Antibody data
- Antibody Data
- Antigen structure
- References [2]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Flow cytometry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 701257 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- P-Selectin Recombinant Rabbit Monoclonal Antibody (3H20L10)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Antibody clone number
- 3H20L10
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
Submitted references Carbon nano-onion-mediated dual targeting of P-selectin and P-glycoprotein to overcome cancer drug resistance.
The cancer glycocalyx mediates intravascular adhesion and extravasation during metastatic dissemination.
Wang H, Liang Y, Yin Y, Zhang J, Su W, White AM, Bin Jiang, Xu J, Zhang Y, Stewart S, Lu X, He X
Nature communications 2021 Jan 12;12(1):312
Nature communications 2021 Jan 12;12(1):312
The cancer glycocalyx mediates intravascular adhesion and extravasation during metastatic dissemination.
Offeddu GS, Hajal C, Foley CR, Wan Z, Ibrahim L, Coughlin MF, Kamm RD
Communications biology 2021 Feb 26;4(1):255
Communications biology 2021 Feb 26;4(1):255
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of P-selectin in Jurkat whole cell extracts using a P-selectin recombinant rabbit monoclonal antibody (Product # 701257) at a dilution of 1 µg/mL. Samples were detected using chemiluminescence (ECL). Results show a band at ~140kDa.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of P-selectin/CD62P was performed by loading 30 µg of Jurkat and K562 cell lysates using Novex®NuPAGE® 4-12% Bis-Tris gel (Product # NP0321BOX), XCell SureLock Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Proteins were transferred to a PVDF membrane and blocked with 5% skim milk for 1 hour at room temperature. P-selectin/CD62P was detected at ~140 kDa using P-selectin/CD62P Recombinant Rabbit Monoclonal Antibody (Product # 701257) at a 1:1000 dilution in 2.5% skim milk at 4°C overnight on a rocking platform. Detection was performed using an HRP-conjugated Goat anti-Rabbit secondary antibody (Product # G-21234) at a 1:5000 dilution and chemiluminescent detection was performed using Pierce™ ECL Western blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
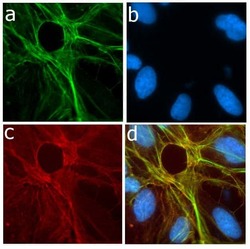
- Experimental details
- Immunofluorescent analysis of P-selectin in U2OS cells using a P-selectin recombinant rabbit monoclonal antibody (Product # 701257) followed by detection using an Alexa Fluor 488-conjugated goat anti-rabbit secondary antibody (green) (Image A). Nuclei were stained using DAPI (Image B) and actin stained with Alexa Fluor 594 phalloidin (red) (image C). Image D is a composite image showing membrane localization of P-selectin.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
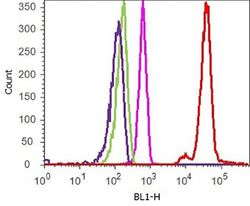
- Experimental details
- Flow cytometry analysis of P-selectin/CD62P was performed on Jurkat cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0. 25% Tritonª X-100 for 20 minutes, and blocked with 5% BSA for 1 hour at room temperature. Cells were labeled with ABfinityª P-selectin/CD62P recombinant rabbit monoclonal antibody (Product # 701257, red histogram) or with rabbit isotype control (pink histogram) at a dilution of 1:250 in 2.5% BSA. After incubation at room temperature for 3 hours, the cells were labeled with Alexa Fluor¨ 488 goat anti-Rabbit Secondary antibody (Product # A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune¨ Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
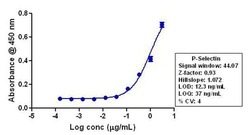
- Experimental details
- Indirect ELISA analysis of P-selectin in Jurkat lysate coated onto the plate using a P-selectin recombinant rabbit monoclonal antibody (Product # 701257) at various dilutions. A non-linear regression analysis was performed (4 PL) and LOD and LOQ for the antibody were determined.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
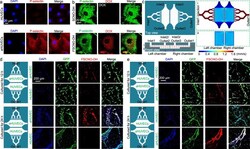
- Experimental details
- Fig. 5 P-selectin targeting capability of the FSCNO nanoparticles under both static and dynamic/microfluidic cultures. a Confocal images showing the expression of P-selectin in activated human umbilical vein endothelial cells (aHUVECs) but not in HUVECs without activation. b Confocal images of precooled aHUVECs after incubated with SCNO-DH and FSCNO-DH nanoparticles (10 mug mL -1 for DOX and 5 mug mL -1 for HM). The cells were precooled in ice for 3 h to stop its metabolic and uptake activity. c Top and side views of the microfluidic device for studying the P-selectin targeting capability of the FSCNO-DH nanoparticles under dynamic culture condition. Detached cells can be injected into device through inlet2 or inlet3 and cultured in the chambers. Medium containing nanoparticles can be injected through inlet1 and flows out the device via outlet1. Also shown are the computational modeling results of velocity distribution at the middle planes of the channels and chambers, in the top and bottom PDMS parts as indicated by the red dashed lines i and ii, respectively. The data show that the velocity of injected nanoparticle solution (inlet1) is nearly homogeneous in the two chambers. d, e Scheme and confocal images of green fluorescence protein (GFP)-expression HUVECs (in the chamber next to outlet1 for d and in the chamber next to inlet1 for e ) and aHUVECs (in the chamber next to inlet1 for d and in the chamber next to outlet1 for e ) cultured in the microfluidic device for 12 and
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA