Antibody data
- Antibody Data
- Antigen structure
- References [36]
- Comments [0]
- Validations
- Immunohistochemistry [2]
- Flow cytometry [1]
- Other assay [16]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 11-9459-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD45 Monoclonal Antibody (2D1), FITC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 2D1 monoclonal antibody reacts with all isoforms of human CD45, also known as Leukocyte Common Antigen (LCA). CD45 is expressed by all hematopoietic cells excluding circulating erythrocytes and platelets. The cytoplasmic portion of CD45 has tyrosine phosphatase enzymatic activity and plays an important role in activation of lymphocytes. Applications Reported: This 2D1 antibody has been reported for use in flow cytometric analysis. Applications Tested: This 2D1 antibody has been pre-titrated and tested by flow cytometric analysis of human peripheral blood leukocytes. This can be used at 5 µL (1.0 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488 nm; Emission: 520 nm; Laser: Blue Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Green dye
- Isotype
- IgG
- Antibody clone number
- 2D1
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Human serum albumin promotes self-renewal and expansion of umbilical cord blood CD34(+) hematopoietic stem/progenitor cells.
LIM Mineralization Protein-1 Enhances the Committed Differentiation of Dental Pulp Stem Cells through the ERK1/2 and p38 MAPK Pathways and BMP Signaling.
Cryopreserved PM21-Particle-Expanded Natural Killer Cells Maintain Cytotoxicity and Effector Functions In Vitro and In Vivo.
Immune composition and its association with hematologic recovery after chemotherapeutic injury in acute myeloid leukemia.
Plasma Extracellular Vesicle Subtypes May be Useful as Potential Biomarkers of Immune Activation in People With HIV.
Low-Level Laser Irradiation Promotes Proliferation and Differentiation on Apical Papilla Stem Cells.
Bcl-xL mutant promotes cartilage differentiation of BMSCs by upregulating TGF-β/BMP expression levels.
VAP-PLGA microspheres (VAP-PLGA) promote adipose-derived stem cells (ADSCs)-induced wound healing in chronic skin ulcers in mice via PI3K/Akt/HIF-1α pathway.
Down-Regulated Exosomal MicroRNA-221 - 3p Derived From Senescent Mesenchymal Stem Cells Impairs Heart Repair.
Neutralizing Antibodies Protect against Oral Transmission of Lymphocryptovirus.
Single residue in CD28-costimulated CAR-T cells limits long-term persistence and antitumor durability.
Elucidating the fundamental fibrotic processes driving abdominal adhesion formation.
Sodium Selenite Improves The Therapeutic Effect Of BMSCs Via Promoting The Proliferation And Differentiation, Thereby Promoting The Hematopoietic Factors.
TGF‑β induces periodontal ligament stem cell senescence through increase of ROS production.
High-yield isolation of menstrual blood-derived endometrial stem cells by direct red blood cell lysis treatment.
Targeting enhancer switching overcomes non-genetic drug resistance in acute myeloid leukaemia.
Transcriptomic Profiling of the Tumor Microenvironment Reveals Distinct Subgroups of Clear Cell Renal Cell Cancer: Data from a Randomized Phase III Trial.
Microglia innately develop within cerebral organoids.
Bone marrow niche trafficking of miR-126 controls the self-renewal of leukemia stem cells in chronic myelogenous leukemia.
BNIP3/Bcl-2-mediated apoptosis induced by cyclic tensile stretch in human cartilage endplate-derived stem cells.
Isolation and characterization of stem cells from differentially degenerated human lumbar zygapophyseal articular cartilage.
A fully defined static suspension culture system for large-scale human embryonic stem cell production.
Similarities and differences between helminth parasites and cancer cell lines in shaping human monocytes: Insights into parallel mechanisms of immune evasion.
Regulatory Innate Lymphoid Cells Control Innate Intestinal Inflammation.
Region and cell-type resolved quantitative proteomic map of the human heart.
Increased expression of triggering receptor expressed on myeloid cells-1 in the population with obesity and insulin resistance.
A Member of the Nuclear Receptor Superfamily, Designated as NR2F2, Supports the Self-Renewal Capacity and Pluripotency of Human Bone Marrow-Derived Mesenchymal Stem Cells.
Tumor immune microenvironment characterization in clear cell renal cell carcinoma identifies prognostic and immunotherapeutically relevant messenger RNA signatures.
NR2F2 regulates bone marrow-derived mesenchymal stem cell-promoted proliferation of Reh cells.
MIF Plays a Key Role in Regulating Tissue-Specific Chondro-Osteogenic Differentiation Fate of Human Cartilage Endplate Stem Cells under Hypoxia.
Vitamin D machinery and metabolism in porcine adipose-derived mesenchymal stem cells.
Human adipose-derived stem cells partially rescue the stroke syndromes by promoting spatial learning and memory in mouse middle cerebral artery occlusion model.
Vascular niche promotes hematopoietic multipotent progenitor formation from pluripotent stem cells.
Human and mouse skeletal muscle stem cells: convergent and divergent mechanisms of myogenesis.
Epithelial cell differentiation of human mesenchymal stromal cells in decellularized lung scaffolds.
Differentiation of human adipose-derived stem cells into neuron-like cells which are compatible with photocurable three-dimensional scaffolds.
Hua J, Jiao T, Qiao Y, Zhang S, Xiao T, Zhang Y, Yan J
Annals of translational medicine 2023 Jan 31;11(2):62
Annals of translational medicine 2023 Jan 31;11(2):62
LIM Mineralization Protein-1 Enhances the Committed Differentiation of Dental Pulp Stem Cells through the ERK1/2 and p38 MAPK Pathways and BMP Signaling.
Mu R, Chen B, Bi B, Yu H, Liu J, Li J, He M, Rong L, Liu B, Liu K, Zhu L, Shi X, Shuai Y, Jin L
International journal of medical sciences 2022;19(8):1307-1319
International journal of medical sciences 2022;19(8):1307-1319
Cryopreserved PM21-Particle-Expanded Natural Killer Cells Maintain Cytotoxicity and Effector Functions In Vitro and In Vivo.
Oyer JL, Croom-Perez TJ, Dieffenthaller TA, Robles-Carillo LD, Gitto SB, Altomare DA, Copik AJ
Frontiers in immunology 2022;13:861681
Frontiers in immunology 2022;13:861681
Immune composition and its association with hematologic recovery after chemotherapeutic injury in acute myeloid leukemia.
Kenswil KJG, Pisterzi P, Feyen J, Ter Borg M, Rombouts E, Braakman E, Raaijmakers MHGP
Experimental hematology 2022 Jan;105:32-38.e2
Experimental hematology 2022 Jan;105:32-38.e2
Plasma Extracellular Vesicle Subtypes May be Useful as Potential Biomarkers of Immune Activation in People With HIV.
Bazié WW, Boucher J, Vitry J, Goyer B, Routy JP, Tremblay C, Trottier S, Jenabian MA, Provost P, Alary M, Gilbert C
Pathogens & immunity 2021;6(1):1-28
Pathogens & immunity 2021;6(1):1-28
Low-Level Laser Irradiation Promotes Proliferation and Differentiation on Apical Papilla Stem Cells.
Gutiérrez D, Rouabhia M, Ortiz J, Gaviria D, Alfonso C, Muñoz A, Inostroza C
Journal of lasers in medical sciences 2021;12:e75
Journal of lasers in medical sciences 2021;12:e75
Bcl-xL mutant promotes cartilage differentiation of BMSCs by upregulating TGF-β/BMP expression levels.
Xiao K, Yang L, Xie W, Gao X, Huang R, Xie M
Experimental and therapeutic medicine 2021 Jul;22(1):736
Experimental and therapeutic medicine 2021 Jul;22(1):736
VAP-PLGA microspheres (VAP-PLGA) promote adipose-derived stem cells (ADSCs)-induced wound healing in chronic skin ulcers in mice via PI3K/Akt/HIF-1α pathway.
Jiang W, Zhang J, Zhang X, Fan C, Huang J
Bioengineered 2021 Dec;12(2):10264-10284
Bioengineered 2021 Dec;12(2):10264-10284
Down-Regulated Exosomal MicroRNA-221 - 3p Derived From Senescent Mesenchymal Stem Cells Impairs Heart Repair.
Sun L, Zhu W, Zhao P, Zhang J, Lu Y, Zhu Y, Zhao W, Liu Y, Chen Q, Zhang F
Frontiers in cell and developmental biology 2020;8:263
Frontiers in cell and developmental biology 2020;8:263
Neutralizing Antibodies Protect against Oral Transmission of Lymphocryptovirus.
Singh S, Homad LJ, Akins NR, Stoffers CM, Lackhar S, Malhi H, Wan YH, Rawlings DJ, McGuire AT
Cell reports. Medicine 2020 Jun 23;1(3)
Cell reports. Medicine 2020 Jun 23;1(3)
Single residue in CD28-costimulated CAR-T cells limits long-term persistence and antitumor durability.
Guedan S, Madar A, Casado-Medrano V, Shaw C, Wing A, Liu F, Young RM, June CH, Posey AD Jr
The Journal of clinical investigation 2020 Jun 1;130(6):3087-3097
The Journal of clinical investigation 2020 Jun 1;130(6):3087-3097
Elucidating the fundamental fibrotic processes driving abdominal adhesion formation.
Foster DS, Marshall CD, Gulati GS, Chinta MS, Nguyen A, Salhotra A, Jones RE, Burcham A, Lerbs T, Cui L, King ME, Titan AL, Ransom RC, Manjunath A, Hu MS, Blackshear CP, Mascharak S, Moore AL, Norton JA, Kin CJ, Shelton AA, Januszyk M, Gurtner GC, Wernig G, Longaker MT
Nature communications 2020 Aug 13;11(1):4061
Nature communications 2020 Aug 13;11(1):4061
Sodium Selenite Improves The Therapeutic Effect Of BMSCs Via Promoting The Proliferation And Differentiation, Thereby Promoting The Hematopoietic Factors.
Yan D, Tang B, Yan L, Zhang L, Miao M, Chen X, Sui G, Zhang Q, Liu D, Wang H
OncoTargets and therapy 2019;12:9685-9696
OncoTargets and therapy 2019;12:9685-9696
TGF‑β induces periodontal ligament stem cell senescence through increase of ROS production.
Fan C, Ji Q, Zhang C, Xu S, Sun H, Li Z
Molecular medicine reports 2019 Oct;20(4):3123-3130
Molecular medicine reports 2019 Oct;20(4):3123-3130
High-yield isolation of menstrual blood-derived endometrial stem cells by direct red blood cell lysis treatment.
Sun Y, Ren Y, Yang F, He Y, Liang S, Guan L, Cheng F, Liu Y, Lin J
Biology open 2019 May 2;8(5)
Biology open 2019 May 2;8(5)
Targeting enhancer switching overcomes non-genetic drug resistance in acute myeloid leukaemia.
Bell CC, Fennell KA, Chan YC, Rambow F, Yeung MM, Vassiliadis D, Lara L, Yeh P, Martelotto LG, Rogiers A, Kremer BE, Barbash O, Mohammad HP, Johanson TM, Burr ML, Dhar A, Karpinich N, Tian L, Tyler DS, MacPherson L, Shi J, Pinnawala N, Yew Fong C, Papenfuss AT, Grimmond SM, Dawson SJ, Allan RS, Kruger RG, Vakoc CR, Goode DL, Naik SH, Gilan O, Lam EYN, Marine JC, Prinjha RK, Dawson MA
Nature communications 2019 Jun 20;10(1):2723
Nature communications 2019 Jun 20;10(1):2723
Transcriptomic Profiling of the Tumor Microenvironment Reveals Distinct Subgroups of Clear Cell Renal Cell Cancer: Data from a Randomized Phase III Trial.
Hakimi AA, Voss MH, Kuo F, Sanchez A, Liu M, Nixon BG, Vuong L, Ostrovnaya I, Chen YB, Reuter V, Riaz N, Cheng Y, Patel P, Marker M, Reising A, Li MO, Chan TA, Motzer RJ
Cancer discovery 2019 Apr;9(4):510-525
Cancer discovery 2019 Apr;9(4):510-525
Microglia innately develop within cerebral organoids.
Ormel PR, Vieira de Sá R, van Bodegraven EJ, Karst H, Harschnitz O, Sneeboer MAM, Johansen LE, van Dijk RE, Scheefhals N, Berdenis van Berlekom A, Ribes Martínez E, Kling S, MacGillavry HD, van den Berg LH, Kahn RS, Hol EM, de Witte LD, Pasterkamp RJ
Nature communications 2018 Oct 9;9(1):4167
Nature communications 2018 Oct 9;9(1):4167
Bone marrow niche trafficking of miR-126 controls the self-renewal of leukemia stem cells in chronic myelogenous leukemia.
Zhang B, Nguyen LXT, Li L, Zhao D, Kumar B, Wu H, Lin A, Pellicano F, Hopcroft L, Su YL, Copland M, Holyoake TL, Kuo CJ, Bhatia R, Snyder DS, Ali H, Stein AS, Brewer C, Wang H, McDonald T, Swiderski P, Troadec E, Chen CC, Dorrance A, Pullarkat V, Yuan YC, Perrotti D, Carlesso N, Forman SJ, Kortylewski M, Kuo YH, Marcucci G
Nature medicine 2018 May;24(4):450-462
Nature medicine 2018 May;24(4):450-462
BNIP3/Bcl-2-mediated apoptosis induced by cyclic tensile stretch in human cartilage endplate-derived stem cells.
Yuan C, Pu L, He Z, Wang J
Experimental and therapeutic medicine 2018 Jan;15(1):235-241
Experimental and therapeutic medicine 2018 Jan;15(1):235-241
Isolation and characterization of stem cells from differentially degenerated human lumbar zygapophyseal articular cartilage.
Xiao L, Xu S, Wang X, Jin Z, Wang J, Yang B, Xu H
Molecular medicine reports 2018 Dec;18(6):5751-5759
Molecular medicine reports 2018 Dec;18(6):5751-5759
A fully defined static suspension culture system for large-scale human embryonic stem cell production.
Li X, Ma R, Gu Q, Liang L, Wang L, Zhang Y, Wang X, Liu X, Li Z, Fang J, Wu J, Wang Y, Li W, Hu B, Wang L, Zhou Q, Hao J
Cell death & disease 2018 Aug 30;9(9):892
Cell death & disease 2018 Aug 30;9(9):892
Similarities and differences between helminth parasites and cancer cell lines in shaping human monocytes: Insights into parallel mechanisms of immune evasion.
Narasimhan PB, Akabas L, Tariq S, Huda N, Bennuru S, Sabzevari H, Hofmeister R, Nutman TB, Tolouei Semnani R
PLoS neglected tropical diseases 2018 Apr;12(4):e0006404
PLoS neglected tropical diseases 2018 Apr;12(4):e0006404
Regulatory Innate Lymphoid Cells Control Innate Intestinal Inflammation.
Wang S, Xia P, Chen Y, Qu Y, Xiong Z, Ye B, Du Y, Tian Y, Yin Z, Xu Z, Fan Z
Cell 2017 Sep 21;171(1):201-216.e18
Cell 2017 Sep 21;171(1):201-216.e18
Region and cell-type resolved quantitative proteomic map of the human heart.
Doll S, Dreßen M, Geyer PE, Itzhak DN, Braun C, Doppler SA, Meier F, Deutsch MA, Lahm H, Lange R, Krane M, Mann M
Nature communications 2017 Nov 13;8(1):1469
Nature communications 2017 Nov 13;8(1):1469
Increased expression of triggering receptor expressed on myeloid cells-1 in the population with obesity and insulin resistance.
Subramanian S, Pallati PK, Rai V, Sharma P, Agrawal DK, Nandipati KC
Obesity (Silver Spring, Md.) 2017 Mar;25(3):527-538
Obesity (Silver Spring, Md.) 2017 Mar;25(3):527-538
A Member of the Nuclear Receptor Superfamily, Designated as NR2F2, Supports the Self-Renewal Capacity and Pluripotency of Human Bone Marrow-Derived Mesenchymal Stem Cells.
Zhu N, Wang H, Wang B, Wei J, Shan W, Feng J, Huang H
Stem cells international 2016;2016:5687589
Stem cells international 2016;2016:5687589
Tumor immune microenvironment characterization in clear cell renal cell carcinoma identifies prognostic and immunotherapeutically relevant messenger RNA signatures.
Şenbabaoğlu Y, Gejman RS, Winer AG, Liu M, Van Allen EM, de Velasco G, Miao D, Ostrovnaya I, Drill E, Luna A, Weinhold N, Lee W, Manley BJ, Khalil DN, Kaffenberger SD, Chen Y, Danilova L, Voss MH, Coleman JA, Russo P, Reuter VE, Chan TA, Cheng EH, Scheinberg DA, Li MO, Choueiri TK, Hsieh JJ, Sander C, Hakimi AA
Genome biology 2016 Nov 17;17(1):231
Genome biology 2016 Nov 17;17(1):231
NR2F2 regulates bone marrow-derived mesenchymal stem cell-promoted proliferation of Reh cells.
Zhu N, Wang H, Wei J, Wang B, Shan W, Lai X, Zhao Y, Yu J, Huang H
Molecular medicine reports 2016 Aug;14(2):1351-6
Molecular medicine reports 2016 Aug;14(2):1351-6
MIF Plays a Key Role in Regulating Tissue-Specific Chondro-Osteogenic Differentiation Fate of Human Cartilage Endplate Stem Cells under Hypoxia.
Yao Y, Deng Q, Song W, Zhang H, Li Y, Yang Y, Fan X, Liu M, Shang J, Sun C, Tang Y, Jin X, Liu H, Huang B, Zhou Y
Stem cell reports 2016 Aug 9;7(2):249-62
Stem cell reports 2016 Aug 9;7(2):249-62
Vitamin D machinery and metabolism in porcine adipose-derived mesenchymal stem cells.
Valle YL, Almalki SG, Agrawal DK
Stem cell research & therapy 2016 Aug 17;7(1):118
Stem cell research & therapy 2016 Aug 17;7(1):118
Human adipose-derived stem cells partially rescue the stroke syndromes by promoting spatial learning and memory in mouse middle cerebral artery occlusion model.
Zhou F, Gao S, Wang L, Sun C, Chen L, Yuan P, Zhao H, Yi Y, Qin Y, Dong Z, Cao L, Ren H, Zhu L, Li Q, Lu B, Liang A, Xu GT, Zhu H, Gao Z, Ma J, Xu J, Chen X
Stem cell research & therapy 2015 May 9;6(1):92
Stem cell research & therapy 2015 May 9;6(1):92
Vascular niche promotes hematopoietic multipotent progenitor formation from pluripotent stem cells.
Gori JL, Butler JM, Chan YY, Chandrasekaran D, Poulos MG, Ginsberg M, Nolan DJ, Elemento O, Wood BL, Adair JE, Rafii S, Kiem HP
The Journal of clinical investigation 2015 Mar 2;125(3):1243-54
The Journal of clinical investigation 2015 Mar 2;125(3):1243-54
Human and mouse skeletal muscle stem cells: convergent and divergent mechanisms of myogenesis.
Bareja A, Holt JA, Luo G, Chang C, Lin J, Hinken AC, Freudenberg JM, Kraus WE, Evans WJ, Billin AN
PloS one 2014;9(2):e90398
PloS one 2014;9(2):e90398
Epithelial cell differentiation of human mesenchymal stromal cells in decellularized lung scaffolds.
Mendez JJ, Ghaedi M, Steinbacher D, Niklason LE
Tissue engineering. Part A 2014 Jun;20(11-12):1735-46
Tissue engineering. Part A 2014 Jun;20(11-12):1735-46
Differentiation of human adipose-derived stem cells into neuron-like cells which are compatible with photocurable three-dimensional scaffolds.
Gao S, Zhao P, Lin C, Sun Y, Wang Y, Zhou Z, Yang D, Wang X, Xu H, Zhou F, Cao L, Zhou W, Ning K, Chen X, Xu J
Tissue engineering. Part A 2014 Apr;20(7-8):1271-84
Tissue engineering. Part A 2014 Apr;20(7-8):1271-84
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of CD45 was performed using formalin-fixed paraffin-embedded human tonsil tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a microwave at 100 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without CD45 Monoclonal Antibody (2D1), FITC, eBioscience™ (Product # 11-9459-42) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and deconvoluted externally.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of CD45 was performed using formalin-fixed paraffin-embedded human tonsil tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a microwave at 100 degree Celsius for 10 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without CD45 Monoclonal Antibody (2D1), FITC, eBioscience™ (Product # 11-9459-42) at 1:100 dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and deconvoluted externally.
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
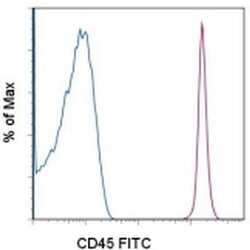
- Experimental details
- Normal human peripheral blood cells with stained with Mouse IgG1 K Isotype Control FITC (Product # 11-4714-42) (blue histogram) or Anti-Human CD45 FITC (purple histogram). Cells in the lymphocyte gate were used for analysis.
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 In silico validation of the immune cell scoring method. a In silico validation of immune cell scores using simulated mixing proportions. RNA-Seq profiles of FACS-sorted NK cells, macrophages, CD 4 + and CD 8 + T cells, and non-immune CD 45 - cells were mixed with known proportions to obtain a ""clean"" mixture. Noise was added at varying SNRs. Mixing levels were then inferred by ssGSEA from the ""clean"" and noisy mixtures. The Spearman correlations between the simulated and inferred levels ( top panel ) and the bootstrap p values for these correlation values ( bottom panel ) are shown on the y-axes (Additional file 1 : Figure S18 and "" Methods "" for the calculation of the bootstrap p values). b Validation of IIS with methylation-based leukocyte fractions. Spearman correlations between the two orthogonal scores are shown on the x-axis for 13 tumor types. c Validation of TIS with TCR beta chain abundance. Both scores are computationally inferred from RNA-Seq data but employ different approaches to measure T cell levels. Spearman correlations are shown on the x-axis for 19 tumor types
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. EV quantification and cell origin in association with HIV. Purified plasma EVs fromuninfected (Control, n = 8) and HIV+ patients (n = 17) were stained with the lipophilic fluorescent tracer dye DiD to count total vesicles ( Figure 2A and B ), then labeled with antibodies directed against receptors CD45 ( Figure 2C and D ), CD4 ( Figure 2E and F ), and CD8 ( Figure 2 G and H ) to evaluate cell origin by cytofluorometry. Asterisks denote significant difference (** P
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Experimental diagram (A) , hADSC surface antigen profiling with FACS showed that hADSCs positively express CD29, CD44, CD73 and CD105, negatively express CD34 and CD45 (B) . FACS, flow activated cell sorting; hADSCs, human adipose-derived stem cells.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
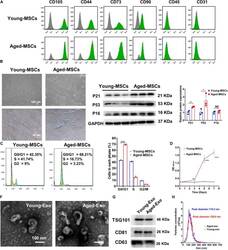
- Experimental details
- FIGURE 1 Characterization of young and aged MSCs and exosomes. (A) Surface marker profiling of young-MSCs and aged-MSCs. (B) SA-beta-Gal staining showed that senescence increased significantly in aged MSCs. (C) Representative immunoblot images and quantitative analysis of p21, p53, and p16 protein level in young and aged-MSCs. ( n = 3). (D) Quantitation of cell cycle phases by propidium iodide staining. ( n = 3). (E) The CCK-8 assay showed that aged MSCs grew more slowly than young MSCs. ( n = 6). (F) Young and aged exosomes were observed using TEM. (G) The exosome surface markers were analyzed by Western blot. (H) Nanoparticle tracking analysis was used to analyze the particle size and concentration of Young-Exo and Aged-Exo. * p < 0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001; NS, not significant.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Culture and identification of bone marrow mesenchymal stem cells. (A) Light microscopy of BMSCs (scale bar=100 um for the left image and 200 um for the right image). (B) Percentage of CD34-, CD45-, CD73-, CD90- and CD105-positive BMSCs were detected by flow cytometry. BMSC, bone marrow mesenchymal stem cell.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
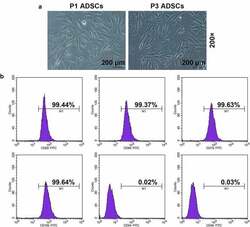
- Experimental details
- Figure 1. Morphology and immune phenotype of adipose-derived stem cells (ADSCs) were identified by morphological observation and flow cytometry. (a) Morphology of the primary (P1) and third passage (P3) of ADSCs. Images were acquired at 200x magnification. (b) Immune phenotype of ADSCs. The average data from three independent experiments were shown as mean +- standard deviation
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 oMG expressed microglia-characteristic cell surface markers and showed similar functional immune and phagocytic properties as adult MG. a Flow cytometric analyses of the expression pattern of microglial extracellular markers on CD11b+-gated oMG (oMG 1, 3, and 5) compared to adult MG derived from three separate brain regions from adult MG1.1. (eight organoids were pooled per donor (oMG 1, 3, and 5) after 52 days in culture). b Morphology of magnetic automated cell sorted CD11b+ oMG 1 and adult MG in bright field microscope after 1 week in culture. Scale bar 40 mum. c mRNA expression, determined by qRT-PCR, of pro-inflammatory cytokines IL6 and IL1B after 6 h stimulation with LPS was significantly higher in oMG compared to adult MG (Mann-Whitney test IL6 and IL1B: U = 0, n = 4, p = 0.03). LPS-stimulated response relative to control condition without LPS. ( n = 4 experiments, eight organoids pooled per experiment; adult MG1.1) (* p < 0.05). d Anti-inflammatory response of oMG and adult MG was compared by qRT-PCR for expression of anti-inflammatory genes CD163 and MRC1 upon 72 h stimulation with dexamethasone. Dexamethasone-stimulated response relative to control condition without dexamethasone. (oMG, n = 3 separate experiments in which oMG were isolated from > 4 pooled cerebral organoids from iPSC 1 per experiment; adult MG, n = 4). e Phagocytosis capacity was tested oMG 1 and adult MG by performing a phagocytosis assay with iC3b-coated green-yellow
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 01 Figure S1 Isotype control for TREM-1 and TREM-2 expression in monocytes and neutrophils of the study subjects. Cell phenotype was performed using anti-human antibodies and isotypes for monocytes (CD45+, CD14+, HLA-DR+) represented in figure A and neutrophils (CD45+, CD16+) in figure B. A) The representative figures for isotype control for monocytes gating; live monocytes were gated from FSC/SSC gating. Then CD45+ were gated using the basal level of CD45 Isotype and analyzed for CD14 and HLA-DR expression with respective CD14 and HLA-DR Isotypes. The double positive populations (CD14+ HLA-DR+) were further gated for expression of TREM-1+ and TREM-2+ using the basal level of TREM-1 and TREM-2 Isotypes. B) The representative figures for Isotype control for neutrophils gating; live granulocytes were gated from FSC/SSC gating. Then CD45+ were gated using the basal level of CD45 Isotype and analyzed for CD16 expression using the basal level of CD16 Isotype. CD16+ populations were further gated for expression of TREM-1 and TREM-2 using the basal level of TREM-1 and TREM-2 Isotypes. Figure S2 Hematoxylin and Eosin staining for fatty liver grading and inflammation. H & E in biopsy samples of SO - D - , SO + D - and SO + D + groups respectively in liver (images Aa, Ab & Ac), omental fat (images Ba, Bb & Bc) and subcutaneous fat (images Ca, Cb & Cc). Liver biopsy samples showed steatosis in SO + D - and fibrosis and cirrhosis in SO + D + groups. Size of the adipocyte was larger in su
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Characterization of PDLSCs. (A) PDL cell clusters exhibited radiating or whirlpool-like morphology. The central structure in this image is a fragment of PDL tissue. Scale bar, 200 mum (B) CD146 + PDLSCs were small, round, fusiform and triangular. Scale bar, 100 mum. (C) PDLSCs were positive for the stem cell markers CD44, CD90 and CD105, but negative for CD34 and CD45, as detected by flow cytometry. PDL, periodontal ligament; PDLSCs, PDL stem cells.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 3 Cancer cell lines and mf significantly upregulates the cell surface expressions of PDL1, PDL2, CD206 and VCAM-1 on human monocytes. Human monocytes were cultured in media alone, or with CMFDA-labeled three different cancer cell lines (MDA, OVCAR, U87), or with live mf of Brugia malayi for 48hr. Cells were harvested and cell surface expression PDL1, PDL2, CD206, VCAM-1, and CD163 was measured using flow cytometry gated on CD45 + /CMFDA - monocytes. (A) One representative set (n = 15) of flow histograms demonstrating cell surface expression in unexposed human monocytes and after exposure to mf or different cancer cell lines. (B). The data are expressed as the geometric mean with 95% confidence interval of the mean fluorescent intensity of unexposed and exposed monocytes ( n = 15). * P< 0.05, ** P< 0.005.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Comparison of MSCs derived from 2D-hESCs and 3D-hESCs. a The morphology of 2D- and 3D-hESC-MSCs at different stages of differentiation. b Flow cytometry analysis revealed specific MSC surface markers (CD44, CD29, and CD105) with negative controls (CD34, CD19, and CD45) in 2D- and 3D-hESC-MSCs. c Immunostaining of differentiated 3D-hESC-MSCs expressing an adipocyte marker (FABP-4), osteocytes maker (osteocalcin), and chondrocytes marker (aggrecan)
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3. The phenotype of MenSCs. (A,B) To determine the immunophenotype of MenSCs, P3 MenSCs-DGC ( n =5) and MenSCs-RLB ( n =5) were stained by corresponding conjugated antibodies and analyzed by FACS. Both MenSCs-DGC and MenSCs-RLB positively expressed classical ASCs' markers (CD29, CD44, CD73, CD90 and CD105) and HLA-ABC; they did not express hematopoietic stem cell markers (CD34 and CD45) and HLA-DR. (C) The quantification of flow cytometry results of A and B.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Identification of BMSCs in mice. Morphological observations at different magnifications (40x, 100x, and 200x) showed that the bone marrow stem cells (BMSCs) isolated from mice were homogeneously elongated ( A ). Various cell surface markers of BMSCs were detected on the cells, including CD90, CD73, CD105, CD44, and CD45 ( B ). The values are from triplicate determinations. A P value less than 0.05 was considered to be statistically significant. ***Indicates a P value less than 0.001.
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Immunohistochemistry
Immunohistochemistry