MA1-215
antibody from Invitrogen Antibodies
Targeting: THRB
ERBA-BETA, ERBA2, GRTH, NR1A2, PRTH, THR1, THRB1, THRB2
Antibody data
- Antibody Data
- Antigen structure
- References [19]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [2]
- Immunohistochemistry [4]
- Other assay [16]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-215 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- THRA/THRB Monoclonal Antibody (C3)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- MA1-215 detects thyroid hormone receptor (TR) from human tissues. This antibody recognizes TR beta-1 and also cross-reacts with TR alpha-1.
- Antibody clone number
- C3
- Concentration
- Conc. Not Determined
Submitted references Extracellular histones, a new class of inhibitory molecules of CNS axonal regeneration.
Early life variations in temperature exposure affect the epigenetic regulation of the paraventricular nucleus in female rat pups.
The protective variant rs7173049 at LOXL1 locus impacts on retinoic acid signaling pathway in pseudoexfoliation syndrome.
MKK6 controls T3-mediated browning of white adipose tissue.
Reversing thyroid-hormone-mediated repression of a HSV-1 promoter via computationally guided mutagenesis.
Thyroid hormone-dependent epigenetic suppression of herpes simplex virus-1 gene expression and viral replication in differentiated neuroendocrine cells.
The p160 coactivator PAS-B motif stabilizes nuclear receptor binding and contributes to isoform-specific regulation by thyroid hormone receptors.
Inhibition of apoptotic potency by ligand stimulated thyroid hormone receptors located in mitochondria.
The rat thyroid hormone receptor (TR) Deltabeta3 displays cell-, TR isoform-, and thyroid hormone response element-specific actions.
Rapid signaling at the plasma membrane by a nuclear receptor for thyroid hormone.
Alternative mRNA splicing of SMRT creates functional diversity by generating corepressor isoforms with different affinities for different nuclear receptors.
Thyroid-hormone-dependent negative regulation of thyrotropin beta gene by thyroid hormone receptors: study with a new experimental system using CV1 cells.
Multiple messenger ribonucleic acid variants regulate cell-specific expression of human thyroid hormone receptor beta1.
Nontranscriptional modulation of intracellular Ca2+ signaling by ligand stimulated thyroid hormone receptor.
Thyroid hormone receptor DNA binding is required for both positive and negative gene regulation.
Thyroid hormone action in the absence of thyroid hormone receptor DNA-binding in vivo.
Ligand-dependent degradation of retinoid X receptors does not require transcriptional activity or coactivator interactions.
Cloning and characterization of two novel thyroid hormone receptor beta isoforms.
Conformational changes of human beta 1 thyroid hormone receptor induced by binding of 3,3',5-triiodo-L-thyronine.
Siddiq MM, Hannila SS, Zorina Y, Nikulina E, Rabinovich V, Hou J, Huq R, Richman EL, Tolentino RE, Hansen J, Velenosi A, Kwon BK, Tsirka SE, Maze I, Sebra R, Beaumont KG, Toro CA, Cardozo CP, Iyengar R, Filbin MT
Brain communications 2021;3(4):fcab271
Brain communications 2021;3(4):fcab271
Early life variations in temperature exposure affect the epigenetic regulation of the paraventricular nucleus in female rat pups.
Lauby SC, McGowan PO
Proceedings. Biological sciences 2020 Oct 28;287(1937):20201991
Proceedings. Biological sciences 2020 Oct 28;287(1937):20201991
The protective variant rs7173049 at LOXL1 locus impacts on retinoic acid signaling pathway in pseudoexfoliation syndrome.
Berner D, Hoja U, Zenkel M, Ross JJ, Uebe S, Paoli D, Frezzotti P, Rautenbach RM, Ziskind A, Williams SE, Carmichael TR, Ramsay M, Topouzis F, Chatzikyriakidou A, Lambropoulos A, Sundaresan P, Ayub H, Akhtar F, Qamar R, Zenteno JC, Cruz-Aguilar M, Astakhov YS, Dubina M, Wiggs J, Ozaki M, Kruse FE, Aung T, Reis A, Khor CC, Pasutto F, Schlötzer-Schrehardt U
Human molecular genetics 2019 Aug 1;28(15):2531-2548
Human molecular genetics 2019 Aug 1;28(15):2531-2548
MKK6 controls T3-mediated browning of white adipose tissue.
Matesanz N, Bernardo E, Acín-Pérez R, Manieri E, Pérez-Sieira S, Hernández-Cosido L, Montalvo-Romeral V, Mora A, Rodríguez E, Leiva-Vega L, Lechuga-Vieco AV, Ruiz-Cabello J, Torres JL, Crespo-Ruiz M, Centeno F, Álvarez CV, Marcos M, Enríquez JA, Nogueiras R, Sabio G
Nature communications 2017 Oct 11;8(1):856
Nature communications 2017 Oct 11;8(1):856
Reversing thyroid-hormone-mediated repression of a HSV-1 promoter via computationally guided mutagenesis.
Figliozzi RW, Chen F, Hsia SV
Journal of cell science 2017 Nov 1;130(21):3740-3748
Journal of cell science 2017 Nov 1;130(21):3740-3748
Thyroid hormone-dependent epigenetic suppression of herpes simplex virus-1 gene expression and viral replication in differentiated neuroendocrine cells.
Figliozzi RW, Chen F, Balish M, Ajavon A, Hsia SV
Journal of the neurological sciences 2014 Nov 15;346(1-2):164-73
Journal of the neurological sciences 2014 Nov 15;346(1-2):164-73
The p160 coactivator PAS-B motif stabilizes nuclear receptor binding and contributes to isoform-specific regulation by thyroid hormone receptors.
Privalsky ML, Lee S, Hahm JB, Young BM, Fong RN, Chan IH
The Journal of biological chemistry 2009 Jul 17;284(29):19554-63
The Journal of biological chemistry 2009 Jul 17;284(29):19554-63
Inhibition of apoptotic potency by ligand stimulated thyroid hormone receptors located in mitochondria.
Saelim N, Holstein D, Chocron ES, Camacho P, Lechleiter JD
Apoptosis : an international journal on programmed cell death 2007 Oct;12(10):1781-94
Apoptosis : an international journal on programmed cell death 2007 Oct;12(10):1781-94
The rat thyroid hormone receptor (TR) Deltabeta3 displays cell-, TR isoform-, and thyroid hormone response element-specific actions.
Harvey CB, Bassett JH, Maruvada P, Yen PM, Williams GR
Endocrinology 2007 Apr;148(4):1764-73
Endocrinology 2007 Apr;148(4):1764-73
Rapid signaling at the plasma membrane by a nuclear receptor for thyroid hormone.
Storey NM, Gentile S, Ullah H, Russo A, Muessel M, Erxleben C, Armstrong DL
Proceedings of the National Academy of Sciences of the United States of America 2006 Mar 28;103(13):5197-201
Proceedings of the National Academy of Sciences of the United States of America 2006 Mar 28;103(13):5197-201
Alternative mRNA splicing of SMRT creates functional diversity by generating corepressor isoforms with different affinities for different nuclear receptors.
Goodson ML, Jonas BA, Privalsky ML
The Journal of biological chemistry 2005 Mar 4;280(9):7493-503
The Journal of biological chemistry 2005 Mar 4;280(9):7493-503
Thyroid-hormone-dependent negative regulation of thyrotropin beta gene by thyroid hormone receptors: study with a new experimental system using CV1 cells.
Nakano K, Matsushita A, Sasaki S, Misawa H, Nishiyama K, Kashiwabara Y, Nakamura H
The Biochemical journal 2004 Mar 1;378(Pt 2):549-57
The Biochemical journal 2004 Mar 1;378(Pt 2):549-57
Multiple messenger ribonucleic acid variants regulate cell-specific expression of human thyroid hormone receptor beta1.
Frankton S, Harvey CB, Gleason LM, Fadel A, Williams GR
Molecular endocrinology (Baltimore, Md.) 2004 Jul;18(7):1631-42
Molecular endocrinology (Baltimore, Md.) 2004 Jul;18(7):1631-42
Nontranscriptional modulation of intracellular Ca2+ signaling by ligand stimulated thyroid hormone receptor.
Saelim N, John LM, Wu J, Park JS, Bai Y, Camacho P, Lechleiter JD
The Journal of cell biology 2004 Dec 6;167(5):915-24
The Journal of cell biology 2004 Dec 6;167(5):915-24
Thyroid hormone receptor DNA binding is required for both positive and negative gene regulation.
Shibusawa N, Hollenberg AN, Wondisford FE
The Journal of biological chemistry 2003 Jan 10;278(2):732-8
The Journal of biological chemistry 2003 Jan 10;278(2):732-8
Thyroid hormone action in the absence of thyroid hormone receptor DNA-binding in vivo.
Shibusawa N, Hashimoto K, Nikrodhanond AA, Liberman MC, Applebury ML, Liao XH, Robbins JT, Refetoff S, Cohen RN, Wondisford FE
The Journal of clinical investigation 2003 Aug;112(4):588-97
The Journal of clinical investigation 2003 Aug;112(4):588-97
Ligand-dependent degradation of retinoid X receptors does not require transcriptional activity or coactivator interactions.
Osburn DL, Shao G, Seidel HM, Schulman IG
Molecular and cellular biology 2001 Aug;21(15):4909-18
Molecular and cellular biology 2001 Aug;21(15):4909-18
Cloning and characterization of two novel thyroid hormone receptor beta isoforms.
Williams GR
Molecular and cellular biology 2000 Nov;20(22):8329-42
Molecular and cellular biology 2000 Nov;20(22):8329-42
Conformational changes of human beta 1 thyroid hormone receptor induced by binding of 3,3',5-triiodo-L-thyronine.
Bhat MK, Parkison C, McPhie P, Liang CM, Cheng SY
Biochemical and biophysical research communications 1993 Aug 31;195(1):385-92
Biochemical and biophysical research communications 1993 Aug 31;195(1):385-92
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot of human thyroid hormone receptor using Product # MA1-215.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Thyroid Hormone Receptor in HeLa Cells. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with a Thyroid Hormone Receptor monoclonal antibody (Product # MA1-215) at a dilution of 1:20 overnight at 4 C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35503). Thyroid Hormone Receptor staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Thyroid Hormone Receptor in HepG2 Cells. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with a Thyroid Hormone Receptor monoclonal antibody (Product # MA1-215) at a dilution of 1:20 overnight at 4 C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35503). Thyroid Hormone Receptor staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Images were taken at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Human thyroid tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature and probed with a Thyroid Hormone Receptor monoclonal antibody (Product # MA1-215) at a dilution of 1:20 or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Thyroid Hormone Receptor showing staining in the nucleus and weak staining in the cytoplasm of paraffin-embedded human colon tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Thyroid Hormone Receptor Mouse Monoclonal Antibody (Product # MA1-215) diluted in 3% BSA-PBS at a dilution of 1:20 for 1 hour at 37ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Thyroid Hormone Receptor showing staining in the cytoplasm and weak staining in the nucleus of paraffin-embedded human thyroid tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Thyroid Hormone Receptor Mouse Monoclonal Antibody (Product # MA1-215) diluted in 3% BSA-PBS at a dilution of 1:20 for 1 hour at 37ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Thyroid Hormone Receptor showing staining in the cytoplasm and weak staining in the nucleus of paraffin-embedded mouse thyroid tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Thyroid Hormone Receptor Mouse Monoclonal Antibody (Product # MA1-215) diluted in 3% BSA-PBS at a dilution of 1:20 for 1 hour at 37ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. T 3 -bound TR beta A1 increases IP 3 -induced Ca 2 + wave period. (a) Spatial-temporal stacks of IP 3 (~300 nM)-induced Ca 2+ wave activity in a representative control (water injected) oocyte, a T 3 -treated (100 nM) oocyte expressing TR beta A1 and a T 3 (100 nM) treated oocyte. Each image is 745 x 745 mum. (b) Western blot showing expression of TR beta A1. Protein extracts from all groups were collected and loaded at 0.5 oocytes per lane onto 10% SDS-PAGE. The membrane was probed with a monoclonal mouse anti-human TRs antibody (MA1-215) and labeled with an HRP-conjugated secondary antibody. (c) Histogram of average interwave period for each group of oocytes. n values in parentheses represent the total number of oocytes pooled from at least two frogs. Error bars correspond to the mean +- SEM. The asterisks (**) denote a statistic significant difference (ANOVA single factor, P < 0.0001).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. Transcriptional activity of TR beta A1 requires xRXR alpha and both cognate ligands. Transcriptional activity was monitored with the TRE-reporter vector, pSEAP (TRE) . (a) Lanes 1 and 2 are negative (pSEAP (-ve) ) and positive (pSEAP (+ve) ) vector controls. Oocytes expressing TR beta A1 or TR beta A1 plus xRXR alpha were incubated with 100 nM T 3 (lanes 3-5) plus 100 nM RA (lane 5) for 3 d. Cytosolic extracts from each group of oocytes was prepared and loaded onto a 10% SDS-PAGE at 2.5 oocytes equivalents per lane. SEAP was detected with the polyclonal rabbit anti-human SEAP antibody and an HRP-conjugated secondary antibody. The SP labeled arrow indicates SEAP immunoreactivity, which was present only in oocytes expressing TR beta A1 and xRXR alpha exposed to both T 3 and RA. (b) Transcriptional activity of TR beta A1 requires the pBOX within the DBD and the NLS. Oocytes expressing xTR beta A1DeltapBox-NLS and xRXR alpha show no SEAP immunoreactivity when incubated with T 3 (lane 6) or T 3 plus RA (lane 7). Western blot analysis shows that xRXR alpha , TR beta A1, and xTR beta A1DeltapBox-NLS are expressed at comparable levels (Western blots below lanes 4-7). TR beta A1 and xTR beta A1DeltapBox-NLS were detected with the monoclonal mouse anti-human TRs antibody (MA1-215). xRXR alpha was detected with a polyclonal rabbit anti-human RXR antibody (Sc-774).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Acute modulation of Ca 2 + signaling does not require heterodimerization of TR beta A1 with xRXR alpha . (a) Spatial-temporal stacks of IP 3 -induced Ca 2+ wave activity in control oocytes compared with oocytes expressing TR beta A1 or TR beta A1 with xRXR alpha . T 3 (100 nM) and RA (100 nM) were added as indicated 10-15 min before injection with IP 3 (~300 nM). Scale is the same as Fig. 1 . (b) Western blots of oocytes expressing TR beta A1 and xRXR alpha . Primary and secondary antibodies were identical to those used in Figs. 1 and 2 . (c). Histogram of average interwave period (second) of each group of oocytes. The asterisks (**) denote a statistic significance using ANOVA single factor (P < 0.0001). Values in parentheses represent the number of oocytes.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. The pBOX and NLSs of TR beta A1 are not required for the acute regulation of Ca 2 + signaling. (a) Schematic figure depicting the position of the pBOX deletion in the DBD and the NLS modification within TR beta A1. (b) Spatial-temporal stack of IP 3 -induced Ca 2+ wave activity in control oocytes compared with oocytes expressing TR mutants DeltapBox-NLS and DeltaNLS. Oocytes expressing the TR mutants were incubated with T 3 (100 nM) 10-15 min before IP 3 (~300 nM) injections. (c) Western blot analysis confirming comparable levels of protein expression for both wild-type and mutant TR beta A1. (d) Histogram of the average Ca 2+ wave periods for each group of oocytes ( n values are in parentheses). Statistic significance over control oocytes is indicated by the asterisks (**; ANOVA single factor, P < 0.0001).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
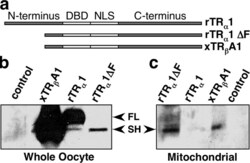
- Figure 8. Xeno pus TR beta A1 and NH 2 -terminal truncated rat TR alpha 1 (rTR alpha 1DeltaF) localize to mitochondria. (a) Schematic diagram of TRs showing that rTR alpha 1DeltaF and xTR beta A1 have a similar NH 2 terminus. (b and c) Western blots of TR alpha 1, rTR alpha 1DeltaF, and xTR beta A1 expression in whole oocytes and mitochondrial extracts respectively. FL, full-length receptor; SH, shortened form of the receptor. Extracts were prepared from 300 oocytes in each group. All oocytes were exposed to 100 nM T 3 for at least 15 min before organelle extraction. TRs were immunoprecipitated with a monoclonal mouse anti-human TRs antibody (MA1-215), captured with immobilized protein G, concentrated, and loaded onto a 10% SDS-PAGE. An HRP-conjugated secondary antibody was used for visualization.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Mkk6 -/- white adipose tissue is predisposed to T3-induced browning. a Activation of p38 and AMPK pathways in epididymal white fat from HFD-fed WT and Mkk6 -/- mice. Immunoblot analysis with the indicated antibodies was performed in extracts from mice starved overnight. b Immunoblot analysis of in vitro differentiated WT and Mkk6 -/- adipocytes. Representative from three different experiments done by duplicate. c Mkk6 -/ pre-adipocytes were infected with shRNA against AMPK, TAK1, TAB1, or scramble as a control. Activation of p38 was assayed by immunoblot of p38 and ATF2 phosphorylation. Representative from three different experiments. d UCP1 protein expression in in vitro differentiated WT and Mkk6 -/- adipocytes. Representative from three different experiments done by duplicate. e Mkk6 -/- pre-adipocytes were infected with shRNA against p38alpha or scramble as a control, and then differentiated to adipocytes. UCP1 levels were assayed by immunoblot n = 4. f Mitochondrial spare respiratory capacity (SRC) was assessed by Seahorse assay in primary WT and Mkk6 -/- adipocytes incubated with glucose oxidation (GO) or fatty acid oxidation (FAO) medium (mean +- SEM, WT n = 22 WT or 24 Mkk6 -/- wells from three mice cultured independently). g Representative transmission electronic microscopy images of mitochondria in eWAT from WT and Mkk6 -/- HFD-fed mice ( n = 3 mice). Scale bar: 500 nm. h Immunoblot analysis of UCP1 protein in in vitro differentiated WT and Mkk6 -/- adipocyte
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot