MA1-935
antibody from Invitrogen Antibodies
Targeting: CFTR
ABC35, ABCC7, CF, CFTR/MRP, dJ760C5.1, MRP7, TNR-CFTR
 Western blot
Western blot Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [12]
- Comments [0]
- Validations
- Immunocytochemistry [3]
- Immunohistochemistry [3]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-935 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CFTR Monoclonal Antibody (CF3)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse
- Host
- Mouse
- Isotype
- IgM
- Antibody clone number
- CF3
- Vial size
- 100 µL
- Concentration
- Conc. Not Determined
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references Therapeutic CFTR Correction Normalizes Systemic and Lung-Specific S1P Level Alterations Associated with Heart Failure.
Heterogeneous expression of CFTR in insulin-secreting β-cells of the normal human islet.
Type IV(B) pili are required for invasion but not for adhesion of Salmonella enterica serovar Typhi into BHK epithelial cells in a cystic fibrosis transmembrane conductance regulator-independent manner.
Exaggerated apoptosis and NF-kappaB activation in pancreatic and tracheal cystic fibrosis cells.
Expression and localization of cystic fibrosis transmembrane conductance regulator in the rat endocrine pancreas.
Differential regulation of cystic fibrosis transmembrane conductance regulator by interferon gamma in mast cells and epithelial cells.
Antibodies for CFTR studies.
Polarization of specific tropomyosin isoforms in gastrointestinal epithelial cells and their impact on CFTR at the apical surface.
PTH stimulates a Cl(-)-dependent and EIPA-sensitive current in chick proximal tubule cells in culture.
Expression and functional characterization of CFTR in mast cells.
Salmonella typhi uses CFTR to enter intestinal epithelial cells.
Production and characterisation of monoclonal and polyclonal antibodies to different regions of the cystic fibrosis transmembrane conductance regulator (CFTR): detection of immunologically related proteins.
Uhl FE, Vanherle L, Matthes F, Meissner A
International journal of molecular sciences 2022 Jan 14;23(2)
International journal of molecular sciences 2022 Jan 14;23(2)
Heterogeneous expression of CFTR in insulin-secreting β-cells of the normal human islet.
Di Fulvio M, Bogdani M, Velasco M, McMillen TS, Ridaura C, Kelly L, Almutairi MM, Kursan S, Sajib AA, Hiriart M, Aguilar-Bryan L
PloS one 2020;15(12):e0242749
PloS one 2020;15(12):e0242749
Type IV(B) pili are required for invasion but not for adhesion of Salmonella enterica serovar Typhi into BHK epithelial cells in a cystic fibrosis transmembrane conductance regulator-independent manner.
Bravo D, Blondel CJ, Hoare A, Leyton L, Valvano MA, Contreras I
Microbial pathogenesis 2011 Nov;51(5):373-7
Microbial pathogenesis 2011 Nov;51(5):373-7
Exaggerated apoptosis and NF-kappaB activation in pancreatic and tracheal cystic fibrosis cells.
Rottner M, Kunzelmann C, Mergey M, Freyssinet JM, Martínez MC
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2007 Sep;21(11):2939-48
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2007 Sep;21(11):2939-48
Expression and localization of cystic fibrosis transmembrane conductance regulator in the rat endocrine pancreas.
Boom A, Lybaert P, Pollet JF, Jacobs P, Jijakli H, Golstein PE, Sener A, Malaisse WJ, Beauwens R
Endocrine 2007 Oct;32(2):197-205
Endocrine 2007 Oct;32(2):197-205
Differential regulation of cystic fibrosis transmembrane conductance regulator by interferon gamma in mast cells and epithelial cells.
Kulka M, Dery R, Nahirney D, Duszyk M, Befus AD
The Journal of pharmacology and experimental therapeutics 2005 Nov;315(2):563-70
The Journal of pharmacology and experimental therapeutics 2005 Nov;315(2):563-70
Antibodies for CFTR studies.
Mendes F, Farinha CM, Roxo-Rosa M, Fanen P, Edelman A, Dormer R, McPherson M, Davidson H, Puchelle E, De Jonge H, Heda GD, Gentzsch M, Lukacs G, Penque D, Amaral MD
Journal of cystic fibrosis : official journal of the European Cystic Fibrosis Society 2004 Aug;3 Suppl 2:69-72
Journal of cystic fibrosis : official journal of the European Cystic Fibrosis Society 2004 Aug;3 Suppl 2:69-72
Polarization of specific tropomyosin isoforms in gastrointestinal epithelial cells and their impact on CFTR at the apical surface.
Dalby-Payne JR, O'Loughlin EV, Gunning P
Molecular biology of the cell 2003 Nov;14(11):4365-75
Molecular biology of the cell 2003 Nov;14(11):4365-75
PTH stimulates a Cl(-)-dependent and EIPA-sensitive current in chick proximal tubule cells in culture.
Laverty G, McWilliams C, Sheldon A, Arnason SS
American journal of physiology. Renal physiology 2003 May;284(5):F987-95
American journal of physiology. Renal physiology 2003 May;284(5):F987-95
Expression and functional characterization of CFTR in mast cells.
Kulka M, Gilchrist M, Duszyk M, Befus AD
Journal of leukocyte biology 2002 Jan;71(1):54-64
Journal of leukocyte biology 2002 Jan;71(1):54-64
Salmonella typhi uses CFTR to enter intestinal epithelial cells.
Pier GB, Grout M, Zaidi T, Meluleni G, Mueschenborn SS, Banting G, Ratcliff R, Evans MJ, Colledge WH
Nature 1998 May 7;393(6680):79-82
Nature 1998 May 7;393(6680):79-82
Production and characterisation of monoclonal and polyclonal antibodies to different regions of the cystic fibrosis transmembrane conductance regulator (CFTR): detection of immunologically related proteins.
Walker J, Watson J, Holmes C, Edelman A, Banting G
Journal of cell science 1995 Jun;108 ( Pt 6):2433-44
Journal of cell science 1995 Jun;108 ( Pt 6):2433-44
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of CFTR using CFTR Monoclonal antibody (CF3) (Product # MA1-935) shows staining in HeLa cells. CFTR staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing CFTR (Product # MA1-935) at a dilution of 1:100-1:200 over night at 4 °C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody. Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
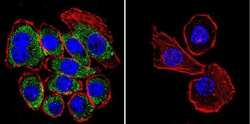
- Experimental details
- Immunofluorescent analysis of CFTR using CFTR Monoclonal antibody (CF3) (Product # MA1-935) shows staining in U251 glioma cells. CFTR staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing CFTR (Product # MA1-935) at a dilution of 1:100-1:200 over night at 4 °C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody. Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of CFTR using CFTR Monoclonal antibody (CF3) (Product # MA1-935) shows staining in WiDr colon carcinoma cells. CFTR staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing CFTR (Product # MA1-935) at a dilution of 1:100-1:200 over night at 4 °C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody. Images were taken at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry was performed on cancer biopsies of deparaffinized Human colon carcinoma tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing CFTR (Product # MA1-935) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Human pancreas tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing CFTR (Product # MA1-935) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Human tonsil tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing CFTR (Product # MA1-935) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
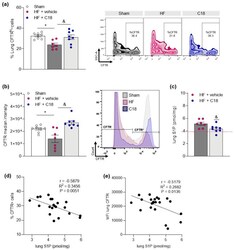
- Experimental details
- Figure 3 Correcting cystic fibrosis transmembrane regulator expression attenuates heart failure-associated pulmonary sphingosine-1-phosphate elevation. ( a ) Proportion of CFTR + cells in the lungs of sham and HF mice after two weeks of vehicle or C18 treatment determined by flow cytometry. Representative zebra plots showing proportion of CFTR + cells in lung tissue. ( b ) MFI quantification of CFTR + cells in lung tissue of sham and HF mice after two weeks of vehicle or C18 treatment determined by flow cytometry and representative histograms. ( c ) Pulmonary S1P tissue concentrations of vehicle and C18-treated HF mice. The pink dotted line indicates sham levels. ( d ) Linear regression showing associations between the proportion of CFTR + cells in lung tissue and pulmonary S1P levels. ( e ) Linear regression showing associations between the MFI of CFTR + cells in lung tissue and pulmonary S1P levels. Data are expressed as mean +- SEM. In ( a , b ), one-way ANOVA with Tukey's post hoc testing where * denotes p
 Explore
Explore Validate
Validate Learn
Learn