Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [3]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-337 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- SREBP1 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- PA1-337 detects SREBP 1 in mouse and rat samples.
- Concentration
- 1 mg/mL
Submitted references Keratin 17 Is Required for Lipid Metabolism in Keratinocytes and Benefits Epidermal Permeability Barrier Homeostasis.
Gycyrrhizic acid alleviates atherosclerotic lesions in rats with diabetes mellitus.
Polygonatum sibiricum F. Delaroche polysaccharide ameliorates HFD‑induced mouse obesity via regulation of lipid metabolism and inflammatory response.
Long term N-acetylcysteine administration rescues liver steatosis via endoplasmic reticulum stress with unfolded protein response in mice.
Sterol regulatory element binding protein and dietary lipid regulation of fatty acid synthesis in the mammary epithelium.
Pang B, Zhu Z, Xiao C, Luo Y, Fang H, Bai Y, Sun Z, Ma J, Dang E, Wang G
Frontiers in cell and developmental biology 2021;9:779257
Frontiers in cell and developmental biology 2021;9:779257
Gycyrrhizic acid alleviates atherosclerotic lesions in rats with diabetes mellitus.
Zhao Y, Li W, Zhang D
Molecular medicine reports 2021 Nov;24(5)
Molecular medicine reports 2021 Nov;24(5)
Polygonatum sibiricum F. Delaroche polysaccharide ameliorates HFD‑induced mouse obesity via regulation of lipid metabolism and inflammatory response.
Liu B, Tang Y, Song Z, Ge J
Molecular medicine reports 2021 Jul;24(1)
Molecular medicine reports 2021 Jul;24(1)
Long term N-acetylcysteine administration rescues liver steatosis via endoplasmic reticulum stress with unfolded protein response in mice.
Tsai CC, Chen YJ, Yu HR, Huang LT, Tain YL, Lin IC, Sheen JM, Wang PW, Tiao MM
Lipids in health and disease 2020 May 25;19(1):105
Lipids in health and disease 2020 May 25;19(1):105
Sterol regulatory element binding protein and dietary lipid regulation of fatty acid synthesis in the mammary epithelium.
Rudolph MC, Monks J, Burns V, Phistry M, Marians R, Foote MR, Bauman DE, Anderson SM, Neville MC
American journal of physiology. Endocrinology and metabolism 2010 Dec;299(6):E918-27
American journal of physiology. Endocrinology and metabolism 2010 Dec;299(6):E918-27
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
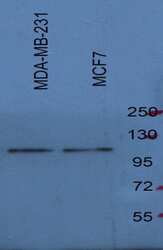
- Experimental details
- Western blot analysis of SREBP1 was performed by loading 20 µg of MCF-7 and MDA-MB-231 ell lysates in RIPA buffer (Product # 89901) and Page Ruler Plus Protein Ladder (Product # 26620) onto a 4-8% sodium dodecyl sulfate polyacrylamide gel (SDS-PAGE). Proteins were transferred to nitrocellulose membrane and blocked in 5% fat-free milk /PBST for one hour at room temperature. SREBP1 was detected at approximately 120 kDa using a SREBP1 polyclonal antibody (Product # PA1-337) at a dilution of 1:1000 5% fat-free milk /PBST overnight at 4°C on a rocking platform, followed by a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5,000-10000 for one hour. Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34078). Data courtesy of Dr Wei Xu's lab at University of Wisconsin Madison.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
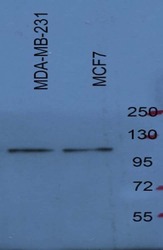
- Experimental details
- Western blot analysis of SREBP1 was performed by loading 20 µg of MCF-7 and MDA-MB-231 ell lysates in RIPA buffer (Product # 89901) and Page Ruler Plus Protein Ladder (Product # 26620) onto a 4-8% sodium dodecyl sulfate polyacrylamide gel (SDS-PAGE). Proteins were transferred to nitrocellulose membrane and blocked in 5% fat-free milk /PBST for one hour at room temperature. SREBP1 was detected at approximately 120 kDa using a SREBP1 polyclonal antibody (Product # PA1-337) at a dilution of 1:1000 5% fat-free milk /PBST overnight at 4°C on a rocking platform, followed by a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5,000-10000 for one hour. Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34078). Data courtesy of Dr Wei Xu's lab at University of Wisconsin Madison.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
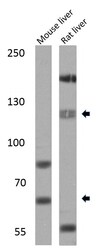
- Experimental details
- Western blot analysis of SREBP1 was performed by loading 25 µg of mouse liver (lane 1) or rat liver (lane 2) lysates onto an SDS polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked at 4ºC overnight. The membrane was probed with a SREBP1 polyclonal antibody (Product # PA1-337) at a dilution of 1:1000 overnight at 4°C, washed in TBST, and probed with an HRP-conjugated secondary antibody for 1 hr at room temperature in the dark. Chemiluminescent detection was performed using Pierce ECL Plus Western Blotting Substrate (Product # 32132). Results show a band at ~68 and 120 kDa.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of SREBP1 (green) showing staining in the cytoplasm of C2C12 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a SREBP1 polyclonal antibody (Product # PA1-337) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
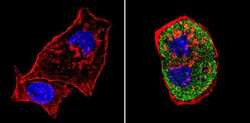
- Experimental details
- Immunofluorescent analysis of SREBP1 (green) showing staining in the cytoplasm of HepG2 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a SREBP1 polyclonal antibody (Product # PA1-337) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
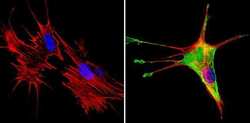
- Experimental details
- Immunofluorescent analysis of SREBP1 (green) showing staining in the cytoplasm of NIH-3T3 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a SREBP1 polyclonal antibody (Product # PA1-337) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
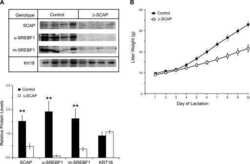
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
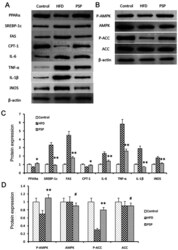
- Experimental details
- Figure 5. Signal pathway changes and lipid-associated genes and inflammatory cytokine protein changes. (A) Western blotting was performed to reveal the protein expressions of the lipid and inflammatory genes as well as the key genes of the AMPK signal pathway. Lipid synthesis-associated genes and inflammatory genes were inhibited by PSP. (B) P-AMPK and the downstream gene p-ACC was clearly activated by PSP, which demonstrated that the PSP could ameliorate HFD-induced mouse obesity through activating the AMPK signal pathway. (C) Semi-quantification of protein expression levels from part (A) (D) Semi-quantification of protein expression levels from part (B) # P>0.05, *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. Effects of GA on macrophage activation, alpha-SMA expression, FAS and SREBP-1c expression in DM-AS rats. (A) Immunohistochemistry was used to detect CD68 and alpha-SMA expression in aortic tissues (magnification, x200). (B) Quantitative analysis of CD68 and alpha-SMA expression from (A). (C) Western blotting detected the expression of FAS and SREBP-1c. ***P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 5 K17 alters the subcellular localization of SREBP-1 and PPARgamma. (A,B) The protein levels of full-length SPEBP-1 (flSREBP-1) and nuclear SPEBP-1 (nSREBP-1) in HaCaT cells were measured after transfection with K17 siRNA or pEGFP-N1-K17. (C,D) Immunofluorescence analysis of FASN and PPARgamma subcellular localization in HaCaT cells after transfection with K17 siRNA or pEGFP-N1-K17. Scale bars = 10 mum. (E) HaCaT cells were treated with pCMV6-XL5-K17 (K17 LV) and a nuclear fraction was prepared. SREBP-1 and PPARgamma protein was quantitated by Western blot. Lamin A/C and beta-actin were used as loading controls for nuclear and cytosol fractions, respectively. (F) Cells were co-immunoprecipitated with anti-K17 antibody and SREBP-1 and PPARgamma proteins were detected by Western blot. Data are expressed as means +- standard error of the mean from three independent experiments. * p < 0.05.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot