Antibody data
- Antibody Data
- Antigen structure
- References [15]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [3]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-9897-80 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Vimentin Monoclonal Antibody (V9), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The V9 monoclonal antibody recognizes human Vimentin, a 57 kDa protein that functions as a structural component of intermediate filaments. Vimentin is expressed in cells derived from the mesenchyme but also in specific populations such as radial glia and immature glial cells, pancreatic precursor cells. It is proposed to be a marker of cardiac differentiation. In neural cells, vimentin expression is gradually replaced by neurofilaments. Reports have also shown surface expression of vimentin on activated macrophages, platelets, as well as apoptotic T cells and neutrophils. This antibody also recognizes canine (dog), rat and chicken vimentin but does not recognize mouse vimentin. Applications Reported: This V9 antibody has been reported for use in western blotting, immunocytochemistry, and immunohistochemical staining of frozen (IHC-F) and formalin-fixed paraffin embedded tissue sections (IHC-P). Applications Tested: This V9 antibody has been tested by immunocytochemistry on paraformaldehyde fixed and permeabilized SK-N-SH or C6 cell lines and by western blot on cell lysates prepared from HeLa cells. This antibody can be used at less than or equal to 1 µg/mL. It is recommended that this antibody be carefully titrated for optimal performance in the assay of interest. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Rat, Canine, Chicken/Avian
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- V9
- Vial size
- 25 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C
Submitted references Conjugation of VEGFR1/R2-targeting peptide with gold nanoparticles to enhance antiangiogenic and antitumoral activity.
Characterization of epithelial cells, connective tissue cells and immune cells in human upper airway mucosa by immunofluorescence multichannel image cytometry: a pilot study.
Induction of Epithelial-mesenchymal Transition in MDCK II Cells.
MicroRNA-384 inhibits nasopharyngeal carcinoma growth and metastasis via binding to Smad5 and suppressing the Wnt/β-catenin axis.
Nonmuscle Myosin II Activation Regulates Cell Proliferation, Cell Contraction, and Myofibroblast Differentiation in Keloid-Derived Fibroblasts.
miR-16 enhances miR-302/367-induced reprogramming and tumor suppression in breast cancer cells.
Omentin-1 is associated with atrial fibrillation in patients with cardiac valve disease.
GMP-compatible and xeno-free cultivation of mesenchymal progenitors derived from human-induced pluripotent stem cells.
Levels of hepatic Th17 cells and regulatory T cells upregulated by hepatic stellate cells in advanced HBV-related liver fibrosis.
Biphasic Squamoid Alveolar Renal Cell Carcinoma: A Distinctive Subtype of Papillary Renal Cell Carcinoma?
Interaction of TWEAK with Fn14 leads to the progression of fibrotic liver disease by directly modulating hepatic stellate cell proliferation.
Expression of intercellular adhesion molecule 1 by hepatocellular carcinoma stem cells and circulating tumor cells.
Cell surface expression of intermediate filament proteins vimentin and lamin B1 in human neutrophil spontaneous apoptosis.
Reexpression of vimentin in differentiated neuroblastoma cells enhances elongation of axonal neurites.
Species-specific recognition patterns of monoclonal antibodies directed against vimentin.
Zanjanchi P, Asghari SM, Mohabatkar H, Shourian M, Shafiee Ardestani M
Journal of nanobiotechnology 2022 Jan 4;20(1):7
Journal of nanobiotechnology 2022 Jan 4;20(1):7
Characterization of epithelial cells, connective tissue cells and immune cells in human upper airway mucosa by immunofluorescence multichannel image cytometry: a pilot study.
Giotakis AI, Dudas J, Glueckert R, Dejaco D, Ingruber J, Fleischer F, Innerhofer V, Pinggera L, Bektic-Tadic L, Gabriel SAM, Riechelmann H
Histochemistry and cell biology 2021 Mar;155(3):405-421
Histochemistry and cell biology 2021 Mar;155(3):405-421
Induction of Epithelial-mesenchymal Transition in MDCK II Cells.
Pastuła A, Lundmark R
Bio-protocol 2021 Feb 5;11(3):e3903
Bio-protocol 2021 Feb 5;11(3):e3903
MicroRNA-384 inhibits nasopharyngeal carcinoma growth and metastasis via binding to Smad5 and suppressing the Wnt/β-catenin axis.
Zeng X, Liao H, Wang F
Cytotechnology 2021 Apr;73(2):203-215
Cytotechnology 2021 Apr;73(2):203-215
Nonmuscle Myosin II Activation Regulates Cell Proliferation, Cell Contraction, and Myofibroblast Differentiation in Keloid-Derived Fibroblasts.
Lu YY, Fang CC, Hong CH, Wu CH, Lin YH, Chang KL, Lee CH
Advances in wound care 2020 Sep;9(9):491-501
Advances in wound care 2020 Sep;9(9):491-501
miR-16 enhances miR-302/367-induced reprogramming and tumor suppression in breast cancer cells.
Hoseinbeyki M, Taha MF, Javeri A
IUBMB life 2020 May;72(5):1075-1086
IUBMB life 2020 May;72(5):1075-1086
Omentin-1 is associated with atrial fibrillation in patients with cardiac valve disease.
Chen Y, Liu F, Han F, Lv L, Tang CE, Xie Z, Luo F
BMC cardiovascular disorders 2020 May 6;20(1):214
BMC cardiovascular disorders 2020 May 6;20(1):214
GMP-compatible and xeno-free cultivation of mesenchymal progenitors derived from human-induced pluripotent stem cells.
McGrath M, Tam E, Sladkova M, AlManaie A, Zimmer M, de Peppo GM
Stem cell research & therapy 2019 Jan 11;10(1):11
Stem cell research & therapy 2019 Jan 11;10(1):11
Levels of hepatic Th17 cells and regulatory T cells upregulated by hepatic stellate cells in advanced HBV-related liver fibrosis.
Li X, Su Y, Hua X, Xie C, Liu J, Huang Y, Zhou L, Zhang M, Li X, Gao Z
Journal of translational medicine 2017 Apr 11;15(1):75
Journal of translational medicine 2017 Apr 11;15(1):75
Biphasic Squamoid Alveolar Renal Cell Carcinoma: A Distinctive Subtype of Papillary Renal Cell Carcinoma?
Hes O, Condom Mundo E, Peckova K, Lopez JI, Martinek P, Vanecek T, Falconieri G, Agaimy A, Davidson W, Petersson F, Bulimbasic S, Damjanov I, Jimeno M, Ulamec M, Podhola M, Sperga M, Pane Foix M, Shelekhova K, Kalusova K, Hora M, Rotterova P, Daum O, Pivovarcikova K, Michal M
The American journal of surgical pathology 2016 May;40(5):664-75
The American journal of surgical pathology 2016 May;40(5):664-75
Interaction of TWEAK with Fn14 leads to the progression of fibrotic liver disease by directly modulating hepatic stellate cell proliferation.
Wilhelm A, Shepherd EL, Amatucci A, Munir M, Reynolds G, Humphreys E, Resheq Y, Adams DH, Hübscher S, Burkly LC, Weston CJ, Afford SC
The Journal of pathology 2016 May;239(1):109-21
The Journal of pathology 2016 May;239(1):109-21
Expression of intercellular adhesion molecule 1 by hepatocellular carcinoma stem cells and circulating tumor cells.
Liu S, Li N, Yu X, Xiao X, Cheng K, Hu J, Wang J, Zhang D, Cheng S, Liu S
Gastroenterology 2013 May;144(5):1031-1041.e10
Gastroenterology 2013 May;144(5):1031-1041.e10
Cell surface expression of intermediate filament proteins vimentin and lamin B1 in human neutrophil spontaneous apoptosis.
Moisan E, Girard D
Journal of leukocyte biology 2006 Mar;79(3):489-98
Journal of leukocyte biology 2006 Mar;79(3):489-98
Reexpression of vimentin in differentiated neuroblastoma cells enhances elongation of axonal neurites.
Dubey M, Hoda S, Chan WK, Pimenta A, Ortiz DD, Shea TB
Journal of neuroscience research 2004 Oct 15;78(2):245-9
Journal of neuroscience research 2004 Oct 15;78(2):245-9
Species-specific recognition patterns of monoclonal antibodies directed against vimentin.
Bohn W, Wiegers W, Beuttenmüller M, Traub P
Experimental cell research 1992 Jul;201(1):1-7
Experimental cell research 1992 Jul;201(1):1-7
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Vimentin Monoclonal Antibody (V9), eBioscience™ (Product # 14-9897-82) and a ~58 kDa band corresponding to VIM was observed across cell lines tested. Whole cell extracts (30 µg lysate) of HeLa (Lane 1), SH-SY5Y (Lane 2), A549 (Lane 3), A-431 (Lane 4) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1 µg/mL) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:20,000) using the iBright™ FL1500 Imaging System (Product # A44115). Chemiluminescent detection was performed using SuperSignal™ West Pico PLUS Chemiluminescent Substrate (Product # 34580).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Vimentin Monoclonal Antibody (V9), eBioscience™(Product # 14-9897-80) and a 53kDa band corresponding to Vimentin was observed across cell lines tested. Whole Cell Extract-WCL (30 µg lysate) of MDA-MB-231 (Lane 1), MCF7 (Lane 2), T-47D (Lane 3), A549 (Lane 4), HeLa (Lane 5), SH-SY5Y (Lane 6), A-431 (Lane 7) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1 µg/mL) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- CRISPR-Cas9 mediated genome editing ofVimentin (as confirmed by next generation sequencing) was achieved by using LentiArray™ Lentiviral sgRNA (Product # A32042, Assay ID CRISPR931291_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Fig (a) Western blot analysis of Vimentin was performed by loading 30 µg of HeLa Wild Type (Lane 1), HeLa Cas9 (Lane 2) and HeLa Cas9 cells transduced with Vimentin Lentiviral sgRNA (Lane 3) whole cell extracts. The samples were electrophoresed using NuPAGE™ Novex™ 4-12% Bis-Tris Protein Gel (Product # NP0322BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with Anti-Vimentin Monoclonal Antibody (V9), eBioscience™ (Product # 14-9897-82) using 1:500 dilution and Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177 1:4,000 dilution). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005). Even though NGS analysis determine the clone as partial KO, there was complete loss of signal in sgRNA transduced cells using the LentiArray™ CRISPR product line confirming that the antibody is specific to Vimentin (Fig (b)).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunocytochemistry of fixed and permeabilized C6 cells using 1 µg/mL of Mouse IgG1 Isotype Control (Product # 14-4714-82) (left) or 1 µg/mL of Anti-Vimentin (right) followed by Anti-Mouse TRITC. Nuclei are counterstained with DAPI.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of VIM was performed using 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with Vimentin Monoclonal Antibody (V9), eBioscience™ (Product # 14-9897-82, 5 µg/mL) in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766, 1:2000), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing Cytosolic localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Vimentin was performed using 70 confluent log phase MDA-MB-231 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with Vimentin Monoclonal Antibody (V9), eBioscience™ (Product # 14-9897-80) at 5 µg/mL in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing cytoskeleton localization. Panel e represents no expression in MCF-7. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Supernatants from HSC increased the percentages of Th17 cells and Tregs. a Phenotypes of primary HSC extracted from HBV-related fibrotic liver tissues in Group 2. Sections were immunostained with desmin, FAP, FSP, vimentin, fibronectin and a-SMA antibodies. One of 20 representative micrographs is shown. b , d Purified CD4+ T cells were cultured alone (Blank) or with 30% indicated supernatants. The values in the quadrants represent the percentages of Th17 cells and Tregs. The data shown are representative dot plots from more than three independent experiments. c , e The statistical analysis of the effect of LX-2 and pHSC supernatant on the percentages of Th17 cells ( c ) and Tregs ( e ). * p < 0.05, ** p < 0.01, *** p < 0.001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
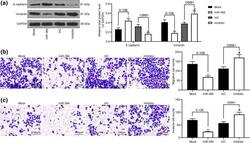
- Fig. 3 miR-384 mimic inhibits EMT, migration and invasion of NPC cells. a levels of mesenchyme marker protein Vimentin and epithelium-marker protein E-cadherin in cells measured by western blot analysis; b , c number of migrated ( b ) and invaded ( c ) cells determined by Transwell assays. Data are exhibited as mean +- SD from three independent experiments; in panel ( a ), data were analyzed using two-way ANOVA, while data in panels ( b ) and ( c ) were analyzed by one-way ANOVA, and Tukey''s multiple comparison test was used for the post-hoc test after ANOVA; * p < 0.05; ** p < 0.01 vs. the Mock group; # p < 0.05 vs. the InC group
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Omentin-1 inhibited TGF-beta1-induced endothelial-mesenchymal transition (EndMT) of human umbilical vein endothelial cells (HUVECs). Vimentin and VE-Cad protein levels in HUVECs were detected via western blotting ( a ). The mRNA levels of vimentin and VE-Cad were detected via RT-qPCR ( b ). Expression of mRNA were normalized to GAPDH. Representative images of immunofluorescence staining for vimentin ( c ) and VE-Cad ( d ) (200x magnification) in HUVECs. The p-SMAD3 and t-SMAD3 protein levels in HUVECs were detected via western blotting ( e ). ( f ) Representative images of HUVECs scratch assay (x 100 magnification). The values were mean +- SEM of three independent experiments. * P < 0.05 vs control group, ** P < 0.01 vs control group, *** P < 0.001 vs control group, + P < 0.05 vs TGF-beta1 group, ++ P < 0.01 vs TGF-beta1 group, +++ P < 0.001 vs TGF-beta1 group
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Chronic rhinosinusitis types and normal tissue; comparison of bright-field and fluorescent-immunolabeled sections. H.E. staining (a, c, e) and immunofluorescence staining (b, d, f) of control (a, b) , chronic rhinosinusitis without polyps (CRSsNP) (c, d) and chronic rhinosinusitis with polyps (CRSwNP) (e, f) tissue. Four-channel fluorescence represents nuclear DAPI staining (blue), cytokeratin (green), vimentin (red) and CD45 combined with CD18 (yellow). Scale bar: 100 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Comparison of histograms of fluorescence signals between isotype controls and patient's tissue. Histograms of fluorescence signals after control incubation in the upper histograms (isotype controls) and specific fluorescence signals after test incubation in the lower histograms (patient's tissue). Presentation of a random cell sample of all patients and their isotype controls. X -axis: mean intensity in logarithmic scale. Y -axis: cell count. The left histograms represented cytokeratin fluorescence signal, the middle histograms vimentin fluorescence signal and the right histograms CD45/CD18 fluorescence signal
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 Cytoskeletal proteins. Cell painting showing the production of the cytoskeletal proteins F-actin and vimentin in induced pluripotent stem cell-derived mesodermal progenitors (line 1013A and BC1) cultured in different media at passages 6, 10, and 15. The nuclei are stained with 4',6-diamidino-2-phenylindole (blue). Scale bar = 20 mum. Abbreviations: MP, mesodermal progenitors; FBS, fetal bovine serum; HPL, human platelet lysate; P, passage. Additional data are shown in Additional file 4 : Figure S3
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry