Antibody data
- Antibody Data
- Antigen structure
- References [23]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [6]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-824 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- PPAR gamma-2 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- PA1-824 detects peroxisome proliferator activated receptor (PPAR) gamma 2 from human and mouse tissues. This antibody does not detect PPAR alpha or PPAR delta. PA1-824 has been successfully used in Western blot and immunofluorescence procedures. By Western blot, this antibody detects an ~56 kDa protein representing PPAR gamma 2 from NIH-3T3 cell lysate. PA1-824 inhibits PPAR gamma 2 DNA binding. PA1-824 immunogen is a synthetic peptide corresponding to residues M(1) G E T L G D S P I D P E S D S(16) C of human PPAR gamma-2. This sequence is not conserved in PPAR gamma-1. PA1-824 can be used with blocking peptide PEP-027.
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Concentration
- 1 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references Dimensionality changes actin network through lamin A/C and zyxin.
A truncated PPAR gamma 2 localizes to mitochondria and regulates mitochondrial respiration in brown adipocytes.
Soya protein β-conglycinin ameliorates fatty liver and obesity in diet-induced obese mice through the down-regulation of PPARγ.
Peroxisome Proliferator-Activated Receptor α Facilitates Osteogenic Differentiation in MC3T3-E1 Cells via the Sirtuin 1-Dependent Signaling Pathway.
Databases for technical aspects of immunohistochemistry.
Sirtuin1 promotes osteogenic differentiation through downregulation of peroxisome proliferator-activated receptor γ in MC3T3-E1 cells.
Effect of retinoic acid and vitamin D3 on osteoblast differentiation and activity in aging.
Lipid-Laden Multilocular Cells in the Aging Thymus Are Phenotypically Heterogeneous.
Dietary β-conglycinin prevents fatty liver induced by a high-fat diet by a decrease in peroxisome proliferator-activated receptor γ2 protein.
An increase in liver PPARγ2 is an initial event to induce fatty liver in response to a diet high in butter: PPARγ2 knockdown improves fatty liver induced by high-saturated fat.
Signaling and biological effects of glucagon-like peptide 1 on the differentiation of mesenchymal stem cells from human bone marrow.
Fat-storing multilocular cells expressing CCR5 increase in the thymus with advancing age: potential role for CCR5 ligands on the differentiation and migration of preadipocytes.
Mechanical loading down-regulates peroxisome proliferator-activated receptor gamma in bone marrow stromal cells and favors osteoblastogenesis at the expense of adipogenesis.
Perivascular cells expressing annexin A5 define a novel mesenchymal stem cell-like population with the capacity to differentiate into multiple mesenchymal lineages.
A human peripheral blood monocyte-derived subset acts as pluripotent stem cells.
ADAM 12 protease induces adipogenesis in transgenic mice.
Overexpression and ribozyme-mediated targeting of transcriptional coactivators CREB-binding protein and p300 revealed their indispensable roles in adipocyte differentiation through the regulation of peroxisome proliferator-activated receptor gamma.
Platelet-derived growth factor promotes the expression of peroxisome proliferator-activated receptor gamma in vascular smooth muscle cells by a phosphatidylinositol 3-kinase/Akt signaling pathway.
Platelet-derived growth factor promotes the expression of peroxisome proliferator-activated receptor gamma in vascular smooth muscle cells by a phosphatidylinositol 3-kinase/Akt signaling pathway.
Diverse signaling pathways modulate nuclear receptor recruitment of N-CoR and SMRT complexes.
Diverse signaling pathways modulate nuclear receptor recruitment of N-CoR and SMRT complexes.
MCF-7 and T47D human breast cancer cells contain a functional peroxisomal response.
MCF-7 and T47D human breast cancer cells contain a functional peroxisomal response.
Zonderland J, Moldero IL, Anand S, Mota C, Moroni L
Biomaterials 2020 May;240:119854
Biomaterials 2020 May;240:119854
A truncated PPAR gamma 2 localizes to mitochondria and regulates mitochondrial respiration in brown adipocytes.
Chang JS, Ha K
PloS one 2018;13(3):e0195007
PloS one 2018;13(3):e0195007
Soya protein β-conglycinin ameliorates fatty liver and obesity in diet-induced obese mice through the down-regulation of PPARγ.
Li D, Ikaga R, Yamazaki T
The British journal of nutrition 2018 Jun;119(11):1220-1232
The British journal of nutrition 2018 Jun;119(11):1220-1232
Peroxisome Proliferator-Activated Receptor α Facilitates Osteogenic Differentiation in MC3T3-E1 Cells via the Sirtuin 1-Dependent Signaling Pathway.
Gong K, Qu B, Wang C, Zhou J, Liao D, Zheng W, Pan X
Molecules and cells 2017 Jun 30;40(6):393-400
Molecules and cells 2017 Jun 30;40(6):393-400
Databases for technical aspects of immunohistochemistry.
Furukawa S, Nagaike M, Ozaki K
Journal of toxicologic pathology 2017 Jan;30(1):79-107
Journal of toxicologic pathology 2017 Jan;30(1):79-107
Sirtuin1 promotes osteogenic differentiation through downregulation of peroxisome proliferator-activated receptor γ in MC3T3-E1 cells.
Qu B, Ma Y, Yan M, Gong K, Liang F, Deng S, Jiang K, Ma Z, Pan X
Biochemical and biophysical research communications 2016 Sep 9;478(1):439-445
Biochemical and biophysical research communications 2016 Sep 9;478(1):439-445
Effect of retinoic acid and vitamin D3 on osteoblast differentiation and activity in aging.
Bosetti M, Sabbatini M, Calarco A, Borrone A, Peluso G, Cannas M
Journal of bone and mineral metabolism 2016 Jan;34(1):65-78
Journal of bone and mineral metabolism 2016 Jan;34(1):65-78
Lipid-Laden Multilocular Cells in the Aging Thymus Are Phenotypically Heterogeneous.
Langhi LG, Andrade LR, Shimabukuro MK, van Ewijk W, Taub DD, Borojevic R, de Mello Coelho V
PloS one 2015;10(10):e0141516
PloS one 2015;10(10):e0141516
Dietary β-conglycinin prevents fatty liver induced by a high-fat diet by a decrease in peroxisome proliferator-activated receptor γ2 protein.
Yamazaki T, Kishimoto K, Miura S, Ezaki O
The Journal of nutritional biochemistry 2012 Feb;23(2):123-32
The Journal of nutritional biochemistry 2012 Feb;23(2):123-32
An increase in liver PPARγ2 is an initial event to induce fatty liver in response to a diet high in butter: PPARγ2 knockdown improves fatty liver induced by high-saturated fat.
Yamazaki T, Shiraishi S, Kishimoto K, Miura S, Ezaki O
The Journal of nutritional biochemistry 2011 Jun;22(6):543-53
The Journal of nutritional biochemistry 2011 Jun;22(6):543-53
Signaling and biological effects of glucagon-like peptide 1 on the differentiation of mesenchymal stem cells from human bone marrow.
Sanz C, Vázquez P, Blázquez C, Barrio PA, Alvarez Mdel M, Blázquez E
American journal of physiology. Endocrinology and metabolism 2010 Mar;298(3):E634-43
American journal of physiology. Endocrinology and metabolism 2010 Mar;298(3):E634-43
Fat-storing multilocular cells expressing CCR5 increase in the thymus with advancing age: potential role for CCR5 ligands on the differentiation and migration of preadipocytes.
Mello Coelho Vd, Bunbury A, Rangel LB, Giri B, Weeraratna A, Morin PJ, Bernier M, Taub DD
International journal of medical sciences 2009 Dec 4;7(1):1-14
International journal of medical sciences 2009 Dec 4;7(1):1-14
Mechanical loading down-regulates peroxisome proliferator-activated receptor gamma in bone marrow stromal cells and favors osteoblastogenesis at the expense of adipogenesis.
David V, Martin A, Lafage-Proust MH, Malaval L, Peyroche S, Jones DB, Vico L, Guignandon A
Endocrinology 2007 May;148(5):2553-62
Endocrinology 2007 May;148(5):2553-62
Perivascular cells expressing annexin A5 define a novel mesenchymal stem cell-like population with the capacity to differentiate into multiple mesenchymal lineages.
Brachvogel B, Moch H, Pausch F, Schlötzer-Schrehardt U, Hofmann C, Hallmann R, von der Mark K, Winkler T, Pöschl E
Development (Cambridge, England) 2005 Jun;132(11):2657-68
Development (Cambridge, England) 2005 Jun;132(11):2657-68
A human peripheral blood monocyte-derived subset acts as pluripotent stem cells.
Zhao Y, Glesne D, Huberman E
Proceedings of the National Academy of Sciences of the United States of America 2003 Mar 4;100(5):2426-31
Proceedings of the National Academy of Sciences of the United States of America 2003 Mar 4;100(5):2426-31
ADAM 12 protease induces adipogenesis in transgenic mice.
Kawaguchi N, Xu X, Tajima R, Kronqvist P, Sundberg C, Loechel F, Albrechtsen R, Wewer UM
The American journal of pathology 2002 May;160(5):1895-903
The American journal of pathology 2002 May;160(5):1895-903
Overexpression and ribozyme-mediated targeting of transcriptional coactivators CREB-binding protein and p300 revealed their indispensable roles in adipocyte differentiation through the regulation of peroxisome proliferator-activated receptor gamma.
Takahashi N, Kawada T, Yamamoto T, Goto T, Taimatsu A, Aoki N, Kawasaki H, Taira K, Yokoyama KK, Kamei Y, Fushiki T
The Journal of biological chemistry 2002 May 10;277(19):16906-12
The Journal of biological chemistry 2002 May 10;277(19):16906-12
Platelet-derived growth factor promotes the expression of peroxisome proliferator-activated receptor gamma in vascular smooth muscle cells by a phosphatidylinositol 3-kinase/Akt signaling pathway.
Fu M, Zhu X, Wang Q, Zhang J, Song Q, Zheng H, Ogawa W, Du J, Chen YE
Circulation research 2001 Nov 23;89(11):1058-64
Circulation research 2001 Nov 23;89(11):1058-64
Platelet-derived growth factor promotes the expression of peroxisome proliferator-activated receptor gamma in vascular smooth muscle cells by a phosphatidylinositol 3-kinase/Akt signaling pathway.
Fu M, Zhu X, Wang Q, Zhang J, Song Q, Zheng H, Ogawa W, Du J, Chen YE
Circulation research 2001 Nov 23;89(11):1058-64
Circulation research 2001 Nov 23;89(11):1058-64
Diverse signaling pathways modulate nuclear receptor recruitment of N-CoR and SMRT complexes.
Lavinsky RM, Jepsen K, Heinzel T, Torchia J, Mullen TM, Schiff R, Del-Rio AL, Ricote M, Ngo S, Gemsch J, Hilsenbeck SG, Osborne CK, Glass CK, Rosenfeld MG, Rose DW
Proceedings of the National Academy of Sciences of the United States of America 1998 Mar 17;95(6):2920-5
Proceedings of the National Academy of Sciences of the United States of America 1998 Mar 17;95(6):2920-5
Diverse signaling pathways modulate nuclear receptor recruitment of N-CoR and SMRT complexes.
Lavinsky RM, Jepsen K, Heinzel T, Torchia J, Mullen TM, Schiff R, Del-Rio AL, Ricote M, Ngo S, Gemsch J, Hilsenbeck SG, Osborne CK, Glass CK, Rosenfeld MG, Rose DW
Proceedings of the National Academy of Sciences of the United States of America 1998 Mar 17;95(6):2920-5
Proceedings of the National Academy of Sciences of the United States of America 1998 Mar 17;95(6):2920-5
MCF-7 and T47D human breast cancer cells contain a functional peroxisomal response.
Kilgore MW, Tate PL, Rai S, Sengoku E, Price TM
Molecular and cellular endocrinology 1997 May 16;129(2):229-35
Molecular and cellular endocrinology 1997 May 16;129(2):229-35
MCF-7 and T47D human breast cancer cells contain a functional peroxisomal response.
Kilgore MW, Tate PL, Rai S, Sengoku E, Price TM
Molecular and cellular endocrinology 1997 May 16;129(2):229-35
Molecular and cellular endocrinology 1997 May 16;129(2):229-35
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of PPAR gamma-2 was performed by loading 25 ug of NIH-3T3 (Lane 1), 3T3-L1 (Lane 2), and Hela cell lysates (Lane 3) and a molecular weight protein ladder onto an SDS polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked with a blocking buffer at 4_C overnight. The membrane was probed with a PPAR gamma-2 polyclonal antibody (Product # PA1-824) at a dilution of 1:1000 overnight at 4ÁC, washed in TBST, and probed with an HRP-conjugated secondary antibody for 1 hr at room temperature in the dark. Chemiluminescent detection was performed using Pierce ECL Plus Western Blotting Substrate (Product # 32132). Results show a band at 56 kDa in all three cell lines.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of PPAR gamma-2 (green) showing positive staining in the nucleus and cytoplasm of 3T3-L1 cells (right) compared with a negative control in the absence of primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes, blocked with 3% BSA-PBS for 30 minutes at room temperature and probed with a PPAR gamma-2 polyclonal antibody (Product # PA1-824) in 3% BSA-PBS at a dilution of 1:200 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight 488-conjugated goat-anti-rabbit IgG secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with DAPI for 5-10 minutes in the dark. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of PPAR gamma-2 (green) showing positive staining in the nucleus and cytoplasm of Hela cells (right) compared with a negative control in the absence of primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes, blocked with 3% BSA-PBS for 30 minutes at room temperature and probed with a PPAR gamma-2 polyclonal antibody (Product # PA1-824) in 3% BSA-PBS at a dilution of 1:200 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight 488-conjugated goat-anti-rabbit IgG secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with DAPI for 5-10 minutes in the dark. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of PPAR gamma-2 (green) showing positive staining in the nucleus and cytoplasm of NIH-3T3 cells (right) compared with a negative control in the absence of primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes, blocked with 3% BSA-PBS for 30 minutes at room temperature and probed with a PPAR gamma-2 polyclonal antibody (Product # PA1-824) in 3% BSA-PBS at a dilution of 1:200 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight 488-conjugated goat-anti-rabbit IgG secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with DAPI for 5-10 minutes in the dark. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of PPAR gamma (red) in 3T3-L1 differentiated day 7 cells. The cells were fixed with 4% paraformaldehyde in PBS for 15 minutes at room temperature, permeabilized with 0.1% Triton X-100 for 15 minutes, and blocked with 3% BSA for 30 minutes at room temperature. Cells were stained with a PPAR gamma rabbit polyclonal antibody (Product # PA1-824) at a dilution of 1:200 in blocking buffer for 1 hour at room temperature, and then incubated with a Goat anti-Rabbit IgG (H+L) Secondary Antibody, Dylight 680 (Product # 35568) at a dilution of 1:1000 for at least 30 minutes at room temperature in the dark (red). Lipids (green) were stained with HCS LipidTOX neutral lipid stain (Product # H34475) at a dilution of 1:200 for at least 30 minutes at room temperature in the dark. Nuclei (blue) were stained with Hoechst 33342 (Product # 62249). Images were taken on a EVOS FL Auto Imaging System at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
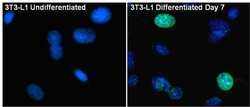
- Experimental details
- Immunofluorescent analysis of PPAR gamma (green) in 3T3-L1 Undifferentiated (left panel) and Differentiated day 7 cells (right panel. The cells were fixed with 4% paraformaldehyde in PBS for 15 minutes at room temperature, permeabilized with 0.1% Triton X-100 for 15 minutes, and blocked with 3% BSA for 30 minutes at room temperature. Cells were stained with a PPAR gamma rabbit polyclonal antibody (Product # PA1-824) at a dilution of 1:200 in blocking buffer for 1 hour at room temperature, and then incubated with a Goat anti-Rabbit IgG (H+L) Superclonal Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:1000 for at least 30 minutes at room temperature in the dark. Nuclei (blue) were stained with Hoechst 33342 (Product # 62249). Images were taken on a EVOS FL Auto Imaging System at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of PPAR gamma (green) in 3T3-L1 Undifferentiated (left panel) and Differentiated day 7 cells (right panel. The cells were fixed with 4% paraformaldehyde in PBS for 15 minutes at room temperature, permeabilized with 0.1% Triton X-100 for 15 minutes, and blocked with 3% BSA for 30 minutes at room temperature. Cells were stained with a PPAR gamma rabbit polyclonal antibody (Product # PA1-824) at a dilution of 1:200 in blocking buffer for 1 hour at room temperature, and then incubated with a Goat anti-Rabbit IgG (H+L) Superclonal Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:1000 for at least 30 minutes at room temperature in the dark. Nuclei (blue) were stained with Hoechst 33342 (Product # 62249). Images were taken on a EVOS FL Auto Imaging System at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 6 Heterogeneity of intrathymic LLMC phenotypes. A : Left panel: Preadipocytes immunostained for PPARgamma2 in the thymic parenchyma close to the capsule and blood vessels. Small panels show representative photomicrographs of intrathymic lipid-laden preadipocytes immunolabeled for PPARgamma2 (green) or PLIN (red); merged images in the right panel. B: Left panel shows thymic brown adipocytes expressing UCP-1 near blood vessels in the parenchyma. Small panels show representative photomicrographs of intrathymic brown adipocytes immunolabeled for UCP1 (green) or PLIN (red); merged images in the right panel C: intrathymic macrophagic LLMC expressing PLIN (red) and Iba 1 (green). D: Pericytic LLMC (white arrowheads) expressing PLIN (red) and NG2 (green) adjacent to the capsule. Insets show higher magnification of perilipin and NG2 staining around the lipid droplets of the pericytic LLMC. E : myofibroblastic LLMC stained with antibody for PLIN (red) and alphaSMA (green) surrounding a blood vessel. F: Thymic epithelial cells positively stained with antibody anti-Pan cytokeratin (CK, green) did not express PLIN (red). Nuclei stained with DAPI or Hoechst dye (blue). BV: blood vessels, Ca: capsule, S: septae. Scale bars in A, left panels: 15mum; right panel: 7 mum in B, left panels: 25mum; right panel: 5mum; in C: 10mum; in D: 20mum, inset: 10mum; in E: 15mum; in F: 30mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 3 The truncated PPARgamma2 localizes in the mitochondrial matrix. (A) The truncated form of PPARgamma2 in mitochondria is protected from proteinase K digestion. Purified brown adipose tissue mitochondria (60 mug) were treated with increasing amounts of proteinase K in the absence or presence of 1% Triton X-100. (B) Immuno-transmission electron microscopic analysis of the truncated PPARgamma2 in brown adipocytes. Black dots indicated by arrow heads represent immunogold particles reacted with PPARgamma (H100) antibody. Mitochondrial localization of immunogold particles was examined in 4-5 grids per group (20-30 mitochondria/grid), and the relative number of immunogold particles localized in the mitochondria was shown in the right panel. Data are presented as the mean +- SEM. Data represent mean +-SEM. ****P
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot