Antibody data
- Antibody Data
- Antigen structure
- References [6]
- Comments [0]
- Validations
- Western blot [4]
- Immunocytochemistry [2]
- Immunohistochemistry [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-154 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Cyclin A2 Monoclonal Antibody (E23.1)
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human, Mouse, Rat, Bovine
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- E23.1
- Vial size
- 100 µg
- Concentration
- 1 mg/mL
- Storage
- -20°C
Submitted references Lysophosphatidic Acid Mediates Imiquimod-Induced Psoriasis-like Symptoms by Promoting Keratinocyte Proliferation through LPAR1/ROCK2/PI3K/AKT Signaling Pathway.
Survivin as a potential therapeutic target of acetylsalicylic acid in pituitary adenomas.
Single-tubule RNA-Seq uncovers signaling mechanisms that defend against hyponatremia in SIADH.
APOLLON protein promotes early mitotic CYCLIN A degradation independent of the spindle assembly checkpoint.
Differential apoptosis by gallotannin in human colon cancer cells with distinct p53 status.
Disrupting Skp2-cyclin A interaction with a blocking peptide induces selective cancer cell killing.
Kim D, Kim HJ, Baek JO, Roh JY, Jun HS
International journal of molecular sciences 2021 Oct 5;22(19)
International journal of molecular sciences 2021 Oct 5;22(19)
Survivin as a potential therapeutic target of acetylsalicylic acid in pituitary adenomas.
Németh K, Szücs N, Czirják S, Reiniger L, Szabó B, Barna G, Karászi K, Igaz P, Zivkovic V, Korbonits M, Patócs A, Butz H
Oncotarget 2018 Jun 26;9(49):29180-29192
Oncotarget 2018 Jun 26;9(49):29180-29192
Single-tubule RNA-Seq uncovers signaling mechanisms that defend against hyponatremia in SIADH.
Lee JW, Alsady M, Chou CL, de Groot T, Deen PMT, Knepper MA, Ecelbarger CM
Kidney international 2018 Jan;93(1):128-146
Kidney international 2018 Jan;93(1):128-146
APOLLON protein promotes early mitotic CYCLIN A degradation independent of the spindle assembly checkpoint.
Kikuchi R, Ohata H, Ohoka N, Kawabata A, Naito M
The Journal of biological chemistry 2014 Feb 7;289(6):3457-67
The Journal of biological chemistry 2014 Feb 7;289(6):3457-67
Differential apoptosis by gallotannin in human colon cancer cells with distinct p53 status.
Al-Ayyoubi S, Gali-Muhtasib H
Molecular carcinogenesis 2007 Mar;46(3):176-86
Molecular carcinogenesis 2007 Mar;46(3):176-86
Disrupting Skp2-cyclin A interaction with a blocking peptide induces selective cancer cell killing.
Ji P, Sun D, Wang H, Bauzon F, Zhu L
Molecular cancer therapeutics 2007 Feb;6(2):684-91
Molecular cancer therapeutics 2007 Feb;6(2):684-91
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
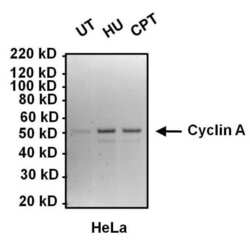
- Experimental details
- Western blot analysis of Cyclin A was performed by loading 10 µg of lysates from HeLa cells left untreated (UT), or treated with 2mM hydroxyurea (HU) or 100 nM camptothecin (CPT) for 16 hours onto a 4-12% Bis-Tris polyacrylamide gel. Proteins were transferred to a nitrocellulose membrane and blocked with 5% BSA in TBST for at least 1 hour. Membranes were probed with a Cyclin A monoclonal antibody (Product # MA1-154) at a dilution of 1:1000 overnight at 4°C on a rocking platform, washed in TBST, and probed with an HRP-conjugated goat anti-mouse IgG secondary antibody (Product # 31430) at a dilution of 1:20,000 for 1 hour. Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34076).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
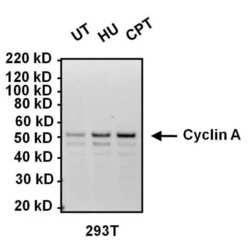
- Experimental details
- Western blot analysis of Cyclin A was performed by loading 10 µg of lysates from HEK293T cells left untreated (UT), or treated with 2mM hydroxyurea (HU) or 100 nM camptothecin (CPT) for 16 hours onto a 4-12% Bis-Tris polyacrylamide gel. Proteins were transferred to a nitrocellulose membrane and blocked with 5% BSA in TBST for at least 1 hour. Membranes were probed with a Cyclin A monoclonal antibody (Product # MA1-154) at a dilution of 1:1000 overnight at 4°C on a rocking platform, washed in TBST, and probed with an HRP-conjugated goat anti-mouse IgG secondary antibody (Product # 31430) at a dilution of 1:20,000 for 1 hour. Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34076).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
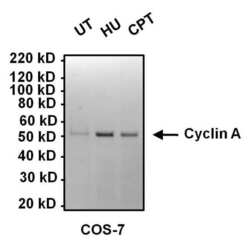
- Experimental details
- Western blot analysis of Cyclin A was performed by loading 10 µg of COS7 cell lysates from cells left untreated (UT), or treated with 2mM hydroxyurea (HU), or 100 nM camptothecin (CPT) for 16 hours per lane onto a 4-12% Bis-Tris polyacrylamide gel. Proteins were transferred to a nitrocellulose membrane and blocked with 5% BSA in TBST for at least 1 hour. The membrane was probed with a Cyclin A monoclonal antibody (Product # MA1-154) at a dilution of 1:1000 overnight at 4°C on a rocking platform, washed in TBST, and probed with an HRP-conjugated goat anti-mouse IgG secondary antibody (Product # 31430) at a dilution of 1:20,000 for 1 hour. Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34076).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
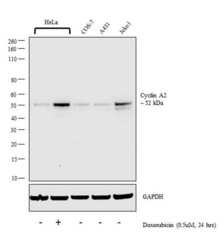
- Experimental details
- Western blot analysis was performed on whole cell extract (30 µg lysate) of HeLa (Lane 1), HeLa treated with doxorubicin (0.5uM, 24 hrs) (Lane 2), COS-7 (Lane 3), A431 (Lane 4) and Jeko1(Lane 5). The blot was probed with Anti-Cyclin A2 Monoclonal Antibody (E23.1) (Product # MA1-154, 1:1000 dilution) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A28177, 0.25 µg/ml, 1:4000 dilution). A 52 kDa band corresponding to Cyclin A2 was observed in all cell the lines tested and was increased upon Doxorubicin treatment.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
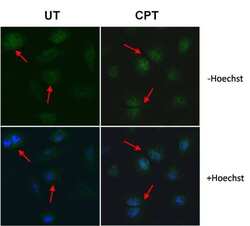
- Experimental details
- Immunofluorescent analysis of Cyclin A (green) in HeLa cells left untreated (UT, left panels) or treated with 100nM camptothecin (CPT, right panels). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS and blocked with 1% Blocker BSA (Product # 37525) for 15 minutes at room temperature. Cells were probed with a Cyclin A monoclonal antibody (Product # MA1-154) at a dilution of 1:50 for at least 1 hour at room temperature, washed with PBS, and incubated with a DyLight 488-conjugated goat anti-mouse IgG secondary antibody (Product # 35502). Nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249; lower panels) for 30 minutes. Images were taken on a Thermo Scientific ToxInsight Instrument at 20X magnification. Note: Red arrows indicate nuclear translocation of Cyclin A in response to treatment.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Cyclin A2 was performed using 70% confluent log phase HeLa cells treated with Camptothecin (100nM, 16 hrs). The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Cyclin A2 Monoclonal Antibody (Product # MA1-154) at 1:50 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing accumulation of Cyclin A2 in the nucleus upon treatment. Panel e represents the control cells showing cytoplasmic staining. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
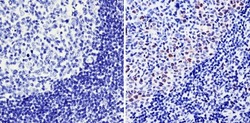
- Experimental details
- Immunohistochemistry analysis of Cyclin A showing staining in the nucleus of paraffin-embedded human tonsil tissue (right) compared with a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Cyclin A monoclonal antibody (Product # MA1-154) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
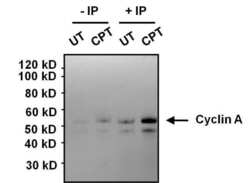
- Experimental details
- Immunoprecipitation analysis of Cyclin A was performed on HeLa cell lysates, from cells left untreated (UT) or treated with 100 nM camptothecin (CPT). Antigen-antibody complexes were formed by incubating 375 µg of lysate with 1 µg of a Cyclin A monoclonal antibody (Product # MA1-154) overnight on an end-over-end rotator at 4øC. The immune complexes were captured on 100 µL Protein A/G Plus Agarose (Product # 20423), washed extensively, and eluted with 5X Lane Marker Reducing Sample Buffer (Product # 39000). Samples, including control input cell lysates (-IP, left 2 lanes), were resolved on a 4-12% Bis-Tris polyacrylamide gel, transferred to a nitrocellulose membrane and blocked with 5% BSA/TBST for 1 hour. The membrane was probed with a Cyclin A monoclonal antibody (Product # MA1-154) at a dilution of 1:1000 overnight rotating at 4øC, washed in TBST, and probed with Clean-Blot IP Detection Reagent (Product # 21230). Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34076).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
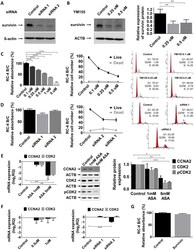
- Experimental details
- Figure 3 Effect of survivin inhibition (A) Survivin silencing by siRNA on RC-4 B/C cells. (B) Survivin inhibition by YM155, a small molecule inhibitor in RC-4 B/C cells. The effect of YM155 (C) and survivin siRNA (D) on pituitary adenoma cell viability, proliferation and cell cycle phases of RC-4 B/C cells (see details in the text). (E) Decrease of CCNA2, CDK2 mRNA and CCNA2, total CDK2 and p-CDK2 protein abundance following ASA treatment in RC-4 B/C cells. Left: mRNA, middle and right : western blot and densitometry. (F) YM155 and survivin siRNA transfection did not significantly alter CCNA2 and CDK2 expression in RC-4 B/C cells. (G) Survivin overexpression did not reduce the viability of RC-4 B/C cells. * : p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
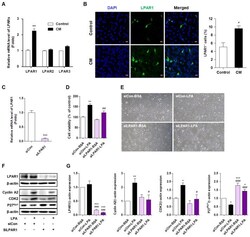
- Experimental details
- Figure 5 LPAR1 mediates the lysophosphatidic acid (LPA)-induced HaCaT cell proliferation. ( A ) HaCaT cells were seeded in six-well plates and treated with cytokine mixture (CM; 20 ng/mL tumor necrosis factor (TNF)-alpha and 10 ng/mL interleukin (IL)-6) for 4 h. LPAR1, LPAR2, and LPAR3 mRNA expression was analyzed by qRT-PCR. ( B ) HaCaT cells were seeded in four-well chambers and treated with CM for 24 h. Immunocytochemical analysis was performed using an anti-LPAR1 antibody and an Alexa Fluor 488-conjugated secondary antibody. The nuclei were counterstained with DAPI (blue). Representative staining images (left panel) and percentage of LPAR1 positive cells were quantified using confocal microscope (original magnification, 400x; the scale bars represent 20 mum). ( C - E ) HaCaT cells were transfected with a control siRNA (siCon) or LPAR1 siRNA (siLPAR1) for 6 h and treated with LPA for 24 h. ( C ) The mRNA expression of LPAR1 was analyzed by qRT-PCR. ( D ) Cell viability was analyzed by CCK8 assay and calculated as a percentage relative to that of siCon-BSA cells. ( E ) The cell morphology was observed by light microscopy. Original magnification, 100x. ( F , G ) HaCaT cells were transfected with siCon or siLPAR1 for 6 h and treated with LPA for 6 h. The levels of the proteins LPAR1, cyclin A2, CDK2, and p27 Kip1 were analyzed by Western blotting, quantified using ImageJ software, and normalized to those of beta-actin. (F) Representative images of the blots and ( G ) relative
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunoprecipitation
Immunoprecipitation