6 h to 5 days. f The reactivity to ATRA in skin equivalents (Bell's model, THS model, and TRS model) was evaluated by mRNA expression levels of HAS3 and COL1A1 in the IAM assay model. *P < 0.01 and * *P < 0.001, as assessed by Tukey-Kramer test following two-way analysis of variance (ANOVA; P < 0.001);
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Intestinal barrier was destroyed in NEC patients and mice. a Representative sections from ileac tissue of Ctrl or NEC patients stained with anti-claudin-1, anti-occludin, and anti-ZO-1 antibodies. Neonatal intestinal atresia patients were used as controls. b Statistical analysis of the average optical density of immunohistochemical staining images of ( a ) (n = 5 per group). c and d Western blot analysis of claudin-1, occludin, and ZO-1 protein levels in small intestinal epithelial cells of NEC mice and relative gray calculation (n = 5 ~ 6 per group, representative data from four independent experiments). e The concentration of FITC-dextran 4 kDa (FD4) in the serum of Ctrl or NEC mice. Ctrl, dam-fed, n = 5 ~ 6 per group. Four independent experiments were performed. Data are shown as mean values +- SD. Statistical analyses were performed with Student's two-tailed unpaired t -test. * p < 0.05; ** p < 0.01; n.s., not significant. Scale bars represent 20 mum in ( a )
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
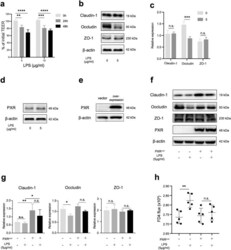
- Experimental details
- Fig. 6 PXR protected barrier function from LPS damage. a TEER changes in Caco-2 cell monolayers treated with LPS for 24 or 48 h. b and c Western blot analysis of claudin-1, occludin, and ZO-1 protein levels in Caco-2 cells treated with LPS (n = 4 per group, representative data from three independent experiments). d Western blot analysis of PXR in Caco-2 treated with LPS. e Confirmation of PXR overexpression by Western blot. f and g Western blot analysis of the components of tight junctions (Claudin-1, Occludin, and ZO-1) in Caco-2 cells treated with LPS for 48 h (n = 4 per group, representative data from three independent experiments). h FD4 flux of basolateral medium was measured after Caco-2 cells were treated with LPS or not treated for 48 h (n = 5 per group, representative data from four independent experiments). Data are expressed as mean values +- SD. Statistical analyses were performed with Student's two-tailed unpaired t -test in ( c and g ), * p < 0.05; ** p < 0.01; n.s., not significant; TEER, transepithelial electrical resistance; FD4, FITC-dextran 4 kDa
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
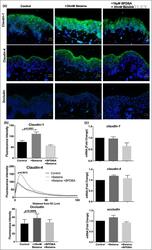
- Experimental details
- 4 Figure Betaine increases tight junction protein expression in human organ-cultured skin. (a) Claudin-1, claudin-4 and occludin immunofluorescence in ex vivo human skin cultured for 72 h under control conditions, in the presence of 35 mmol L -1 betaine, 10 mumol L -1 N -(1-benzyl-4-piperidinyl)-2,4-dichlorobenzamide ( BPDBA ), and both 10 mumol L -1 BPDBA and 35 mmol L -1 betaine. (b) Quantification of fluorescence intensity showed that the presence of BPDBA mitigated the increase in expression of all three tight junction proteins caused by betaine. (c) Claudin-1, claudin-4 and occludin gene expression was not affected by the presence of betaine. Data are presented as the mean +- SEM , two-way anova , n = 7. Bars = 50 mum. SC , stratum corneum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
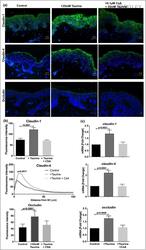
- Experimental details
- 5 Figure Taurine increases tight junction protein expression in human organ-cultured skin. (a) Claudin-1, claudin-4 and occludin immunofluorescence in ex vivo human skin cultured for 72 h under control conditions, in the presence of 35 mmol L -1 taurine and following the addition of 0*1 mumol L -1 ciclosporin A (CsA) and 35 mmol L -1 taurine. (b) Quantification of fluorescence intensity showed that the presence of CsA mitigated the increase in expression of all three tight junction proteins caused by betaine. (c) Claudin-1, claudin-4 and occludin gene expression was similarly affected by the presence of taurine. Data are presented as the mean +- SEM , two-way anova , n = 7. Bars = 50 mum. SC , stratum corneum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
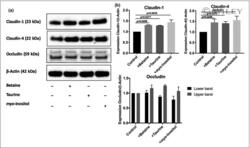
- Experimental details
- 7 Figure Immunoblotting of tight junction proteins in human primary keratinocytes. (a) Immunoblotting analysis showed that the expression of claudin-1 and claudin-4 was induced in differentiated normal human epidermal keratinocytes incubated with 5 mmol L -1 organic osmolytes. (b) Densitometric quantification showing the ratio of tight junction protein to beta-actin protein expression. Data are presented as the mean +- SEM , two-way anova , n = 3.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
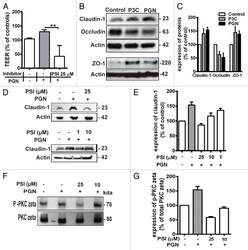
- Experimental details
- Figure 4. ( A ) Pretreatment with 25 uM PKC zeta pseudosubstrate inhibitor (PSI) significantly prevented the increase in TEER induced by PGN. All TEER values are expressed as percentage relative to control (the baseline value was > 520 Omega*cm 2 ). Values represent mean +- SD * P < 0.05, ** P < 0.01, *** P < 0.001. The data were analyzed using one-way analysis of variance (ANOVA). All the experiments were performed atleast 3 times during different days with number of replicates (n = 4). Western blotting ( B ) for claudin-1, occludin and ZO-1. Upregulation of claudin-1 and ZO-1 was observed in Calu-3 cells after treatment with P3C and PGN. Occludin was decreased. The images are representative of 3 independent experiments. beta-actin was used as a lane loading control. The number of replicates is (n = 2). ( C ) the corresponding expression levels of claudin-1, occluding and ZO-1 are shown as bar graphs. ( D ) western blotting for claudin-1. The PSI inhibited the claudin-1 expression induced by PGN treatment in a concentration dependent manner (25 uM, 10 uM and 1 uM). E, the corresponding expression levels of claudin-1 are shown as bar graphs. ( F ) western blotting for phospho-PKC zeta in Calu-3 cell monolayers treated with PGN. Stimulation by PGN increased phosphorylation of PKC zeta in the treated cells which was inhibited by pretreatment with PSI 25 uM and 10 muM. Expression of phospho-PKC zeta and total PKC zeta was examined using western-blot analyses of total cell lysate
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 10.1371/journal.pone.0168346.g001 Fig 1 Urine cells on cytospin slides stained for podocalyxin (PCX, red), claudin-1 (CL1, green), and Dapi (blue). (A-D) A Faby podocyte which is PCX+/CL1- and shows characteristic vacuolated cytoplasm. (E-H) A urine cells which is PCX+/CL1+ consistent with a parietal epithelial cell (PEC) phenotype. (I-L) An apoptotic podocyte (PCX+/CL1-) in the urine from a Fabry patient with shrunken cytoplasm and small nucleus (arrow) compared to its adjacent cells. (M-P) A podocyte (PCX+/CL1-) in the urine from a healthy subject does not show vacuolated cytoplasm.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Immunophenotype of perineurial cells, arachnoid, and suprachoroidalcells. Immunohistochemical profile of perineurial cells (panels A-E),optic nerve sheath/arachnoid (F-J), and suprachoroidal cells (K-O,indicated by black arrows). Panel A shows ciliary nerves and sclera ofthe posterior eye with an inset showing endoneurium (endon.),perineurium (perin.), and epineurium (epin.) of a ciliary nerve.Perineurial cells are immunoreactive for 2 EMA antibodies (B, C), Glut-1(D), and claudin-1 (E). Panel F shows optic nerve sheath with arachnoid,which is immunoreactive for both EMA antibodies (G, H), weakly reactivefor claudin-1 (J), and negative for Glut-1 (I). Panel K shows retina,choroid, thin suprachoroidal cells, and sclera. Suprachoroidal cells,indicated by black arrows, are immunoreactive for EMA (L, M), and, inthese examples, immunonegative for Glut-1 (N) and claudin-1 (O).Examples are from multiple samples with panels B-E and L-O at 600x(panel E, O scale bars = 20 um) and panels G-J at 400x (panel J scalebar = 20 um). Lower-power, PAS-stained images (A, F, K) at 100x.Abbreviations: EMA, epithelial membrane antigen; PAS, periodicacid-Schiff.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. A heat map of staining intensity of suprachoroidal fibroblasts for EMAantibodies (CellMarque, Ventana), CD34, claudin-1, and Glut-1 in theposterior pole, equator, and anterior pole of the eye. Each antibody hasspecimens 1-8 listed in columns with anatomic region of interest listedin rows. A scale for staining intensity is present at the right of thefigure. Abbreviation: EMA, epithelial membrane antigen.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Increase in plasma membrane surface localization of the D97S mutant by endocytosis inhibitor or primaquine treatment. ( A , B ) MDCK cells stably expressing the FLAG-tagged D97S mutant were treated with MDC and MbetaCD for 3 h. After collecting cell lysates, the aliquots were blotted with anti-FLAG and anti-beta-actin antibodies. The band densities of proteins are represented relative to the values with 0 muM. ( B ) The cells were stained with anti-FLAG (CLDN16) and anti-ZO-1 antibodies. ( C , D ) MDCK cells stably expressing the FLAG-tagged D97S mutant were incubated with primaquine (PQ, 100 muM). The biotinylated proteins were blotted with anti-FLAG and anti-CLDN1 antibodies. ( E ) The cell lysates of MDCK cells expressing the WT or D97S mutant were blotted with anti-FLAG and anti-beta-actin antibodies. The band densities of proteins are represented relative to the values at 0 h. The full-length blot images are shown in Supplementary Figs S6 - S8 . ( F ) The cells were incubated with anti-FLAG (CLDN16) and anti-ZO-1 antibodies. The scale bar indicates 10 mum. n = 4 in four independent experiments. ** P < 0.01 and * P < 0.05 compared with 0 muM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
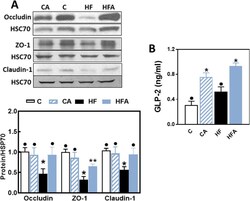
- Experimental details
- Fig. 3 AC mitigates HFD-mediated decrease in TJ protein expression and modulates hormones that regulate TJ structure and function. Mice were fed a control diet (empty bars), the control diet supplemented with 40 mg AC/kg body weight (dashed bars), a HFD (black bars), or the HFD supplemented with 40 mg AC /kg body weight (blue bars) for 14 weeks. A- Ileum protein levels of occludin, ZO-1 and claudin-1 were measured by Western blot. Bands were quantified and values normalized to HSC70 levels (loading control). Values were referred to those of the control group (empty bars). B - Plasma GLP-2 concentrations were measured by ELISA. Results are shown as mean +- SE of 6-8 animals/treatment. Values having different superscripts are significantly different (p < 0.05, One-way ANOVA test). (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.) Fig. 3
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
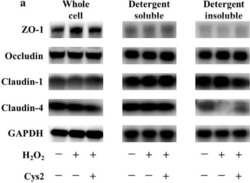
- Experimental details
- Fig. 3 Cystine (Cys2) improved the decrease of the ratio of claudin-4 in the detergent-insoluble fractions (IS) to soluble fractions (S). Caco-2 cells were separated the detergent-insoluble fractions and soluble fractions after 2 h of incubation with 0.5 mM H 2 O 2 . a Whole cell extracts, detergent-soluble fractions and insoluble fractions of Caco-2 cells were immunoblotted for ZO-1, occludin, claudin-1, claudin-4, and GAPDH. The expression level of the TJ proteins zonula occludens-1 (ZO-1) ( b ), occludin ( c ), claudin-1 ( d ), and claudin-4 ( e ) in the whole cell extracts was measured and the ratio of TJ proteins in the detergent-insoluble fractions to soluble fractions was calculated. Control cells (Con) were monolayers pretreated with DMEM and not incubated with 0.5 mM H 2 O 2 . Protein expression was normalized to GAPDH levels and expressed as mean fold change relative to the Con group. Values are represented as mean +- SEM ( n = 6)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
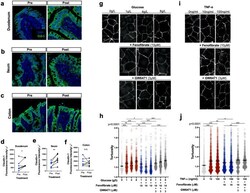
- Experimental details
- Figure 4 Fenofibrate upregulates expression of tight junctions on intestinal epithelial cells. Immunofluorescent staining of claudin-1 (CLD-1) expression in the ( a ) duodenum, ( b ) ileum, and ( c ) colon of diabetic dogs pre- and post-fenofibrate administration. ( d - f ) Semi-quantitation of CLD-1 protein expression in the intestinal compartments. ( g ) Representative ZO-1 staining and ( h ) quantification of barrier tortuosity in Caco-2 cells treated with different concentrations of glucose + 10 muM fenofibrate + 3 muM GW6741 or ( i , j ) TNF-alpha + fenofibrate + GW6741 for 24 h. * P < 0.05, ** P < 0.01, *** P < 0.001 by ( a - f ) mixed models ANOVA and ( g - j ) one-way ANOVA with Tukey's multiple comparison test. N.S., not significant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Claudin1 immunohistochemistry in the distal colon. Representative immunohistological images are displayed below at 20X objective. All DiNP treatment groups were compared to control. Quantification of CLDN1 is in the graph on the right. The data are presented as means +- standard error of the mean (SEM). n = 4-6 samples/group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
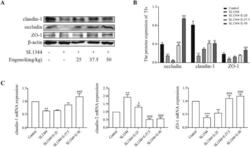
- Experimental details
- Eugenol improved the expressions of the duodenal tight junction proteins ZO-1, claudin-1 and occludin in Salmonella -infected broilers. (A) WB analysis: the protein bands of critical proteins. (B) WB analysis: the quantification of the protein levels by determining the band intensity and normalized to beta-actin band levels. (C) The mRNA expressions of tight junctions (ZO-1, claudin-1 and claudin-2). All experiments were repeated in triplicates. The data are expressed as the Mean +- SE (n = 15). ** P < 0.01, *** P < 0.001 vs. Control. # P < 0.05, ## P < 0.01, ### P < 0.001 vs. SL1344 infection. Figure 5
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
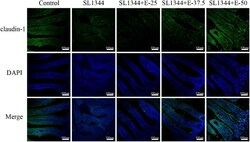
- Experimental details
- Eugenol improved the expression of tight junction protein claudin-1 in the duodenum of Salmonella -infected broilers by immunofluorescence. Green fluorescence represents claudin-1, and blue (DAPI dye) represents the nuclei. Figure 6
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
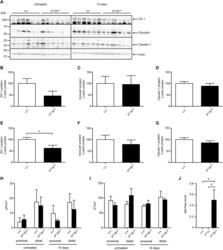
- Experimental details
- FIGURE 5. A, Representative immunoblots of colon lysates from untreated (left panel) and treated (7 days DSS and 7 days recovery, 14 days, right panel) &plus&sol&plus and fr CR &solfr CR rats with ZO-1, occludin, claudin-1, and actin. Protein quantification of corresponding immunoblots for untreated (B-D) and treated (14 days, E-G) animals for (B, E) ZO-1, (C, F) occludin, and (D, G) claudin-1; n = 6 for each genotype. Actin was used as loading control. H, Short circuit current (I sc ) and (I) transepidermal resistance (TER) in proximal and distal colon of &plus&sol&plus and fr CR &solfr CR animals (untreated, n = 3 per genotype) or following 7 days of DSS treatment and 7 days of recovery (14 days, n = 3 per genotype). J, Diarrhea score in untreated &plus&sol&plus , fr CR &sol&plus , and fr CR &solfr CR animals (n = 4 per genotype). &ast P < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 7 D88 mitigates the morphologic alterations of the intestinal lining and attenuates intestinal inflammation. a Representative confocal image ( n = 2) of distal ileum with Claudin-1 immunofluorescence staining. The experiment was repeated independently two times with similar results. Samples for confocal imaging were harvested 22 h post-burn and infection. Green fluorescence represents Claudin-1, and blue fluorescence represents the DAPI stain, the white line represents the scale bar (100 um). b Levels of tumor necrosis factor (TNF-alpha) in the distal ileum 22 h post-burn and infection. The total protein was isolated from the distal ileum, and the concentration of TNF-alpha in the sample was quantified using ELISA. c The levels of interleukin (IL-6) in the distal ileum were also quantified using ELISA. b , c Data represent at least n = 5, each dot in the bars represents data from one mouse. The error bars denote +- SEM. One-way ANOVA followed by Tukey post-test was applied. The no-treatment group data (Burn + PA14) were compared to the vehicle-treated and the D88-treated groups. *, **, and *** indicates significant differences compared to the control at P < 0.05, P < 0.01, and P < 0.001, respectively. ns represents no significant difference.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
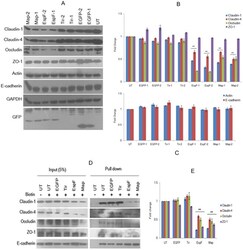
- Experimental details
- Figure 4 Constitutive expression of EspF and Map depletes tight junction proteins. ( A ) Cell lysates derived from untransfected, EGFP, EGFP-Tir, EGFP-EspF and EGFP-Map cell lines were separated by electrophoresis on 12% SDS-polyacrylamide gels and transferred to PVDF membranes. Equal amounts of cell lysates were loaded after normalizing with GAPDH. Blots were probed with the indicated primary antibodies and HRP-conjugated secondary antibodies. Full-length blots are presented in Supplementary Figure S3 . ( B , C ) The x-ray films were scanned and quantitative analysis was performed by measuring band intensities using ImageJ software. The expression of different tight junction proteins in these cell lines was normalized with respect to untransfected cells and fold changes were calculated. Data are represented as means +- s.e.m. from six independent experiments using two biological replicates for all cell lines; **represents p-values < 0.001. ( D ) The cell surface proteins were labeled with membrane impermeable Sulfo-NHS-Biotin and immobilized on streptavidin-agarose beads. The beads were washed and subjected to SDS-PAGE. E-cadherin was used as a loading control for cell surface proteins. Full-length blots are presented in Supplementary Figure S4 . ( E ) The levels of cell surface proteins were estimated by densitometric scanning of x-ray films using ImageJ software. Shown are fold changes with respect to untransfected cells. Error bars represent means +- s.e.m. from three i
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
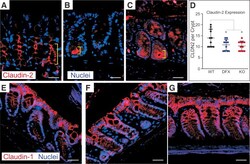
- Experimental details
- Figure 10 Claudin-2 expression is limited to the base colon of crypt in NKCC1 WT/DFX and NKCC1 DFX/DFX mice. Representative IF images of NKCC1 WT/WT , NKCC1 WT/DFX , and NKCC1 DFX/DFX mouse colon sections stained with ( A-C ) claudin-2 and ( D-F ) claudin-1 antibodies (red). Sections also show DAPI (blue) or nuclei staining. ( D-F ) In contrast, claudin-1 expression at the lateral membrane was similar in all 3 genotypes. Scale bars : 20 mum. Number of claudin-2-positive cells per crypt were counted on 5 fields, 11 crypts per genotype. There was a significant decrease in the number of cells expressing claudin-2 in the mutant mice. P = .043 for NKCC1-DFX and P = .019 for KO. There was no difference between NKCC1-DFX and NKCC1 KO mice ( P = .93). ( G ) Data are shown as whisker boxes and statistical significance was calculated by 1-way analysis of variance followed by Tukey posttests. CLDN2, claudin-2.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 2: Endogenous clds are variably distributed between the apical tight junction and lateral membrane in MDCK II cells. (A) Top, Maximum-intensity projections of immunofluorescence confocal colocalization of cldn2, cldn1, ZO-1, and merge (left to right panels) show that cldn1 is more laterally disposed than cldn2 or ZO-1. Bottom, Colocalization of cldn4, cldn3, ZO-1, and merge (left to right panels) reveals cldn4 and cldn3 at the lateral membrane as well as colocalized with ZO-1. Arrows in enlargement show apparent concentrations of lateral cldns likely related to lateral membrane infoldings (see Supplemental Figure S1). (B) z -Axis images as in A show ZO-1 focused at the apical junctional region and cldns colocalized with ZO-1 but also distributed along the lateral membrane.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
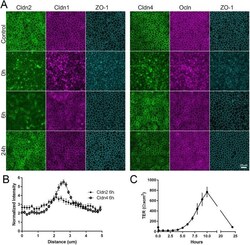
- Experimental details
- FIGURE 9: Cldn2 relocalizes to cell contacts after calcium switch assay more slowly than does cldn1 or cldn4. (A) MDCK II cells cultured on semipermeable membranes were incubated in low-calcium media for 15 h; calcium was added back, and cells were collected and fixed for immunofluorescence at various times after readdition. TER was measured in parallel. (A) Immunofluorescence analysis--(left panel) cldn2, cldn1, and ZO-1 and (right panel) cldn4, ocln, and ZO-1 in untreated monolayers (top images), after 15 h in low calcium (second row), 6 h after calcium readdition (third row), and 24 h after calcium readdition--reveals that by 6 h of calcium return, most the cldn1, cldn4, ocln, and ZO-1 but not cldn2 has returned to cell contacts; the difference in cell-contact localization between cldn2 and 4 is quantified in B. (C) TER measurements show that measurable TER is lost in low calcium but overshoots control levels by more than 10-fold before returning to control values at 24 h.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
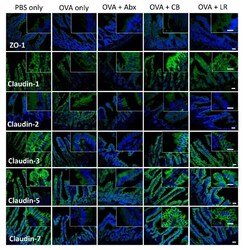
- Experimental details
- Figure 6 Representative confocal images of TJ-related proteins. Cryosections of the jejunal tissue were immunolabeled for ZO-1 and claudin-1, -2, -3, -5, and -7 (in green). Nuclei were visualized with DAPI (in blue). Size bars, 50 mum (small) and 20 mum (large). Abx, antibiotics; CB, C. butyricum ; LR, L. reuteri .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
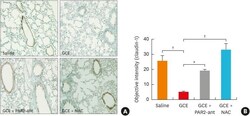
- Experimental details
- Fig. 6 GCE significantly disturbs claudin-1 expression in a mouse model. Claudin-1 expression is significantly restored by PAR2-ant and NAC treatment in the lung, as indicated by immunohistochemistry (200 x magnification) (A) and intensity quantification (B). GCE, German cockroach extract; PAR2-ant, protease-activated receptor 2-antagonist; NAC, N-acetylcysteine. * P < 0.01; + P < 0.001. The experiments were repeated 5 times.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Cholesterol is enriched in the TJ-containing PM fraction. (A) C1L cells were fixed and stained with an anti-claudin-1 pAb. (B) C1L cells were fixed and stained with an anti-claudin-1 pAb (green) and phalloidin (red). Bars, 10 um. (C) Immunoblot analysis of the PM and IM fractions of C1L cells. Each membrane fraction (5 ug) was separated by SDS-PAGE, transferred to a nitrocellulose membrane, and probed with antibodies against the indicated marker proteins (left). Coomassie brilliant blue (CBB) staining is shown on the right. (D) Positive ion mass spectra of SM species in the PM fractions of L and C1L cells. The SM molecular species corresponding with each peak are indicated. The x and y axes show the total carbon chain length and the number of carbon-carbon double bonds of individual lipid molecular species, respectively. (E) Quantification of the indicated SM species in the PM fractions of L cells and C1L cells. (F) Quantification of the cholesterol-to-phospholipid ratio in the PM fractions of L and C1L cells. (G) Immunoblot analysis of the DRM and non-DRM fractions of WT and alpha-catenin-KO EpH4 cells using pAbs against the DRM marker proteins claudin-3 and caveolin-1. Results in C, D, and G are representative of three independent experiments. (H) Quantification of the ratio of the claudin-3 level in the DRM fraction to that in the non-DRM fraction in WT and alpha-catenin-KO EpH4 cells. Error bars show SD calculated based on three independent experiments (Student'
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Immunofluorescence staining of tight junction (ZO-1, Occludin and Claudin-1) in colon. White arrows point out the tight junction disruption (600 times of magnification). C, control, HFD, high-fat diet, GC, geniposide and chlorogenic acid combination, QHD, Qushi Huayu Decoction, NaB, sodium butyrate.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
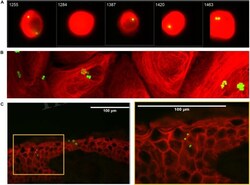
- Experimental details
- FIGURE 2 Internalization of GFP S. aureus by Normal Human Epidermal Keratinocytes (NHEK). (A) Representative Amnis (r) Image Stream Analysis of NHEK co-cultured with 10 7 CFU/mL FITC LiSA for 1 h at 37degC, then treated for 30 min with 2% penicillin/streptomycin (P/S) to eliminate extracellular bacteria and cultured for a further 4 h prior to staining and analysis. Red stain: Alexa Fluor (r) 647 mouse anti-human cytokeratin 14/15/16/19. Green stain: FITC S. aureus. (B) Representative confocal microscopy 2D image of NHEK after co-culture with 10 7 CFU/mL GFP S. aureus for 1 h at 37degC. (C) Representative immunofluorescent staining of healthy human skin explant after 3 h infection with GFP S. aureus (10 7 CFU/mL) at 37degC. Red: anti-rabbit claudin-1 antibody. Green: GFP S. aureus. Yellow box (left panel) highlights area of right panel magnified. All data are representative of three independent experiments. Images taken at 40x magnification. Scale bars represent 100 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 JMJD1C regulates AMPK in a CAMKK2-dependent manner. (A,B) JMJD1C does not affect activation of LKB1 or levels of its activation partners STRAD and MO25 in NRCMs. The cells were infected with adenovirus expressing Jmjd1c , sh Jmjd1c , or the control constructs for 24 h, followed by Ang II treatment for an additional 24 h. n = 3 in each group. (C) Effects of JMJD1C on CAMKK2 expression. NRCMs were treated as in (A) . ** p < 0.01 vs. Ad-Ctrl or shCtrl. n = 3 in each group. (D,E) Effects of JMJD1C on CAMKK2 mRNA expression. NRCMs were treated as in (A) . ** p < 0.01 vs. Ad-Ctrl or shCtrl. (F) Chromatin immunoprecipitation (ChIP) assay showing the binding of JMJD1C, JMJD2A, and JMJD3 on the FHL1, beta-MHC, and CAMKK2 promoters. IgG was used for negative control, and the enrichment was normalized to the input. *** p < 0.001 vs. IgG. (G) ChIP assay showing that JMJD1C overexpression reduced H3K9me1 enrichment at the CAMKK2 promoter in cardiomyocytes. *** p < 0.001 (H) . The inhibition of CAMKK2 blocks the effects of JMJD1C on AMPK activation in NRCMs. The cells were treated with adenovirus expressing sh Jmjd1c or shCtrl; then the cells were treated with Ang II and the CAMKK2 inhibitor STO-609 (20 muM) for an additional 24 h. All results are from three independent experiments. ** p < 0.01 vs. Ad-shCtrl+PBS; ## p < 0.01 vs. Ad-shCtrl+STO-609. n = 3 in each group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. Effects of LMWF on the distribution of claudin-1 in the seminiferous epithelium. ( A ) Non-specific staining was detected only in the basal and adluminal compartments of seminiferous epithelium of control sections - negative control. ( B - C ) Immunohistochemical staining of RT transverse cross-sections treated with 0.91% w/v NaCl. ( D - F ) Immunostaining of claudin-1 following treatment with LMWF demonstrated no difference in the distribution of claudin-1 when compared to the control group treated with 0.91% w/v NaCl. Hematoxylin was used for counterstaining.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot