Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 16-7052-38 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-5 Monoclonal Antibody (TRFK5), Functional Grade, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The TRFK5 antibody reacts with mouse and human interleukin-5 (IL-5). The TRFK5 antibody is a neutralizing antibody. Mouse IL-5 is a disulfide-linked homodimer, containing two 113 amino acid peptides; these resolve as 32-34 kDa bands in SDS-PAGE. IL-5 is produced by T cells and has been known as eosinophil-differentiating factor (EDF), B cell growth factor II (BCGFII), and T cell-replacing factor (TRF). IL-5 induces eosinophil differentiation and promotes eosinophil survival and activation. In mice, IL-5 has been shown to stimulate B cell proliferation and antibody production. Applications Reported:The TRFK5 antibody has been reported for use in ELISA capture, ELISPOT capture, neutralization of IL-5 bioactivity, and IHC. Applications Tested: The Functional Grade Purified TRFK5 antibody has been tested by LAL assay to verify low endotoxin levels, by ELISA, and in bioassay for neutralization of mouse IL-5 bioactivity. The TRFK5 antibody at 0.06 µg/mL has been found to inhibit by 50% the biological effects of 1.0 ng/mL mouse IL-5 in an MC/9 cell proliferation assay. Detailed information and protocols about cytokine bioassays and in vitro cytokine neutralization using antibodies can be found in the BestProtocols® section. The TRFK5 antibody has been tested as the capture antibody in a sandwich ELISA for analysis of mouse IL-5 in combination with the biotin TRFK4 (13-7051) antibody for detection and recombinant mouse IL-5 (14-8051) as the standard. A suitable range of concentrations of this antibody for ELISA capture is 1-4 µg/mL. A standard curve consisting of doubling dilutions of the recombinant standard over the range of 1000 pg/mL - 8 pg/mL should be included in each ELISA plate. The TRFK5 antibody has been tested as the capture antibody in a sandwich ELISA for analysis of human IL-5 in combination with the biotinylated JES1-5A10 (13-7059) antibody for detection and recombinant human IL-5 (14-80591) as the standard. A suitable range of concentrations of this antibody for ELISA capture is 1-4 µg/mL. A standard curve consisting of doubling dilutions of the recombinant standard over the range of 1000 pg/mL - 8 pg/mL should be included in each ELISA plate. Storage and handling: Use in a sterile environment. Filtration: 0.2 µm post-manufacturing filtered. Purity: Greater than 90%, as determined by SDS-PAGE. Endotoxin Level:Less than 0.001 ng/µg antibody, as determined by LAL assay. Aggregation:Less than 10%, as determined by HPLC.
- Reactivity
- Human, Mouse
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- TRFK5
- Vial size
- 5 mg
- Concentration
- 1 mg/mL
- Storage
- 4°C
Submitted references Il4ra-independent vaginal eosinophil accumulation following helminth infection exacerbates epithelial ulcerative pathology of HSV-2 infection.
Bcl11b is essential for licensing Th2 differentiation during helminth infection and allergic asthma.
Fat-associated lymphoid clusters control local IgM secretion during pleural infection and lung inflammation.
Rapid, multiparameter profiling of cellular secretion using silicon photonic microring resonator arrays.
Loss of transgene following ex vivo gene transfer is associated with a dominant Th2 response: implications for cutaneous gene therapy.
Chetty A, Darby MG, Vornewald PM, Martín-Alonso M, Filz A, Ritter M, McSorley HJ, Masson L, Smith K, Brombacher F, O'Shea MK, Cunningham AF, Ryffel B, Oudhoff MJ, Dewals BG, Layland LE, Horsnell WGC
Cell host & microbe 2021 Apr 14;29(4):579-593.e5
Cell host & microbe 2021 Apr 14;29(4):579-593.e5
Bcl11b is essential for licensing Th2 differentiation during helminth infection and allergic asthma.
Lorentsen KJ, Cho JJ, Luo X, Zuniga AN, Urban JF Jr, Zhou L, Gharaibeh R, Jobin C, Kladde MP, Avram D
Nature communications 2018 Apr 26;9(1):1679
Nature communications 2018 Apr 26;9(1):1679
Fat-associated lymphoid clusters control local IgM secretion during pleural infection and lung inflammation.
Jackson-Jones LH, Duncan SM, Magalhaes MS, Campbell SM, Maizels RM, McSorley HJ, Allen JE, Bénézech C
Nature communications 2016 Sep 1;7:12651
Nature communications 2016 Sep 1;7:12651
Rapid, multiparameter profiling of cellular secretion using silicon photonic microring resonator arrays.
Luchansky MS, Bailey RC
Journal of the American Chemical Society 2011 Dec 21;133(50):20500-6
Journal of the American Chemical Society 2011 Dec 21;133(50):20500-6
Loss of transgene following ex vivo gene transfer is associated with a dominant Th2 response: implications for cutaneous gene therapy.
Lu Z, Ghazizadeh S
Molecular therapy : the journal of the American Society of Gene Therapy 2007 May;15(5):954-61
Molecular therapy : the journal of the American Society of Gene Therapy 2007 May;15(5):954-61
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Naive mouse splenocytes (SJL/J strain) were stimulated with 2 µg/mL ConA in HL-1 media for 48 hours (right well) in a Mouse IL-5 ELISPOT assay plate. Left well is medium alone control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Frequencies of CD4 + T-cells expressing Gata3 and Th2 cytokines in asthma and helminth infection are reduced in Bcl11b fl/fl dLck-iCre mice by an intrinsic defect. a Flow cytometry analysis of IL-4 (left column), IL-5 (middle column), and IL-13 (right column) against GATA3 in the CD4 + T-cell population of the lung parenchyma of HDM asthma-induced mice. b Frequencies of IL-4 + ( n = 4), IL-5 + ( n = 4), and IL-13 + ( n = 8) (top panel) and GATA3 + ( n = 10) (bottom panel) CD4 + T-cells from the lungs of the indicated groups. Data are derived from two (IL-4, IL-5, and IL-13 data) or five (GATA3 data) independent experiments. c Flow cytometry analysis of IL-4 (left column), IL-5 (middle column), and IL-13 (right column) against GATA3 in the CD4 + T-cell population from the mesenteric (m)LNs of H. polygyrus infected mice. d Frequencies of IL-4 + ( n = 4), IL-5 + ( n = 4), and IL-13 + ( n = 4) (top panel) and GATA3 + ( n = 8) (bottom panel) CD4 + T-cells from mLNs of the indicated groups. Data are derived from two (IL-4, IL-5, and IL-13) or four (GATA3) independent experiments. e Flow cytometry analysis of GATA3 in CD4 + T-cells (left column) and IL-4 (middle and right columns) in the GATA3 hi (middle column) and GATA3 lo (right column) CD4 + T-cells from the mLNs of H. polygyrus bakeri infected mice. f The ratio of GATA3 hi /GATA3 lo CD4 + T-cells (top) and frequencies of IL-4 secreting cells within the GATA3 hi and GATA3 lo CD4 + T-cells (bottom) from the mLNs of H. poly
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Influence of N. brasiliensis exposure on uncolonized FGT, increase in FGT eosinophils following Nb exposure (A) Female mice were hormone-synchronized 7 days prior to Nb infection. (B) At day 9 post-Nb infection (Nb 9dpi), levels of IL-33, IL-4, and IL-5 in FGT homogenates or lavages were assessed by ELISA or Luminex.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Nb-exacerbated HSV-2 pathology and FGT eosinophil infiltration is Il4ra independent WT and Il4ra -/- mice were infected with HSV-2 following Nb exposure as previously described. (A) HSV-2 progression was determined by daily pathology scoring. (B) Viral shedding was measured by plaque assay of day 6 vaginal washes. (C) Genital levels of IL-5 and IFN-gamma at day 2 post-HSV-2 infection, determined by Luminex and ELISA, respectively. Dotted line represents LLOQ of Luminex analysis. At 6 dpi, vaginal tissue was analyzed by H&E staining. (D) Representative sections (n = 3-4), displaying ulceration and inflammation of vaginal tissue. Images were taken at x50 magnification. HSV-2-ulcerated vaginal epithelium is indicated by black dotted lines and qualified as percentage (%) of ulcerated epithelium. (E) Representative Sirius-red-stained sections (n = 3-4) of virus-induced (Ei) epithelial ulcers and (Eii) stromal inflammation. Black arrows indicate eosinophil presence in the vaginal epithelial layer. Images were taken at x400 and x1,000 magnification. (F) Numbers (x10 3 ) of FGT eosinophils in WT and Il4ra -/- co-infected mice compared with HSV-2-only controls. Data are representative of two independent experiments with 3-6 mice per group (mean +- SEM). Statistical significance was calculated by two-way ANOVA with Bonferroni correction for multiple comparisons. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
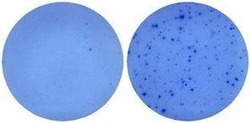
- Experimental details
- Naive mouse splenocytes (SJL/J strain) were stimulated with 2 µg/mL ConA in HL-1 media for 48 hours (right well) in a Mouse IL-5 ELISPOT assay plate. Left well is medium alone control.
 Explore
Explore Validate
Validate Learn
Learn Immunohistochemistry
Immunohistochemistry Blocking/Neutralizing
Blocking/Neutralizing Other assay
Other assay